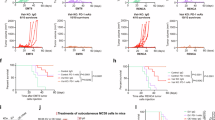
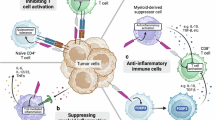
Abstract
The limited success of current immunotherapies emphasizes the need for new targets and combination treatments. V-domain Ig suppressor of T cell activation (VISTA) is a promising immune checkpoint target in cancer immunotherapy, but its regulatory mechanism is poorly understood. Through CRISPR knockout screening and proteomic analysis, we identify tripartite motif containing 25 (TRIM25) as a positive regulator for VISTA largely through antagonizing its degradation signaling. Moreover, ERK-mediated phosphorylation of VISTA at Thr284 enhances its interaction with TRIM25, leading to VISTA stabilization. A VISTA-derived phospho-peptide competitively disrupts TRIM25–VISTA interaction, thereby reducing VISTA expression and potentiating the anti-tumor efficacy of PD-1/PD-L1 blockade. Moreover, single-cell RNA sequencing analysis shows that tumor-infiltrating cytotoxic CD8+ T cells are increased in mice with T cell-specific knockout of Trim25. Of note, genetic ablation of Trim25 in T cells not only improves anti-PD-L1 immunotherapy, but also significantly ameliorates CAR T anti-tumor activity in various mouse tumor models. Collectively, this study unveils a mechanism for VISTA regulation in T cells and highlights targeting TRIM25–VISTA as a potential strategy to enhance tumor immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data are available in the main text or the supplementary materials. The raw DNA sequencing data of the CRISPR screen have been deposited under SRA PRJNA1183302. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the iProX partner repository with the dataset identifier PXD057240. The scRNA-seq FASTQ files are available at GEO under accession GSE281314. All the materials generated in this study are available upon reasonable request to the lead contact.
References
Freeman, G. J. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 192, 1027–1034 (2000).
Pauken, K. E., Torchia, J. A., Chaudhri, A., Sharpe, A. H. & Freeman, G. J. Emerging concepts in PD-1 checkpoint biology. Semin Immunol. 52, 101480 (2021).
Leach, D. R., Krummel, M. F. & Allison, J. P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 271, 1734–1736 (1996).
Boutros, C. et al. Safety profiles of anti-CTLA-4 and anti-PD-1 antibodies alone and in combination. Nat. Rev. Clin. Oncol. 13, 473–486 (2016).
Chen, L. & Han, X. Anti-PD-1/PD-L1 therapy of human cancer: past, present, and future. J. Clin. Invest. 125, 3384–3391 (2015).
Zhang, J., Dang, F., Ren, J. & Wei, W. Biochemical aspects of PD-L1 regulation in cancer immunotherapy. Trends Biochem. Sci. 43, 1014–1032 (2018).
Zou, W., Wolchok, J. D. & Chen, L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci. Transl. Med. 8, 328rv324 (2016).
Sharma, P. & Allison, J. P. The future of immune checkpoint therapy. Science 348, 56–61 (2015).
Liu, R., Li, H. F. & Li, S. PD-1-mediated inhibition of T cell activation: Mechanisms and strategies for cancer combination immunotherapy. Cell Insight 3, 100146 (2024).
Motzer, R. J. et al. Nivolumab for metastatic renal cell carcinoma: results of a randomized phase II trial. J. Clin. Oncol. 33, 1430–1437 (2015).
Wang, H. et al. Immune checkpoint blockade and CAR-T cell therapy in hematologic malignancies. J. Hematol. Oncol. 12, 59 (2019).
Wang, L. et al. VISTA, a novel mouse Ig superfamily ligand that negatively regulates T cell responses. J. Exp. Med. 208, 577–592 (2011).
Flies, D. B., Wang, S., Xu, H. & Chen, L. Cutting edge: A monoclonal antibody specific for the programmed death-1 homolog prevents graft-versus-host disease in mouse models. J. Immunol. 187, 1537–1541 (2011).
Sakr, M. A. et al. GI24 enhances tumor invasiveness by regulating cell surface membrane-type 1 matrix metalloproteinase. Cancer Sci. 101, 2368–2374 (2010).
Yoon, K. W. et al. Control of signaling-mediated clearance of apoptotic cells by the tumor suppressor p53. Science 349, 1261669 (2015).
Yuan, L., Tatineni, J., Mahoney, K. M. & Freeman, G. J. VISTA: A mediator of quiescence and a promising target in cancer immunotherapy. Trends Immunol. 42, 209–227 (2021).
Johnston, R. J. et al. VISTA is an acidic pH-selective ligand for PSGL-1. Nature 574, 565–570 (2019).
Mehta, N. et al. Structure and functional binding epitope of V-domain Ig suppressor of T cell activation. Cell Rep. 28, 2509–2516.e5 (2019).
Han, X. et al. PD-1H (VISTA)-mediated suppression of autoimmunity in systemic and cutaneous lupus erythematosus. Sci. Transl. Med. 11, eaax1159 (2019).
Wang, L. et al. Disruption of the immune-checkpoint VISTA gene imparts a proinflammatory phenotype with predisposition to the development of autoimmunity. Proc. Natl. Acad. Sci. USA 111, 14846–14851 (2014).
Nowak, E. C. et al. Immunoregulatory functions of VISTA. Immunol. Rev. 276, 66–79 (2017).
ElTanbouly, M. A. et al. VISTA is a checkpoint regulator for naïve T cell quiescence and peripheral tolerance. Science 367, eaay0524 (2020).
Hong, S. et al. Analysis of VISTA expression and function in renal cell carcinoma highlights VISTA as a potential target for immunotherapy. Protein Cell 10, 840–845 (2019).
Villarroel-Espindola, F. et al. Spatially resolved and quantitative analysis of VISTA/PD-1H as a novel immunotherapy target in human non-small cell lung cancer. Clin. Cancer Res. 24, 1562–1573 (2018).
Rosenbaum, S. R. et al. FOXD3 regulates VISTA expression in melanoma. Cell Rep. 30, 510–524.e6 (2020).
Kang, J. Y. et al. VISTA-targeted antibody-drug conjugates exhibit potent antitumor effects on malignant pleural mesothelioma. Medcomm-Oncol. 3, e78 (2024).
Flies, D. B., Higuchi, T. & Chen, L. Mechanistic assessment of PD-1H coinhibitory receptor-induced T Cell tolerance to allogeneic antigens. J. Immunol. 194, 5294–5304 (2015).
Hu, L. et al. Ablation of T cell-associated PD-1H enhances functionality and promotes adoptive immunotherapy. JCI Insight 7, e148247 (2022).
Flies, D. B. et al. Coinhibitory receptor PD-1H preferentially suppresses CD4(+) T cell-mediated immunity. J. Clin. Invest. 124, 1966–1975 (2014).
Liu, J. et al. Immune-checkpoint proteins VISTA and PD-1 nonredundantly regulate murine T-cell responses. Proc. Natl. Acad. Sci. USA 112, 6682–6687 (2015).
Rape, M. Ubiquitylation at the crossroads of development and disease. Nat. Rev. Mol. Cell Biol. 19, 59–70 (2018).
Komander, D. & Rape, M. The ubiquitin code. Annu. Rev. Biochem. 81, 203–229 (2012).
Clague, M. J., Urbe, S. & Komander, D. Breaking the chains: deubiquitylating enzyme specificity begets function. Nat. Rev. Mol. Cell Biol. 20, 338–352 (2019).
Jia, R. & Bonifacino, J. S. Negative regulation of autophagy by UBA6-BIRC6-mediated ubiquitination of LC3. Elife 8, e50034 (2019).
Mei, P. et al. E3 ligase TRIM25 ubiquitinates RIP3 to inhibit TNF induced cell necrosis. Cell Death Differ. 28, 2888–2899 (2021).
Hatakeyama, S. TRIM family proteins: roles in autophagy, immunity, and carcinogenesis. Trends Biochem. Sci. 42, 297–311 (2017).
Li, J. et al. Tumor-specific GPX4 degradation enhances ferroptosis-initiated antitumor immune response in mouse models of pancreatic cancer. Sci. Transl. Med. 15, eadg3049 (2023).
Chen, Y. et al. TRIM25 promotes glioblastoma cell growth and invasion via regulation of the PRMT1/c-MYC pathway by targeting the splicing factor NONO. J. Exp. Clin. Cancer Res 43, 39 (2024).
Komander, D. & Rape, M. The ubiquitin code. Annu. Rev. Biochem. 81, 203–229 (2012).
Gack, M. U. et al. TRIM25 RING-finger E3 ubiquitin ligase is essential for RIG-I-mediated antiviral activity. Nature 446, 916–920 (2007).
Wu, S. F. et al. RIG-I regulates myeloid differentiation by promoting TRIM25-mediated ISGylation. Proc. Natl. Acad. Sci. USA 117, 14395–14404 (2020).
Park, J. M. et al. Modification of PCNA by ISG15 plays a crucial role in termination of error-prone translesion DNA synthesis. Mol. Cell 54, 626–638 (2014).
Pair, F. S. & Yacoubian, T. A. 14-3-3 proteins: novel pharmacological targets in neurodegenerative diseases. Trends Pharm. Sci. 42, 226–238 (2021).
Stevers, L. M. et al. Modulators of 14-3-3 Protein-Protein Interactions. J. Med. Chem. 61, 3755–3778 (2018).
Zhu, G., Herlyn, M. & Yang, X. TRIM15 and CYLD regulate ERK activation via lysine-63-linked polyubiquitination. Nat. Cell Biol. 23, 978–991 (2021).
Xiao, X. et al. ERK and USP5 govern PD-1 homeostasis via deubiquitination to modulate tumor immunotherapy. Nat. Commun. 14, 2859 (2023).
Orimo, A. et al. Underdeveloped uterus and reduced estrogen responsiveness in mice with disruption of the estrogen-responsive finger protein gene, which is a direct target of estrogen receptor alpha. Proc. Natl. Acad. Sci. USA 96, 12027–12032 (1999).
Zhang, L. et al. Single-cell analyses inform mechanisms of myeloid-targeted therapies in colon cancer. Cell 181, 442–459.e9 (2020).
Mager, L. F. et al. Microbiome-derived inosine modulates response to checkpoint inhibitor immunotherapy. Science 369, 1481–1489 (2020).
Kakavand, H. et al. Negative immune checkpoint regulation by VISTA: a mechanism of acquired resistance to anti-PD-1 therapy in metastatic melanoma patients. Mod. Pathol. 30, 1666–1676 (2017).
Gao, J. et al. VISTA is an inhibitory immune checkpoint that is increased after ipilimumab therapy in patients with prostate cancer. Nat. Med. 23, 551–555 (2017).
Meloni, B. P. et al. Neuroprotective peptides fused to arginine-rich cell penetrating peptides: Neuroprotective mechanism likely mediated by peptide endocytic properties. Pharm. Ther. 153, 36–54 (2015).
Li, W. et al. Quality control, modeling, and visualization of CRISPR screens with MAGeCK-VISPR. Genome Biol. 16, 281 (2015).
Rodig, N. et al. Endothelial expression of PD-L1 and PD-L2 down-regulates CD8+ T cell activation and cytolysis. Eur. J. Immunol. 33, 3117–3126 (2003).
Xiong, W. et al. USP8 inhibition reshapes an inflamed tumor microenvironment that potentiates the immunotherapy. Nat. Commun. 13, 1700 (2022).
Ho, A., Schwarze, S. R., Mermelstein, S. J., Waksman, G. & Dowdy, S. F. Synthetic protein transduction domains: enhanced transduction potential in vitro and in vivo. Cancer Res. 61, 474–477 (2001).
Acknowledgements
We thank the Core Facility and the Animal Facility of Medical Research Institute of Wuhan University for technical support. This work was supported by grants from the National Key R&D Program of China (2022YFC3401500 and 2023YFC3402100 to J.Z.), the National Natural Science Foundation of China (82273062 to J.Z., 82103149 to H.Y., 82503802 to B.C.), the Fundamental Research Funds for the Central Universities (2042022dx0003 to J.Z., 2042021kf0075 to H.Y.), the Postdoctoral Fellowship Program of CPSF (GZC20241276 to W.X., GZC20250976 to C.H.), the China Postdoctoral Science Foundation (2022M722462 to H.L., 2024M762491 to C.H., 2025M772321 to W.X.), the Postdoctoral Project of Hubei Province (2004HBBHJD057 to C.H.), Natural Science Foundation of Wuhan (2024040701010031 to J.Z.), the Open Projects of Hubei Key Laboratory of Tumor Biological Behavior (220172107 to J.Z.), the Natural Science Foundation of Hubei Province of China (2022CFA008 to J.Z.), the Open Research Fund of the National Center for Protein Sciences at Peking University in Beijing (KF-202504 to J.Z.), the Natural Science Foundation Youth Category A of Hubei Province (JCZRJQ202500008 to H.Z.), the Natural Science Foundation of Chongqing, China (CSTB2024NSCQ-MSX0066 to H.Z.), the translational Medicine and Interdisciplinary Research Joint Fund of Zhongnan Hospital of Wuhan University (ZNJC202312 to J.Z.), and USA National Institutes of Health P50 CA101942 (G.J.F.).
Author information
Authors and Affiliations
Contributions
Y.S. performed most of the experiments with help from Z.Z., Haiou L., X.B., L.C., X.W., L.K., P.D., W.S., X.X., J.S., B.X., C.H., Y.Y., W.X., and H.Y.; J.Z., Y.S., G.J.F., H.Z., and W.W. designed experiments; L.F., B.C., C.J., and Q.Q. collected human colorectal cancer tissues; X.B. and G.J.F. provided essential reagents; J.Z., H.Z., G.J.F., and W.W. guided and supervised the project. J.Z. and Y.S. wrote the manuscript. X.B., Z.Y., G.Q., S.T., Hudan L., W.W., and G.J.F. provided discussion and edited the manuscript. All authors commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
G.J.F. has patents/royalties on the PD-L1/PD-1 pathway from Bristol-Myers-Squibb, Roche, Eli Lilly, and Novartis. G.J.F. has served on advisory boards for iTeos, NextPoint, IgM, GV20, IOME, Bioentre, Santa Ana Bio, Simcere of America, and Geode. G.J.F. has equity in Nextpoint, Triursus, Xios, iTeos, IgM, Invaria, GV20, Bioentre, and Geode. W.W. is a co-founder and consultant for the ReKindle Therapeutics. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, Y., Zhang, Z., Li, H. et al. Destruction of VISTA by TRIM25 ablation in T cells potentiates cancer immunotherapy. Cell Res 35, 1003–1020 (2025). https://doi.org/10.1038/s41422-025-01186-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41422-025-01186-5