Abstract
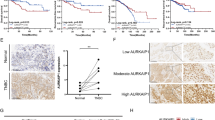
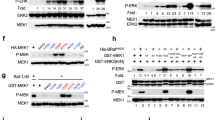
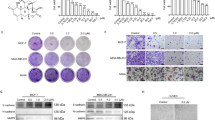
RNase III DROSHA is upregulated in multiple cancers and contributes to tumor progression by hitherto unclear mechanisms. Here, we demonstrate that DROSHA interacts with β-Catenin to transactivate STC1 in an RNA cleavage-independent manner, contributing to breast cancer stem-like cell (BCSC) properties. DROSHA mRNA stability is enhanced by N6-methyladenosine (m6A) modification which is activated by AURKA in BCSCs. AURKA stabilizes METTL14 by inhibiting its ubiquitylation and degradation to promote DROSHA mRNA methylation. Moreover, binding of AURKA to DROSHA transcript further strengthens the binding of the m6A reader IGF2BP2 to stabilize m6A-modified DROSHA. In addition, wild-type DROSHA, but not an m6A methylation-deficient mutant, enhances BCSC stemness maintenance, while inhibition of DROSHA m6A modification attenuates BCSC traits. Our study unveils the AURKA-induced oncogenic m6A modification as a key regulator of DROSHA in breast cancer and identifies a novel DROSHA transcriptional function in promoting the BCSC phenotype.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The RNA-seq and microarray data generated by this study have been deposited in the GEO database under the accession number GSE128428 (RNA-seq) and GSE125705 (microarray). Human breast tumor gene expression and clinical data were derived from TCGA Research Network (http://cancergenome.nih.gov/). Previously published meRIP-seq data and microarray data were re-analyzed. They are available under accession codes: GSE60213, GSE29714, GSE90642 and GSE54365 for meRIP-seq; GSE7513 and GSE15192 for microarray.
References
Han, J. et al. Posttranscriptional crossregulation between Drosha and DGCR8. Cell 136, 75–84 (2009).
Melamed, Z. et al. Alternative splicing regulates biogenesis of miRNAs located across exon-intron junctions. Mol. Cell 50, 869–881 (2013).
Nepal, C. et al. Transcriptional, post-transcriptional and chromatin-associated regulation of pri-miRNAs, pre-miRNAs and moRNAs. Nucleic Acids Res. 44, 3070–3081 (2016).
Wagschal, A. et al. Microprocessor, Setx, Xrn2, and Rrp6 co-operate to induce premature termination of transcription by RNAPII. Cell 150, 1147–1157 (2012).
Lin, S. & Gregory, R. I. MicroRNA biogenesis pathways in cancer. Nat. Rev. Cancer 15, 321–333 (2015).
Hata, A. & Kashima, R. Dysregulation of microRNA biogenesis machinery in cancer. Crit. Rev. Biochem. Mol. Biol. 51, 121–134 (2016).
Lu, W. T. et al. Drosha drives the formation of DNA:RNA hybrids around DNA break sites to facilitate DNA repair. Nat. Commun. 9, 532 (2018).
Gromak, N. et al. Drosha regulates gene expression independently of RNA cleavage function. Cell Rep. 5, 1499–1510 (2013).
Wang, X., Zhao, X., Gao, P. & Wu, M. c-Myc modulates microRNA processing via the transcriptional regulation of Drosha. Sci. Rep. 3, 1942 (2013).
Zhao, B. S., Roundtree, I. A. & He, C. Post-transcriptional gene regulation by mRNA modifications. Nat. Rev. Mol. Cell Biol. 18, 31–42 (2017).
Yang, Y., Hsu, P. J., Chen, Y. S. & Yang, Y. G. Dynamic transcriptomic m6A decoration: writers, erasers, readers and functions in RNA metabolism. Cell Res. 28, 616–624 (2018).
Deng, X., Su, R., Feng, X., Wei, M. & Chen, J. Role of N6-methyladenosine modification in cancer. Curr. Opin. Genet. Dev. 48, 1–7 (2018).
Zhang, S. et al. m6A demethylase ALKBH5 maintains tumorigenicity of glioblastoma stem-like cells by sustaining FOXM1 expression and cell proliferation program. Cancer Cell 31, 591–606 (2017).
Weng, H. et al. METTL14 inhibits hematopoietic stem/progenitor differentiation and promotes leukemogenesis via mRNA m6A modification. Cell Stem Cell 22, 191–205 (2018).
Chen, M. et al. RNA N6-methyladenosine methyltransferase-like 3 promotes liver cancer progression through YTHDF2-dependent posttranscriptional silencing of SOCS2. Hepatology 67, 2254–2270 (2018).
Huang, H. et al. Recognition of RNA N6-methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat. Cell Biol. 20, 285–295 (2018).
Yan, M. et al. Aurora-A kinase: a potent oncogene and target for cancer therapy. Med. Res. Rev. 36, 1036–1079 (2016).
Zheng, F. et al. Nuclear AURKA acquires kinase-independent transactivating function to enhance breast cancer stem cell phenotype. Nat. Commun. 7, 10180 (2016).
Clarke, M. F. Clinical and therapeutic implications of cancer stem cells. N. Engl. J. Med. 380, 2237–2245 (2019).
Geula, S. et al. Stem cells. m6A mRNA methylation facilitates resolution of naive pluripotency toward differentiation. Science 347, 1002–1006 (2015).
Seita, J. et al. Gene expression commons: an open platform for absolute gene expression profiling. PloS One 7, e40321 (2012).
Han, J. et al. The Drosha-DGCR8 complex in primary microRNA processing. Genes Dev. 18, 3016–3027 (2004).
Bourguignon, L. Y., Spevak, C. C., Wong, G., Xia, W. & Gilad, E. Hyaluronan-CD44 interaction with protein kinase C(epsilon) promotes oncogenic signaling by the stem cell marker Nanog and the Production of microRNA-21, leading to down-regulation of the tumor suppressor protein PDCD4, anti-apoptosis, and chemotherapy resistance in breast tumor cells. J. Biol. Chem. 284, 26533–26546 (2009).
Fustin, J. M. et al. RNA-methylation-dependent RNA processing controls the speed of the circadian clock. Cell 155, 793–806 (2013).
Pendleton, K. E. et al. The U6 snRNA m6A methyltransferase METTL16 regulates SAM synthetase intron retention. Cell 169, 824–835 (2017).
Li, Y. et al. Stanniocalcin-1 augments stem-like traits of glioblastoma cells through binding and activating NOTCH1. Cancer Lett. 416, 66–74 (2018).
Cao, J. X. et al. E2F1-regulated DROSHA promotes miR-630 biosynthesis in cisplatin-exposed cancer cells. Biochem. Biophys. Res. Commun. 450, 470–475 (2014).
Wang, X. et al. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature 505, 117–120 (2014).
Niu, Y., Wan, A., Lin, Z., Lu, X. & Wan, G. N6-methyladenosine modification: a novel pharmacological target for anti-cancer drug development. Acta Pharm. Sin. B 8, 833–843 (2018).
Vu, L. P., Cheng, Y. & Kharas, M. G. The Biology of m6A RNA methylation in normal and malignant hematopoiesis. Cancer Discov. 9, 25–33 (2019).
Tan, J. et al. Pharmacologic disruption of polycomb-repressive complex 2-mediated gene repression selectively induces apoptosis in cancer cells. Genes Dev. 21, 1050–1063 (2007).
Mayr, C. et al. 3-deazaneplanocin A may directly target putative cancer stem cells in biliary tract cancer. Anticancer Res. 35, 4697–4705 (2015).
Liu, J. et al. m6A mRNA methylation regulates AKT activity to promote the proliferation and tumorigenicity of endometrial cancer. Nat. Cell Biol. 20, 1074–1083 (2018).
Du, Y. et al. SUMOylation of the m6A-RNA methyltransferase METTL3 modulates its function. Nucleic Acids Res. 46, 5195–5208 (2018).
Zhou, J. et al. Dynamic m(6)A mRNA methylation directs translational control of heat shock response. Nature 526, 591–594 (2015).
Shi, H., Wei, J. & He, C. Where, when, and how: context-dependent functions of RNA methylation writers, readers, and erasers. Mol. Cell 74, 640–650 (2019).
Bertero, A. et al. The SMAD2/3 interactome reveals that TGFbeta controls m6A mRNA methylation in pluripotency. Nature 555, 256–259 (2018).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Cui, B. et al. Stress-induced epinephrine enhances lactate dehydrogenase A and promotes breast cancer stem-like cells. J. Clin. Invest. 129, 1030–1046 (2019).
Dominissini, D., Moshitch-Moshkovitz, S., Salmon-Divon, M., Amariglio, N. & Rechavi, G. Transcriptome-wide mapping of N(6)-methyladenosine by m(6)A-seq based on immunocapturing and massively parallel sequencing. Nat. Protoc. 8, 176–189 (2013).
Meyer, K. D. et al. Comprehensive analysis of mRNA methylation reveals enrichment in 3’ UTRs and near stop codons. Cell 149, 1635–1646 (2012).
Shevchenko, A., Tomas, H., Havlis, J., Olsen, J. V. & Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 1, 2856–2860 (2006).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Detre, S., Saclani Jotti, G. & Dowsett, M. A “quickscore” method for immunohistochemical semiquantitation: validation for oestrogen receptor in breast carcinomas. J. Clin. Pathol. 48, 876–878 (1995).
Creighton, C. J. et al. Residual breast cancers after conventional therapy display mesenchymal as well as tumor-initiating features. Proc. Natl. Acad. Sci. USA 106, 13820–13825 (2009).
Yang, J. et al. The I-TASSER Suite: protein structure and function prediction. Nat. Methods 12, 7–8 (2015).
Feng, L. et al. De novo molecular design of a novel octapeptide that inhibits in vivo melanogenesis and has great transdermal ability. J. Med. Chem. 61, 6846–6857 (2018).
Waterhouse, A. et al. SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res. 46, W296–W303 (2018).
Hu, Y. & Smyth, G. K. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J. Immunol. Methods 347, 70–78 (2009).
Mathelier, A. et al. JASPAR 2014: an extensively expanded and updated open-access database of transcription factor binding profiles. Nucleic Acids Res. 42, D142–D147 (2014).
Tang, Z. et al. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 45, W98–W102 (2017).
Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal. 6, pl1 (2013).
Xuan, J. J. et al. RMBase v2.0: deciphering the map of RNA modifications from epitranscriptome sequencing data. Nucleic Acids Res. 46, D327–D334 (2018).
Lanczky, A. et al. miRpower: a web-tool to validate survival-associated miRNAs utilizing expression data from 2178 breast cancer patients. Breast Cancer Res. Treat. 160, 439–446 (2016).
Acknowledgements
This work was supported by the National Key R&D Program of China (2019YFA0110300 to Q.Liu and 2017YFA0505600-04 to Q.Liu), the National Natural Science Foundation of China (81820108024 to Q.Liu, 81630005 to Q.Liu, 81972786 to J.X., 81573025 to Q.Liu, 81703062 to L.H. and 81703091 to F.A.), Program for Changjiang Scholars and Innovative Research Team in University of Ministry of Education of China (IRT_17R15), Innovative Research Team in University of Liaoning (LT2017001 to Q.Liu), the Natural Science Foundation of Liaoning (2019-BS-081 to F.P.), the “Seedling cultivation” program for young scientific and technological talents of Liaoning (LZ2019067 to B.C. and 2020 to F.P.), the program for climbing Scholars of Liaoning, the Science and Technology Innovation Foundation of Dalian (2020JJ25CY008 to Q.Liu), Dalian High-level Talent Innovation Program (2016RD12 to Q.Liu), International Scientific and Technological Cooperation of Dalian (2015F11GH095 to Q.Liu), the Natural Science Foundation of Guangdong (2016A030311038 and 2017A030313608 to Q.Liu, 2017A020215098 to Z.W.), the Science and Technology Planning Project of Guangzhou (201804020044 to Q.Liu). E.W-F.L.’s work is supported by MRC (MR/N012097/1), CRUK (C37/A12011; C37/18784), Breast Cancer Now (2012MayPR070; 2012NovPhD016; 2014NovPhD326). The authors thank Prof. Yungui Yang (Beijing Institute of Genomics, CAS) for scientific advice and technical assistance, especially meRIP-seq data analysis and the method of meRIP-qPCR assay. The authors thank Prof. Lingqiang Zhang (Beijing Institute of Lifeomics, Beijing, China) for a gift of HA-Ub plasmid.
Author information
Authors and Affiliations
Contributions
Q.Liu, J.X and F.P. conceived and designed the entire project. Q.Liu, F.P., J.X., B.H. and Z.W., designed and supervised the research. F.P., B.C., Q.Liang, S.Z., H.Zou, M.L., H.Zhao, Y.M., J.H. and J.Z. performed the experimental data analyses and/or experimental planning. H.Zou, Y.L., J.L. and Z.L., performed the transcriptome-wide and meRIP-seq data analyses. B.L., S.L., J.C. and F.W. performed the proteomic analyses by LC-MS. P.C. and Z.S. performed MD simulation. F.A. drafted the working model. L.X. collected breast tumor specimens and conjugated normal breast specimens. Q.Liu, J.X., F.P., B.C. and E.W-F.L. contributed reagents/analytic tools and/or grant support. F.P., B.C., Q.Liu, J.X., B.H., Q.Liang, S.Z., Y.Z. and E.W-F.L. wrote and revised the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Peng, F., Xu, J., Cui, B. et al. Oncogenic AURKA-enhanced N6-methyladenosine modification increases DROSHA mRNA stability to transactivate STC1 in breast cancer stem-like cells. Cell Res 31, 345–361 (2021). https://doi.org/10.1038/s41422-020-00397-2
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41422-020-00397-2
This article is cited by
-
Targeting epigenetic regulators as a promising avenue to overcome cancer therapy resistance
Signal Transduction and Targeted Therapy (2025)
-
N6-methyladenosine-modified GPX2 impacts cancer cell stemness and TKI resistance through regulating of redox metabolism
Cell Death & Disease (2025)
-
RNA methylation and breast cancer: insights into m6A, m7G and m5C
Molecular Biology Reports (2025)
-
Targeting “don’t eat me” signal: breast cancer immunotherapy
Breast Cancer Research and Treatment (2025)
-
Functional and clinical significance of the RNA m6A methyltransferase complex in breast cancer
npj Breast Cancer (2025)