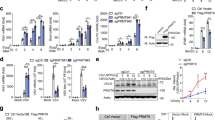
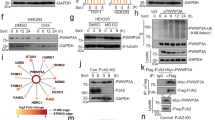
Abstract
Innate immunity in host cells must be rapidly activated to combat invading microbes. Upon RIG-I activation, the transcription of type I interferons is induced within one hour in virus-infected cells. Previous studies have shown that endogenous MAVS spreads signals via aggregation on the mitochondrial membrane, whereas truncated recombinant MAVS forms prion-like filaments in vitro. How MAVS transmits signals so quickly, and the molecular architecture of its membrane aggregates, remains elusive. Here, we report that activated MAVS forms fibrils encircling its resident mitochondrion or connecting neighboring mitochondria with a “ladder-like” structure, allowing the activation of dormant MAVS on encountered mitochondria. This “intermitochondrial activation” process promotes a rapid antiviral response in cells to overcome the immediate danger caused by viruses. Moreover, stuck MAVS fibrils between mitochondria have limited cytosolic protein access and thus relay signals poorly. This study demonstrated that prion-like MAVS fibrils cluster in mitochondria to ensure a rapid antiviral response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Yoneyama M, et al. The RNA helicase RIG-I has an essential function in double-stranded RNA-induced innate antiviral responses. Nat Immunol. 2004;5:730–7.
Chow KT, Gale M Jr., Loo YM. RIG-I and Other RNA Sensors in Antiviral Immunity. Annu Rev Immunol. 2018;36:667–94.
Rehwinkel J, Gack MU. RIG-I-like receptors: their regulation and roles in RNA sensing. Nat Rev Immunol. 2020;20:537–51.
Seth RB, et al. Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-kappaB and IRF 3. Cell. 2005;122:669–82.
Xu LG, et al. VISA is an adapter protein required for virus-triggered IFN-beta signaling. Mol Cell. 2005;19:727–40.
Kumar H, et al. Essential role of IPS-1 in innate immune responses against RNA viruses. J Exp Med. 2006;203:1795–803.
Meylan E, et al. Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nature. 2005;437:1167–72.
Vazquez C, Horner SM. MAVS Coordination of Antiviral Innate Immunity. J Virol. 2015;89:6974–7.
Zeng W, et al. Key role of Ubc5 and lysine-63 polyubiquitination in viral activation of IRF3. Mol Cell. 2009;36:315–25.
Zeng WW, et al. Reconstitution of the RIG-I Pathway Reveals a Signaling Role of Unanchored Polyubiquitin Chains in Innate Immunity. Cell. 2010;141:315–30.
Hou, FJ, et al., MAVS Forms Functional Prion-like Aggregates to Activate and Propagate Antiviral Innate Immune Response (vol 146, pg 448, 2011). Cell, 2011. 146 p. 841–841.
Song B, et al. Ordered assembly of the cytosolic RNA-sensing MDA5-MAVS signaling complex by binding to unanchored K63-linked poly-ubiquitin chains. Immunity. 2021;54:2218–2230 e5.
Liu S, et al. Phosphorylation of innate immune adaptor proteins MAVS, STING, and TRIF induces IRF3 activation. Science. 2015;347:p. aaa2630.
Liu S, et al. MAVS recruits multiple ubiquitin E3 ligases to activate antiviral signaling cascades. Elife. 2013;2:e00785.
Cai X, et al. Prion-like polymerization underlies signal transduction in antiviral immune defense and inflammasome activation. Cell. 2014;156:1207–22.
Wu B, et al. Molecular imprinting as a signal-activation mechanism of the viral RNA sensor RIG-I. Mol Cell. 2014;55:511–23.
Xu H, et al. Structural basis for the prion-like MAVS filaments in antiviral innate immunity. Elife. 2014;3:e01489.
Xu, H, et al. Correction: Structural basis for the prion-like MAVS filaments in antiviral innate immunity. Elife, 2015. 4.
Vaquer-Alicea J, Diamond MI. Propagation of Protein Aggregation in Neurodegenerative Diseases. Annu Rev Biochem. 2019;88:785–810.
Hwang MS, et al. MAVS polymers smaller than 80 nm induce mitochondrial membrane remodeling and interferon signaling. Febs J. 2019;286:1543–60.
Fang R, et al. MAVS activates TBK1 and IKKepsilon through TRAFs in NEMO dependent and independent manner. PLoS Pathog. 2017;13:e1006720.
You FP, et al. PCBP2 mediates degradation of the adaptor MAVS via the HECT ubiquitin ligase AIP4. Nat Immunol. 2009;10:1300–U10.
Mahamid J, et al. Visualizing the molecular sociology at the HeLa cell nuclear periphery. Science. 2016;351:969–72.
Oikonomou CM, Jensen GJ. Cellular Electron Cryotomography: Toward Structural Biology In Situ. Annu Rev Biochem. 2017;86:873–96.
Cai X, Chen ZJ. Prion-like polymerization as a signaling mechanism. Trends Immunol. 2014;35:622–30.
He L, et al. Structure determination of helical filaments by solid-state NMR spectroscopy. Proc Natl Acad Sci USA. 2016;113:E272–81.
Dixit E, et al. Peroxisomes Are Signaling Platforms for Antiviral Innate Immunity. Cell. 2010;141:668–81.
Thoresen DT, et al. A rapid RIG-I signaling relay mediates efficient antiviral response. Mol Cell. 2023;83:90.
Zhu J et al. Arginine monomethylation by PRMT7 controls MAVS-mediated antiviral innate immunity. Mol Cell. 2021.
Cormack BP, Valdivia RH, Falkow S. FACS-optimized mutants of the green fluorescent protein (GFP). Gene. 1996;173:33–8.
Zacharias DA, et al. Partitioning of lipid-modified monomeric GFPs into membrane microdomains of live cells. Science. 2002;296:913–6.
Ma L, et al. Structural insights into the photoactivation of Arabidopsis CRY2. Nat Plants. 2020;6:1432–8.
Bugaj LJ, et al. Optogenetic protein clustering and signaling activation in mammalian cells. Nat Methods. 2013;10:249–52.
Che DL, et al. The Dual Characteristics of Light-Induced Cryptochrome 2, Homo-oligomerization and Heterodimerization, for Optogenetic Manipulation in Mammalian Cells. ACS Synth Biol. 2015;4:1124–35.
Zhao B, et al. A conserved PLPLRT/SD motif of STING mediates the recruitment and activation of TBK1. Nature. 2019;569:718–22.
Fang R, et al. Golgi apparatus-synthesized sulfated glycosaminoglycans mediate polymerization and activation of the cGAMP sensor STING. Immunity. 2021;54:962–975 e8.
Shang G, et al. Cryo-EM structures of STING reveal its mechanism of activation by cyclic GMP-AMP. Nature. 2019;567:389–93.
Du MJ, Chen ZJJ. DNA-induced liquid phase condensation of cGAS activates innate immune signaling. Science. 2018;361:704.
Ishikawa H, Barber GN. STING is an endoplasmic reticulum adaptor that facilitates innate immune signaling. Nature. 2008;455:674–8.
Zhong B, et al. The adaptor protein MITA links virus-sensing receptors to IRF3 transcription factor activation. Immunity. 2008;29:538–50.
Sun WX, et al. ERIS, an endoplasmic reticulum IFN stimulator, activates innate immune signaling through dimerization. Proc Natl Acad Sci USA. 2009;106:8653–8.
Linehan, MM, et al. A minimal RNA ligand for potent RIG-I activation in living mice. Sci Adv, 2018. 4.
Gonzalez-Garcia M, Fusco G, De Simone A. Membrane Interactions and Toxicity by Misfolded Protein Oligomers. Front Cell Dev Biol. 2021;9:642623.
Fusco G, et al. Structural basis of membrane disruption and cellular toxicity by alpha-synuclein oligomers. Science. 2017;358:1440–3.
Bode DC, Baker MD, Viles JH. Ion Channel Formation by Amyloid-beta42 Oligomers but Not Amyloid-beta40 in Cellular Membranes. J Biol Chem. 2017;292:1404–13.
Camilleri A, et al. Tau-induced mitochondrial membrane perturbation is dependent upon cardiolipin. Biochim Biophys Acta Biomembr. 2020;1862:183064.
Mattiazzi M, et al. Mutated human SOD1 causes dysfunction of oxidative phosphorylation in mitochondria of transgenic mice. J Biol Chem. 2002;277:29626–33.
Israelson A, et al. Misfolded mutant SOD1 directly inhibits VDAC1 conductance in a mouse model of inherited ALS. Neuron. 2010;67:575–87.
Rostovtseva TK, et al. alpha-Synuclein Shows High Affinity Interaction with Voltage-dependent Anion Channel, Suggesting Mechanisms of Mitochondrial Regulation and Toxicity in Parkinson Disease. J Biol Chem. 2015;290:18467–77.
Saxton WM, Hollenbeck PJ. The axonal transport of mitochondria. J Cell Sci. 2012;125:2095–104.
Al-Mehdi AB, et al., Perinuclear Mitochondrial Clustering Creates an Oxidant-Rich Nuclear Domain Required for Hypoxia-Induced Transcription. Sci Signal, 2012. 5.
Agarwal S, Ganesh, S. Perinuclear mitochondrial clustering, increased ROS levels, and HIF1 are required for the activation of HSF1 by heat stress. Journal of Cell Science, 2020. 133.
Vives-Bauza C, et al. PINK1-dependent recruitment of Parkin to mitochondria in mitophagy. Proc Natl Acad Sci USA. 2010;107:378–83.
Huang P, Yu T, Yoon Y. Mitochondrial clustering induced by overexpression of the mitochondrial fusion protein Mfn2 causes mitochondrial dysfunction and cell death. Eur J Cell Biol. 2007;86:289–302.
Duvert M, Mazat JP, Barets AL. Intermitochondrial junctions in the heart of the frog, Rana esculenta. A thin-section and freeze-fracture study. Cell Tissue Res. 1985;241:129–37.
Picard M, et al. Trans-mitochondrial coordination of cristae at regulated membrane junctions. Nat Commun. 2015;6:6259.
Skulachev VP. Mitochondrial filaments and clusters as intracellular power-transmitting cables. Trends Biochem Sci. 2001;26:23–9.
Heine KB, Hood WR. Mitochondrial behavior, morphology, and animal performance. Biol Rev Camb Philos Soc. 2020;95:730–7.
Vernay A, et al. MitoNEET-dependent formation of intermitochondrial junctions. Proc Natl Acad Sci USA. 2017;114:8277–82.
Trinkaus VA, et al. In situ architecture of neuronal alpha-Synuclein inclusions. Nat Commun. 2021;12:2110.
Bauerlein FJB, et al. In Situ Architecture and Cellular Interactions of PolyQ Inclusions. Cell. 2017;171:179–187 e10.
Chen HH, et al. Activation of STAT6 by STING Is Critical for Antiviral Innate Immunity. Cell. 2011;147:436–46.
Fang R, et al. NEMO-IKK beta Are Essential for IRF3 and NF-kappa B Activation in the cGAS-STING Pathway. J Immunol. 2017;199:3222–33.
Lau CH, Tin C, Suh Y. CRISPR-based strategies for targeted transgene knock-in and gene correction. Fac Rev. 2020;9:20.
Guo Q, et al. In Situ Structure of Neuronal C9orf72 Poly-GA Aggregates Reveals Proteasome Recruitment. Cell. 2018;172:696–705 e12.
Mastronarde DN. Automated electron microscope tomography using robust prediction of specimen movements. J Struct Biol. 2005;152:36–51.
Hagen WJH, Wan W, Briggs JAG. Implementation of a cryo-electron tomography tilt-scheme optimized for high resolution subtomogram averaging. J Struct Biol. 2017;197:191–8.
Zheng SQ, et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat Methods. 2017;14:331–2.
Zhang K. Gctf: Real-time CTF determination and correction. J Struct Biol. 2016;193:1–12.
Kremer JR, Mastronarde DN, McIntosh JR. Computer visualization of three-dimensional image data using IMOD. J Struct Biol. 1996;116:71–6.
Tegunov D, Cramer P. Real-time cryo-electron microscopy data preprocessing with Warp. Nat Methods. 2019;16:1146–52.
Martinez-Sanchez A, et al. Robust membrane detection based on tensor voting for electron tomography. J Struct Biol. 2014;186:49–61.
Wan W, et al. STOPGAP: A Software Package for Subtomogram Averaging and Refinement. Microsc Microanal. 2020;26:2516–2516.
Jiang W, et al. A transformation clustering algorithm and its application in polyribosomes structural profiling. Nucleic Acids Res. 2022;50:9001–11.
Martinez-Sanchez A, et al. Template-free detection and classification of membrane-bound complexes in cryo-electron tomograms. Nat Methods. 2020;17:209–16.
Walt, S.V. d., Colbert, SC, Varoquaux G. The NumPy Array: A Structure for Efficient Numerical Computation. Comput Sci Eng, 2011. 13: p. 22–30.
Goddard TD, et al. UCSF ChimeraX: Meeting modern challenges in visualization and analysis. Protein Sci. 2018;27:14–25.
Martell JD, et al. Electron microscopy using the genetically encoded APEX2 tag in cultured mammalian cells. Nat Protoc. 2017;12:1792–816.
Broussard JA, et al. Fluorescence resonance energy transfer microscopy as demonstrated by measuring the activation of the serine/threonine kinase Akt. Nat Protoc. 2013;8:265–81.
Acknowledgements
We thank members from the Core Facility at the National Center for Protein Sciences at Peking University for assistance with electron microscopy, optical microscopy, and flow cytometry, particularly Drs. Yiqun Liu and Yingchun Hu, Mses Hongmei Zhang, Pengyuan Dong, Rui Jiao, Mrs. Xinpeng He, Hongzhang Zhou, and Zhenyang Kong for their invaluable technical assistance with electron microscopy; Drs. Jun Ren and Chunyan Shan for their invaluable technical assistance with optical microscopy, and Mrs. Liying Du, Dr. Jia Luo, and Dr. Huan Yang for their invaluable technical assistance with flow cytometry. We thank members from the Laboratory of Electron Microscopy and Cryo-EM Platform of Peking University for assistance with cryo-FIB milling and cryo-ET data collection, particularly Drs. Zhenxi Guo and Changdong Qin, Ms. Xia Pei, and Mr. Bo Shao for device setup. We thank the high-performance computing platform of Peking University for assisting with the computational resources. We thank the Center for Biological Imaging (CBI), Institute of Biophysics, for assisting with cryo-ET data collection. We thank Congyi Zhen for the viruses, Liang Li for the modifications of the calculation-related Python scripts, and Junhan Yang and Shenjia Luo for assisting in image processing.
Funding
This work was supported by the National Natural Science Foundation of China (32130038), the Chinese Ministry of Science and Technology (2024YFA1306501 and 2022YFC2303700), the China Postdoctoral Science Foundation (2021M700242 and 2022T150017), and the Young Elite Scientists Sponsorship Program by CAST (2022QNRC001).
Author information
Authors and Affiliations
Contributions
XY, PW, QG, and ZJ designed the research; XY and PW performed the experiments; LZ, NZ, and XG assisted in the experiments; XY, PW, QG, and ZJ analyzed the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, X., Wang, P., Zhou, N. et al. Prion-like MAVS fibrils stitch mitochondria to promote a rapid antiviral response. Cell Mol Immunol 23, 204–219 (2026). https://doi.org/10.1038/s41423-025-01382-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41423-025-01382-8