Abstract
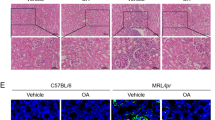
Autoantibodies produced by B cells play a pivotal role in the pathogenesis of systemic lupus erythematosus (SLE). However, both the cellular source of antiphospholipid antibodies and their contributions to the development of lupus nephritis (LN) remain largely unclear. Here, we report a pathogenic role of anti-phosphatidylserine (PS) autoantibodies in the development of LN. Elevated serum PS-specific IgG levels were measured in model mice and SLE patients, especially in those with LN. PS-specific IgG accumulation was found in the kidney biopsies of LN patients. Both transfer of SLE PS-specific IgG and PS immunization triggered lupus-like glomerular immune complex deposition in recipient mice. ELISPOT analysis identified B1a cells as the main cell type that secretes PS-specific IgG in both lupus model mice and patients. Adoptive transfer of PS-specific B1a cells accelerated the PS-specific autoimmune response and renal damage in recipient lupus model mice, whereas depletion of B1a cells attenuated lupus progression. In culture, PS-specific B1a cells were significantly expanded upon treatment with chromatin components, while blockade of TLR signal cascades by DNase I digestion and inhibitory ODN 2088 or R406 treatment profoundly abrogated chromatin-induced PS-specific IgG secretion by lupus B1a cells. Thus, our study has demonstrated that the anti-PS autoantibodies produced by B1 cells contribute to lupus nephritis development. Our findings that blockade of the TLR/Syk signaling cascade inhibits PS-specific B1-cell expansion provide new insights into lupus pathogenesis and may facilitate the development of novel therapeutic targets for the treatment of LN in SLE.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Zhao Z, Ren J, Dai C, Kannapell CC, Wang H, Gaskin F, et al. Nature of T cell epitopes in lupus antigens and HLA-DR determines autoantibody initiation and diversification. Ann Rheum Dis. 2019;78:380–90.
Ma K, Du W, Xiao F, Han M, Huang E, Peng N, et al. IL-17 sustains the plasma cell response via p38-mediated Bcl-xL RNA stability in lupus pathogenesis. Cell Mol Immunol. 2021;18:1739–50.
Zhao Z, Xu B, Wang S, Zhou M, Huang Y, Guo C, et al. Tfh cells with NLRP3 inflammasome activation are essential for high-affinity antibody generation, germinal centre formation and autoimmunity. Ann Rheum Dis. 2022;81:1006–12.
Waters ST, McDuffie M, Bagavant H, Deshmukh US, Gaskin F, Jiang C, et al. Breaking tolerance to double stranded DNA, nucleosome, and other nuclear antigens is not required for the pathogenesis of lupus glomerulonephritis. J Exp Med. 2004;199:255–64.
Fu SM, Dai C, Zhao Z, Gaskin F. Anti-dsDNA antibodies are one of the many autoantibodies in systemic lupus erythematosus. F1000Res. 2015;4:939.
Zuo Y, Estes SK, Ali RA, Gandhi AA, Yalavarthi S, Shi H, et al. Prothrombotic autoantibodies in serum from patients hospitalized with COVID-19. Sci Transl Med. 2020;12:eabd3876.
Tselios K, Sarantopoulos A, Gkougkourellas I, Boura P. The role of anti-phosphatidylserine/prothrombin antibodies in thrombotic manifestations of systemic lupus erythematosus patients. Front Neurol. 2013;4:66.
Radin M, Cecchi I, Rubini E, Schreiber K, Roccatello D, Mahler M, et al. Prevalence and significance of anti-phosphatidylserine antibodies: a pooled analysis in 5992 patients. Thromb Res. 2018;170:28–31.
Zhu H, Luo H, Yan M, Zuo X, Li QZ. Autoantigen microarray for high-throughput autoantibody profiling in systemic lupus erythematosus. Genomics Proteomics Bioinformatics. 2015;13:210–18.
Farina N, Abdulsalam R, McDonnell T, Pericleous C, D’Souza A, Ripoll VM, et al. Antiphospholipid antibody positivity in early systemic lupus erythematosus is associated with subsequent vascular events. Rheumatology. 2022;62:2252–6.
