Abstract
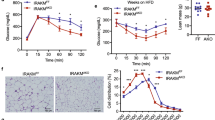
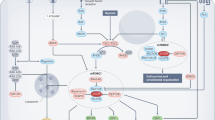
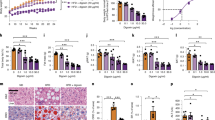
Group 2 innate lymphoid cells (ILC2s) play crucial roles in maintaining adipose tissue homeostasis. Recent studies indicate that ILC2s are dysregulated in obesity. However, the regulatory mechanisms governing adipose tissue ILC2 function remain inadequately explored. In this study, we demonstrated that mechanistic target of rapamycin complex 1 (mTORC1) activity is impaired in adipose tissue ILC2s from obese mice and humans. Deletion of Raptor, a critical adaptor protein in mTORC1, results in reduced numbers of ILC2s and diminished type 2 cytokine production in ILC2s, leading to increased adipose tissue inflammation and insulin resistance. Mechanistically, mTORC1 signaling upregulates PPARγ expression through HIF-1α, which promotes mitochondrial biogenesis and ST2 expression to sustain ILC2 metabolic and functional fitness. Together, our data identify mTORC1 as a crucial regulator that coordinates adipose tissue ILC2 metabolic and immunological homeostasis and prevents obesity-associated insulin resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The raw reads from the RNA-seq data have been deposited at the Gene Expression Omnibus (GEO) repository under the accession numbers GSE225097 and GSE287676. All data supporting the conclusions are available from the authors upon reasonable request.
References
Czech MP. Insulin action and resistance in obesity and type 2 diabetes. Nat Med. 2017;23:804–14.
Powell-Wiley TM, Poirier P, Burke LE, Després J-P, Gordon-Larsen P, Lavie CJ, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143:e984–e1010.
Stefan N, Cusi K. A global view of the interplay between nonalcoholic fatty liver disease and diabetes. Lancet Diab Endocrinol. 2022;10:284–96.
Olefsky JM, Glass CK. Macrophages, inflammation, and insulin resistance. Annu Rev Physiol. 2010;72:219–46.
Feuerer M, Herrero L, Cipolletta D, Naaz A, Wong J, Nayer A, et al. Lean, but not obese, fat is enriched for a unique population of regulatory T cells that affect metabolic parameters. Nat Med. 2009;15:930–9.
Nishimura S, Manabe I, Nagasaki M, Eto K, Yamashita H, Ohsugi M, et al. CD8+ effector T cells contribute to macrophage recruitment and adipose tissue inflammation in obesity. Nat Med. 2009;15:914–20.
Ji Z, Wu S, Xu Y, Qi J, Su X, Shen L. Obesity Promotes EAE Through IL-6 and CCL-2-Mediated T Cells Infiltration. Front Immunol. 2019;10:1881.
Wang H, Shen L, Sun X, Liu F, Feng W, Jiang C, et al. Adipose group 1 innate lymphoid cells promote adipose tissue fibrosis and diabetes in obesity. Nat Commun. 2019;10:3254.
Sun J, Zhang Y, Zhang Q, Hu L, Zhao L, Wang H, et al. Metabolic regulator LKB1 controls adipose tissue ILC2 PD-1 expression and mitochondrial homeostasis to prevent insulin resistance. Immunity. 2024;57:1289–305.e9.
McLaughlin T, Ackerman SE, Shen L & Engleman E. Role of innate and adaptive immunity in obesity-associated metabolic disease. J Clin Invest. 2017;127:5–13.
Shen L, Chng MHY, Alonso MN, Yuan R, Winer DA, Engleman EG. B-1a lymphocytes attenuate insulin resistance. Diabetes. 2015;64:593–603.
Winer DA, Winer S, Shen L, Wadia PP, Yantha J, Paltser G, et al. B cells promote insulin resistance through modulation of T cells and production of pathogenic IgG antibodies. Nat Med. 2011;17:610–7.
Zhang J, Qiu J, Zhou W, Cao J, Hu X, Mi W, et al. Neuropilin-1 mediates lung tissue-specific control of ILC2 function in type 2 immunity. Nat Immunol. 2022;23:237–50.
Brestoff JR, Kim BS, Saenz SA, Stine RR, Monticelli LA, Sonnenberg GF, et al. