Abstract
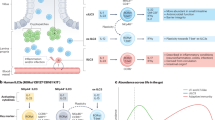
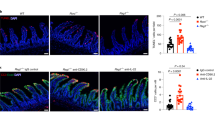
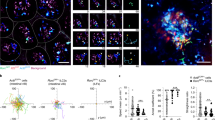
Communication between group 3 innate lymphoid cells (ILC3) and other immune cells, as well as intestinal epithelial cells, is pivotal in regulating intestinal inflammation. This study, for the first time, underscores the importance of crosstalk between intestinal endothelial cells (ECs) and ILC3. Our single-cell transcriptome analysis combined with protein expression detection revealed that ECs significantly increased the population of interleukin (IL)-22+ ILC3 through interactions mediated by endothelin-1 (ET-1) and its receptor endothelin A receptor (EDNRA). Genetic deficiency of EDNRA reduces the proportion of NKp46+ ILC3 and impairs IL-22 production in a T-cell-independent, cell-intrinsic manner, leading to increased intestinal inflammation. Mechanistically, the ET-1–EDNRA axis modulates hypoxia-inducible factor 1 alpha (HIF-1α) through protein kinase B (AKT) signaling, supporting metabolic adaptation toward glycolysis and providing protection against colitis. Moreover, restoring HIF-1α expression or providing exogenous lactate can alleviate colitis associated with EDNRA deficiency and ILC3 glycolytic dysfunction. These findings underscore the importance of communication between intestinal ECs and ILC3 via the ET-1–EDNRA axis in metabolic adaptation processes within ILC3 and maintaining intestinal homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Diefenbach A, Gnafakis S, Shomrat O. Innate lymphoid cell-epithelial cell modules sustain intestinal homeostasis. Immunity. 2020;52:452–63.
Spadoni I, Zagato E, Bertocchi A, Paolinelli R, Hot E, Di Sabatino A, et al. A gut-vascular barrier controls the systemic dissemination of bacteria. Science. 2015;350:830–4.
Naschberger E, Flierl C, Huang J, Erkert L, Gamez-Belmonte R, Gonzalez-Acera M, et al. Analysis of the interferon-γ-induced secretome of intestinal endothelial cells: putative impact on epithelial barrier dysfunction in IBD. Front Cell Dev Biol. 2023;11:1213383.
Zhang K, Hornef MW, DuPont A. The intestinal epithelium as guardian of gut barrier integrity. Cell Microbiol. 2015;17:1561–9.
Ricard N, Bailly S, Guignabert C, Simons M. The quiescent endothelium: signaling pathways regulating organ-specific endothelial normalcy. Nat Rev Cardiol. 2021;18:565–80.
Petrova TV, Koh GY. Organ-specific lymphatic vasculature: from development to pathophysiology. J Exp Med. 2018;215:35–49.
Hooper LV, Macpherson AJ. Immune adaptations that maintain homeostasis with the intestinal microbiota. Nat Rev Immunol. 2010;10:159–69.
Sonnenberg GF, Artis D. Innate lymphoid cells in the initiation, regulation and resolution of inflammation. Nat Med. 2015;21:698–708.
Lindemans CA, Calafiore M, Mertelsmann AM, O’Connor MH, Dudakov JA, Jenq RR, et al. Interleukin-22 promotes intestinal-stem-cell-mediated epithelial regeneration. Nature. 2015;528:560–4.
Das A, Harly C, Yang Q, Bhandoola A. Lineage specification in innate lymphocytes. Cytokine Growth Factor Rev. 2018;42:20–6.
Chu C, Moriyama S, Li Z, Zhou L, Flamar AL, Klose CSN, et al. Antimicrobial functions of group 3 innate lymphoid cells in gut-associated lymphoid tissues are regulated by G-protein-coupled receptor 183. Cell Rep. 2018;23:3750–8.
Vivier E, Artis D, Colonna M, Diefenbach A, Di Santo JP, Eberl G, et al. Innate lymphoid cells: 10 years on. Cell. 2018;174:1054–66.
Zhou W, Sonnenberg GF. Activation and suppression of group 3 innate lymphoid cells in the gut. Trends Immunol. 2020;41:721–33.
Melo-Gonzalez F, Hepworth MR. Functional and phenotypic heterogeneity of group 3 innate lymphoid cells. Immunology. 2017;150:265–75.
Zhong C, Zheng M, Zhu J. Lymphoid tissue inducer-A divergent member of the ILC family. Cytokine Growth Factor Rev. 2018;42:5–12.
Atreya I, Kindermann M, Wirtz S. Innate lymphoid cells in intestinal cancer development. Semin Immunol. 2019;41:101267.
Schroeder JH, Howard JK, Lord GM. Transcription factor-driven regulation of ILC1 and ILC3. Trends Immunol. 2022;43:564–79.
Zhang YZ. Li YY. Inflammatory bowel disease: pathogenesis. World J Gastroenterol. 2014;20:91–9.
Goc J, Lv M, Bessman NJ, Flamar AL, Sahota S, Suzuki H, et al. Dysregulation of ILC3s unleashes progression and immunotherapy resistance in colon cancer. Cell. 2021;184:5015–30.e16.
Geremia A, Arancibia-Cárcamo CV, Fleming MP, Rust N, Singh B, Mortensen NJ, et al. IL-23-responsive innate lymphoid cells are increased in inflammatory bowel disease. J Exp Med. 2011;208:1127–33.
Longman RS, Diehl GE, Victorio DA, Huh JR, Galan C, Miraldi ER, et al. CX₃CR1⁺ mononuclear phagocytes support colitis-associated innate lymphoid cell production of IL-22. J Exp Med. 2014;211:1571–83.
Mortha A, Chudnovskiy A, Hashimoto D, Bogunovic M, Spencer SP, Belkaid Y, et al. Microbiota-dependent crosstalk between macrophages and ILC3 promotes intestinal homeostasis. Science. 2014;343:1249288.
Gentile ME, King IL. Blood and guts: the intestinal vasculature during health and helminth infection. PLoS Pathog. 2018;14:e1007045.
Huang J, Lee HY, Zhao X, Han J, Su Y, Sun Q, et al. Interleukin-17D regulates group 3 innate lymphoid cell function through its receptor CD93. Immunity. 2021;54:673–86.e4.
Horn V, Sonnenberg GF. Group 3 innate lymphoid cells in intestinal health and disease. Nat Rev Gastroenterol Hepatol. 2024;21:428–43.
Cheng HW, Mörbe U, Lütge M, Engetschwiler C, Onder L, Novkovic M, et al. Intestinal fibroblastic reticular cell niches control innate lymphoid cell homeostasis and function. Nat Commun. 2022;13:2027.
Talbot J, Hahn P, Kroehling L, Nguyen H, Li D, Littman DR. Feeding-dependent VIP neuron-ILC3 circuit regulates the intestinal barrier. Nature. 2020;579:575–80.
Yu HB, Yang H, Allaire JM, Ma C, Graef FA, Mortha A, et al. Vasoactive intestinal peptide promotes host defense against enteric pathogens by modulating the recruitment of group 3 innate lymphoid cells. Proc Natl Acad Sci USA. 2021;118:e2106634118.
Pascal M, Kazakov A, Chevalier G, Dubrule L, Deyrat J, Dupin A, et al. The neuropeptide VIP potentiates intestinal innate type 2 and type 3 immunity in response to feeding. Mucosal Immunol. 2022;15:629–41.
Zhang J, Sjøberg KA, Gong S, Wang T, Li F, Kuo A, et al. Endothelial metabolic control of insulin sensitivity through resident macrophages. Cell Metab. 2024;36:2383–401.e9.
Wiggins BG, Wang YF, Burke A, Grunberg N, Vlachaki Walker JM, Dore M, et al. Endothelial sensing of AHR ligands regulates intestinal homeostasis. Nature. 2023;621:821–9.
Stockinger B, Shah K, Wincent E. AHR in the intestinal microenvironment: safeguarding barrier function. Nat Rev Gastroenterol Hepatol. 2021;18:559–70.
Nowarski R, Jackson R, Gagliani N, de Zoete MR, Palm NW, Bailis W, et al. Epithelial IL-18 equilibrium controls barrier function in colitis. Cell. 2015;163:1444–56.
Adolph TE, Meyer M, Schwärzler J, Mayr L, Grabherr F, Tilg H. The metabolic nature of inflammatory bowel diseases. Nat Rev Gastroenterol Hepatol. 2022;19:753–67.
Di Luccia B, Gilfillan S, Cella M, Colonna M, Huang SC. ILC3s integrate glycolysis and mitochondrial production of reactive oxygen species to fulfill activation demands. J Exp Med. 2019;216:2231–41.
Das A, Martinez-Ruiz GU, Bouladoux N, Stacy A, Moraly J, Vega-Sendino M, et al. Transcription factor Tox2 is required for metabolic adaptation and tissue residency of ILC3 in the gut. Immunity. 2024;57:1019–36.e9.
Li X, He J, Gao X, Zheng G, Chen C, Chen Y, et al. GPX4 restricts ferroptosis of NKp46(+)ILC3s to control intestinal inflammation. Cell Death Dis. 2024;15:687.
Masaki T. Historical review: endothelin. Trends Pharm Sci. 2004;25:219–24.
Cheluvappa R, Eri R, Luo AS, Grimm MC. Endothelin and vascular remodeling in colitis pathogenesis-appendicitis and appendectomy limit colitis by suppressing endothelin pathways. Int J Colorectal Dis. 2014;29:1321–8.
Koroleva EP, Halperin S, Gubernatorova EO, Macho-Fernandez E, Spencer CM, Tumanov AV. Citrobacter rodentium-induced colitis: a robust model to study mucosal immune responses in the gut. J Immunol Methods. 2015;421:61–72.
Aujla SJ, Chan YR, Zheng M, Fei M, Askew DJ, Pociask DA, et al. IL-22 mediates mucosal host defense against gram-negative bacterial pneumonia. Nat Med. 2008;14:275–81.
Lee S, Carter PR, Watts MN, Bao JR, Harris NR. Effects of the endothelin-converting enzyme inhibitor SM-19712 in a mouse model of dextran sodium sulfate-induced colitis. Inflamm Bowel Dis. 2009;15:1007–13.
Xu H, Wang Y, Wang W, Fu YX, Qiu J, Shi Y, et al. ILC3s promote intestinal tuft cell hyperplasia and anthelmintic immunity through RANK signaling. Sci Immunol. 2025;10:eadn1491.
Ni L, Lu Q, Tang M, Tao L, Zhao H, Zhang C, et al. Periplaneta americana extract ameliorates dextran sulfate sodium-induced ulcerative colitis via immunoregulatory and PI3K/AKT/NF-κB signaling pathways. Inflammopharmacology. 2022;30:907–18.
Ma Y, Lang X, Yang Q, Han Y, Kang X, Long R, et al. Paeoniflorin promotes intestinal stem cell-mediated epithelial regeneration and repair via PI3K-AKT-mTOR signaling in ulcerative colitis. Int Immunopharmacol. 2023;119:110247.
Zhang X, Zhang F, Li Y, Fan N, Zhao K, Zhang A, et al. Blockade of PI3K/AKT signaling pathway by astragaloside IV attenuates ulcerative colitis by improving the intestinal epithelial barrier. J Transl Med. 2024;22:406.
Brown JB, Cheresh P, Goretsky T, Managlia E, Grimm GR, Ryu H, et al. Epithelial phosphatidylinositol-3-kinase signaling is required for β-catenin activation and host defense against Citrobacter rodentium infection. Infect Immun. 2011;79:1863–72.
Fachi JL, Pral LP, Dos Santos JAC, Codo AC, de Oliveira S, Felipe JS, et al. Hypoxia enhances ILC3 responses through HIF-1α-dependent mechanism. Mucosal Immunol. 2021;14:828–41.
Xue X, Ramakrishnan S, Anderson E, Taylor M, Zimmermann EM, Spence JR, et al. Endothelial PAS domain protein 1 activates the inflammatory response in the intestinal epithelium to promote colitis in mice. Gastroenterology. 2013;145:831–41.
Wang F, Kim BS. Itch: a paradigm of neuroimmune crosstalk. Immunity. 2020;52:753–66.
Tamari M, Ver Heul AM, Kim BS. Immunosensation: neuroimmune cross talk in the skin. Annu Rev Immunol. 2021;39:369–93.
Feuillet V, Ugolini S, Reynders A. Differential regulation of cutaneous immunity by sensory neuron subsets. Trends Neurosci. 2023;46:640–53.
Rafii S, Butler JM, Ding BS. Angiocrine functions of organ-specific endothelial cells. Nature. 2016;529:316–25.
Stürzl M, Kunz M, Krug SM, Naschberger E. Angiocrine regulation of epithelial barrier integrity in inflammatory bowel disease. Front Med. 2021;8:643607.
Andonegui G, Zhou H, Bullard D, Kelly MM, Mullaly SC, McDonald B, et al. Mice that exclusively express TLR4 on endothelial cells can efficiently clear a lethal systemic gram-negative bacterial infection. J Clin Invest. 2009;119:1921–30.
Carrega P, Loiacono F, Di Carlo E, Scaramuccia A, Mora M, Conte R, et al. NCR(+)ILC3 concentrate in human lung cancer and associate with intratumoral lymphoid structures. Nat Commun. 2015;6:8280.
Lai D, Tang J, Chen L, Fan EK, Scott MJ, Li Y, et al. Group 2 innate lymphoid cells protect lung endothelial cells from pyroptosis in sepsis. Cell Death Dis. 2018;9:369.
Silberger DJ, Zindl CL, Weaver CT. Citrobacter rodentium: a model enteropathogen for understanding the interplay of innate and adaptive components of type 3 immunity. Mucosal Immunol. 2017;10:1108–17.
Bando JK, Colonna M. Innate lymphoid cell function in the context of adaptive immunity. Nat Immunol. 2016;17:783–9.
Ahlfors H, Morrison PJ, Duarte JH, Li Y, Biro J, Tolaini M, et al. IL-22 fate reporter reveals origin and control of IL-22 production in homeostasis and infection. J Immunol. 2014;193:4602–13.
Basu R, O’Quinn DB, Silberger DJ, Schoeb TR, Fouser L, Ouyang W, et al. Th22 cells are an important source of IL-22 for host protection against enteropathogenic bacteria. Immunity. 2012;37:1061–75.
Konieczny P, Xing Y, Sidhu I, Subudhi I, Mansfield KP, Hsieh B, et al. Interleukin-17 governs hypoxic adaptation of injured epithelium. Science. 2022;377:eabg9302.
Kierans SJ, Taylor CT. Regulation of glycolysis by the hypoxia-inducible factor (HIF): implications for cellular physiology. J Physiol. 2021;599:23–37.
Moon JS, Hisata S, Park MA, DeNicola GM, Ryter SW, Nakahira K, et al. mTORC1-induced HK1-dependent glycolysis regulates NLRP3 inflammasome activation. Cell Rep. 2015;12:102–15.
Xie M, Yu Y, Kang R, Zhu S, Yang L, Zeng L, et al. PKM2-dependent glycolysis promotes NLRP3 and AIM2 inflammasome activation. Nat Commun. 2016;7:13280.
Sun R, Gu X, Lei C, Chen L, Chu S, Xu G, et al. Neutral ceramidase-dependent regulation of macrophage metabolism directs intestinal immune homeostasis and controls enteric infection. Cell Rep. 2022;38:110560.
Serafini N, Jarade A, Surace L, Goncalves P, Sismeiro O, Varet H, et al. Trained ILC3 responses promote intestinal defense. Science. 2022;375:859–63.
Xiong L, Helm EY, Dean JW, Sun N, Jimenez-Rondan FR, Zhou L. Nutrition impact on ILC3 maintenance and function centers on a cell-intrinsic CD71-iron axis. Nat Immunol. 2023;24:1671–84.
Zhao R, Yang J, Zhai Y, Zhang H, Zhou Y, Hong L, et al. Nucleophosmin 1 promotes mucosal immunity by supporting mitochondrial oxidative phosphorylation and ILC3 activity. Nat Immunol. 2024;25:1565–79.
Bal SM, Golebski K, Spits H. Plasticity of innate lymphoid cell subsets. Nat Rev Immunol. 2020;20:552–65.
Cella M, Gamini R, Sécca C, Collins PL, Zhao S, Peng V, et al. Subsets of ILC3-ILC1-like cells generate a diversity spectrum of innate lymphoid cells in human mucosal tissues. Nat Immunol. 2019;20:980–91.
Krzywinska E, Sobecki M, Nagarajan S, Zacharjasz J, Tambuwala MM, Pelletier A, et al. The transcription factor HIF-1α mediates plasticity of NKp46+ innate lymphoid cells in the gut. J Exp Med. 2022;219:e20210909.
Manosalva C, Quiroga J, Hidalgo AI, Alarcón P, Anseoleaga N, Hidalgo MA, et al. Role of lactate in inflammatory processes: friend or foe. Front Immunol. 2021;12:808799.
Halestrap AP. The monocarboxylate transporter family-structure and functional characterization. IUBMB Life. 2012;64:1–9.
Taylor SJ, Winter MG, Gillis CC, Silva LAD, Dobbins AL, Muramatsu MK, et al. Colonocyte-derived lactate promotes E. coli fitness in the context of inflammation-associated gut microbiota dysbiosis. Microbiome. 2022;10:200.
Végran F, Boidot R, Michiels C, Sonveaux P, Feron O. Lactate influx through the endothelial cell monocarboxylate transporter MCT1 supports an NF-κB/IL-8 pathway that drives tumor angiogenesis. Cancer Res. 2011;71:2550–60.
Cao Y, Li Y, Wang X, Liu S, Zhang Y, Liu G, et al. Dopamine inhibits group 2 innate lymphoid cell-driven allergic lung inflammation by dampening mitochondrial activity. Immunity. 2023;56:320–35.e9.
Xing Z, Li X, He J, Chen Y, Zhu L, Zhang X, et al. OLFM4 modulates intestinal inflammation by promoting IL-22(+)ILC3 in the gut. Commun Biol. 2024;7:914.
Wang Y, Wang J, Liu G, Yi X, Wu J, Cao H, et al. NRP1 instructs IL-17-producing ILC3s to drive colitis progression. Cell Mol Immunol. 2025;22:161–75.
Zeng B, Huang Y, Chen S, Xu R, Xu L, Qiu J, et al. Dextran sodium sulfate potentiates NLRP3 inflammasome activation by modulating the KCa3.1 potassium channel in a mouse model of colitis. Cell Mol Immunol. 2022;19:925–43.
Acknowledgements
We would like to express our sincere gratitude to Professor C. Dong from Tsinghua University for the generous gift of Rorccre mice. We also extend our appreciation to Professor Z. Li from Southern Medical University for kindly providing the CD45.1 mice. The flow cytometry used in this work was supported by the Department of Immunology at the School of Basic Medical Sciences, Southern Medical University.
Funding
This work was supported by grants from the National Natural Science Foundation of China (Nos. 82171706 and 82471736 to YMH), the Guangdong Basic and Applied Basic Research Foundation (Nos. 2022A1515140172 and 2024A1515012897 to YMH), and the Open Fund Project of Guangdong Academy of Medical Sciences (No. YKY-KF202209 to YMH).
Author information
Authors and Affiliations
Contributions
XML, YXG, and YMH conceived the project and designed the research studies. XYL, YMC, JYH, and JT performed most of the experiments described. CLC, LYM, and YZL provided help with the technical assistance and conceptual advice in the experiments. YMH, XYL, and YMC prepared the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The corresponding research plan and ethical approval were provided by the Clinical Trial Ethics Committee of the Sixth Affiliated Hospital of Sun Yat-sen University (Approval ID: E2022243). All animal experimental protocols were approved by the Institutional Animal Care and Use Committee of Southern Medical University Experimental Animal Ethics Committee (Approval number: L2022126).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Chen, Y., He, J. et al. Endothelial cell–ILC3 crosstalk via the ET-1/EDNRA axis promotes NKp46+ILC3 glycolysis to alleviate intestinal inflammation. Cell Mol Immunol 22, 1459–1477 (2025). https://doi.org/10.1038/s41423-025-01345-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41423-025-01345-z