Muller-Calleja N, Hollerbach A, Royce J, Ritter S, Pedrosa D, Madhusudhan T, et al. Lipid presentation by the protein C receptor links coagulation with autoimmunity. Science. 2021;371:eabc0956.
Sprangers B, Monahan M, Appel GB. Diagnosis and treatment of lupus nephritis flares-an update. Nat Rev Nephrol. 2012;8:709–17.
Fischer K, Przepiera-Bedzak H, Brzosko I, Sawicki M, Walecka A, Brzosko M. Anti-phosphatidylethanolamine and anti-phosphatidylserine antibodies-association with renal involvement, atherosclerosis, cardiovascular manifestations, Raynaud phenomenon and disease activity in Polish patients with systemic lupus erythematosus. Biomolecules. 2022;12:1328.
Sawada T, Kurano M, Shirai H, Iwasaki Y, Tahara K, Hayashi H, et al. Serum phosphatidylserine-specific phospholipase A1 as a novel biomarker for monitoring systemic lupus erythematosus disease activity. Int J Rheum Dis. 2019;22:2059–66.
Baumgarth N. The double life of a B-1 cell: self-reactivity selects for protective effector functions. Nat Rev Immunol. 2011;11:34–46.
Wang X, Ma K, Chen M, Ko KH, Zheng BJ, Lu L. IL-17A promotes pulmonary B-1a cell differentiation via induction of Blimp-1 Expression During Influenza Virus Infection. PLoS Pathog. 2016;12:e1005367.
Yang Y, Li X, Ma Z, Wang C, Yang Q, Byrne-Steele M, et al. CTLA-4 expression by B-1a B cells is essential for immune tolerance. Nat Commun. 2021;12:525.
Xu Z, Butfiloski EJ, Sobel ES, Morel L. Mechanisms of peritoneal B-1a cells accumulation induced by murine lupus susceptibility locus Sle2. J Immunol. 2004;173:6050–8.
Duan B, Morel L. Role of B-1a cells in autoimmunity. Autoimmun Rev. 2006;5:403–8.
Diana J, Simoni Y, Furio L, Beaudoin L, Agerberth B, Barrat F, et al. Crosstalk between neutrophils, B-1a cells and plasmacytoid dendritic cells initiates autoimmune diabetes. Nat Med. 2013;19:65–73.
Griffin DO, Holodick NE, Rothstein TL. Human B1 cells in umbilical cord and adult peripheral blood express the novel phenotype CD20+ CD27+ CD43+ CD70. J Exp Med. 2011;208:67–80.
Griffin DO, Rothstein TL. A small CD11b(+) human B1 cell subpopulation stimulates T cells and is expanded in lupus. J Exp Med. 2011;208:2591–8.
Ma K, Wang X, Shi X, Lin X, Xiao F, Ma X, et al. The expanding functional diversity of plasma cells in immunity and inflammation. Cell Mol Immunol. 2020;17:421–2.
Ma K, Li J, Wang X, Lin X, Du W, Yang X, et al. TLR4(+)CXCR4(+) plasma cells drive nephritis development in systemic lupus erythematosus. Ann Rheum Dis. 2018;77:1498–506.
Wu L, Seon GM, Kim Y, Choi SH, Vo QC, Yang HC. Enhancing effect of sodium butyrate on phosphatidylserine-liposome-induced macrophage polarization. Inflamm Res. 2022;71:641–52.
Petri M, Orbai AM, Alarcon GS, Gordon C, Merrill JT, Fortin PR, et al. Derivation and validation of the systemic lupus international collaborating clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64:2677–86.
Fukushi S. Competitive ELISA for the detection of serum antibodies specific for middle east respiratory syndrome coronavirus (MERS-CoV). Methods Mol Biol. 2020;2203:55–65.
Hu K. Become competent within one day in generating boxplots and violin plots for a novice without prior R experience. Methods Protoc. 2020;3:64.
Li H, Zhang YY, Sun YN, Huang XY, Jia YF, Li D. Induction of systemic lupus erythematosus syndrome in BALB/c mice by immunization with active chromatin. Acta Pharm Sin. 2004;25:807–11.
Alsadeq A, Hobeika E, Medgyesi D, Klasener K, Reth M. The role of the Syk/Shp-1 kinase-phosphatase equilibrium in B cell development and signaling. J Immunol. 2014;193:268–76.
Fernandez-Arias C, Rivera-Correa J, Gallego-Delgado J, Rudlaff R, Fernandez C, Roussel C, et al. Anti-self phosphatidylserine antibodies recognize uninfected erythrocytes promoting malarial anemia. Cell Host Microbe. 2016;19:194–203.
Nagata S. Apoptosis and clearance of apoptotic cells. Annu Rev Immunol. 2018;36:489–517.
Qian Y, Conway KL, Lu X, Seitz HM, Matsushima GK, Clarke SH. Autoreactive MZ and B-1 B-cell activation by Faslpr is coincident with an increased frequency of apoptotic lymphocytes and a defect in macrophage clearance. Blood. 2006;108:974–82.
Berland R, Wortis HH. Origins and functions of B-1 cells with notes on the role of CD5. Annu Rev Immunol. 2002;20:253–300.
Clarke AJ, Riffelmacher T, Braas D, Cornall RJ, Simon AK. B1a B cells require autophagy for metabolic homeostasis and self-renewal. J Exp Med. 2018;215:399–413.
Ansel KM, Harris RB, Cyster JG. CXCL13 is required for B1 cell homing, natural antibody production, and body cavity immunity. Immunity. 2002;16:67–76.
Ito T, Ishikawa S, Sato T, Akadegawa K, Yurino H, Kitabatake M, et al. Defective B1 cell homing to the peritoneal cavity and preferential recruitment of B1 cells in the target organs in a murine model for systemic lupus erythematosus. J Immunol. 2004;172:3628–34.
Murakami M, Yoshioka H, Shirai T, Tsubata T, Honjo T. Prevention of autoimmune symptoms in autoimmune-prone mice by elimination of B-1 cells. Int Immunol. 1995;7:877–82.
Wu YY, Georg I, Diaz-Barreiro A, Varela N, Lauwerys B, Kumar R, et al. Concordance of increased B1 cell subset and lupus phenotypes in mice and humans is dependent on BLK expression levels. J Immunol. 2015;194:5692–702.
Deng J, Wang X, Chen Q, Sun X, Xiao F, Ko KH, et al. B1a cells play a pathogenic role in the development of autoimmune arthritis. Oncotarget. 2016;7:19299–311.
Rivera-Correa J, Mackroth MS, Jacobs T, Schulze Zur Wiesch J, Rolling T, Rodriguez A. Atypical memory B-cells are associated with Plasmodium falciparum anemia through anti-phosphatidylserine antibodies. Elife. 2019;8:e48309.
Ha SA, Tsuji M, Suzuki K, Meek B, Yasuda N, Kaisho T, et al. Regulation of B1 cell migration by signals through Toll-like receptors. J Exp Med. 2006;203:2541–50.
Seidl KJ, Wilshire JA, MacKenzie JD, Kantor AB, Herzenberg LA, Herzenberg LA. Predominant VH genes expressed in innate antibodies are associated with distinctive antigen-binding sites. Proc Natl Acad Sci USA. 1999;96:2262–7.
Li H, Jiang Y, Cao H, Radic M, Prak EL, Weigert M. Regulation of anti-phosphatidylserine antibodies. Immunity. 2003;18:185–92.
Ma K, Li J, Fang Y, Lu L. Roles of B cell-intrinsic TLR signals in systemic lupus erythematosus. Int J Mol Sci. 2015;16:13084–105.
Tran NL, Manzin-Lorenzi C, Santiago-Raber ML. Toll-like receptor 8 deletion accelerates autoimmunity in a mouse model of lupus through a Toll-like receptor 7-dependent mechanism. Immunology. 2015;145:60–70.
Desnues B, Macedo AB, Roussel-Queval A, Bonnardel J, Henri S, Demaria O, et al. TLR8 on dendritic cells and TLR9 on B cells restrain TLR7-mediated spontaneous autoimmunity in C57BL/6 mice. Proc Natl Acad Sci USA. 2014;111:1497–502.
Brown GJ, Canete PF, Wang H, Medhavy A, Bones J, Roco JA, et al. TLR7 gain-of-function genetic variation causes human lupus. Nature. 2022;605:349–56.
Wu X, Peng SL. Toll-like receptor 9 signaling protects against murine lupus. Arthritis Rheum. 2006;54:336–42.
Kubo T, Uchida Y, Watanabe Y, Abe M, Nakamura A, Ono M, et al. Augmented TLR9-induced Btk activation in PIR-B-deficient B-1 cells provokes excessive autoantibody production and autoimmunity. J Exp Med. 2009;206:1971–82.
Kreslavsky T, Vilagos B, Tagoh H, Poliakova DK, Schwickert TA, Wohner M, et al. Essential role for the transcription factor Bhlhe41 in regulating the development, self-renewal and BCR repertoire of B-1a cells. Nat Immunol. 2017;18:442–55.
Macias-Garcia A, Heizmann B, Sellars M, Marchal P, Dali H, Pasquali JL, et al. Ikaros is a negative regulator of B1 cell development and function. J Biol Chem. 2016;291:9073–86.
Patole PS, Zecher D, Pawar RD, Grone HJ, Schlondorff D, Anders HJ. G-rich DNA suppresses systemic lupus. J Am Soc Nephrol. 2005;16:3273–80.
Burke JM, Shustov A, Essell J, Patel-Donnelly D, Yang J, Chen R, et al. An open-label, phase II trial of entospletinib (GS-9973), a selective spleen tyrosine kinase inhibitor, in diffuse large B-cell lymphoma. Clin Lymphoma Myeloma Leuk. 2018;18:e327–31.
Schweighoffer E, Nys J, Vanes L, Smithers N, Tybulewicz VLJ. TLR4 signals in B lymphocytes are transduced via the B cell antigen receptor and SYK. J Exp Med. 2017;214:1269–80.
Acknowledgements
We thank Dr. Kwok Wah Chan, Dr. Xi Yang, and Mr. King-Hung Ko for their kind help in the renal histopathological assessment and technical assistance. We also thank the service of the Medical Faculty Core Facility and Laboratory Animal Unit, The University of Hong Kong.
Funding
This work was supported by funding for Chongqing International Institute for Immunology (2020YJC10), National Natural Science Foundation of China (81901635, 82171782, 82260326, 81971464), Shenzhen Science and Technology Program (CYJ20210324114602008), Hong Kong Research Grants Council Theme-Based Research Scheme (T12-703/19 R) and the Centre for Oncology and Immunology under the Health@InnoHK Initiative by the Innovation and Technology Commission, Hong Kong, China.
Author information
Authors and Affiliations
Contributions
KM, WD, SW, and JT designed and performed the mouse experiments and statistical analysis. JL, YX, XZ, JW, CL, HW, and FX performed the experiments with human samples. XK, KR, RQ, HZ, YZ, CW, DL, and LH participated in the data analysis. LL, QL, and LW supervised the study, and KM wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All human experiments were approved by the Institutional Medical Ethics Committee of Third Military Medical University (Reference Number: 2016-77-tmmu) and Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong Western Cluster (HKU/HA HKW IRB) (Reference Number: UW 16-342). Written permission was obtained from all subjects who participated in the study. All animal experiments were approved by the Committee on the Use of Live Animals in Teaching and Research (CULATR, No: 3392-14) at the University of Hong Kong.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, K., Du, W., Wang, S. et al. B1-cell-produced anti-phosphatidylserine antibodies contribute to lupus nephritis development via TLR-mediated Syk activation. Cell Mol Immunol 20, 881–894 (2023). https://doi.org/10.1038/s41423-023-01049-2
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41423-023-01049-2
Keywords
This article is cited by
-
Dehydrocorydaline treatment suppresses Th17 differentiation via the mTOR-mediated STAT3-RORγt signaling in systemic lupus erythematosus
Molecular Medicine (2026)
-
The influence of the gut microbiota on B cells in autoimmune diseases
Molecular Medicine (2025)
-
IL-18 drives the Bhlhe40-mediated pathogenic Th17 cell response and exacerbates autoimmune disease progression
Cellular & Molecular Immunology (2025)
-
Luminex-based pilot study identifies novel serum autoantibodies associated with disease activity in patients with lupus nephritis
Clinical Rheumatology (2025)
-
Age-Associated B Cells in Autoimmune Diseases: Pathogenesis and Clinical Implications
Clinical Reviews in Allergy & Immunology (2025)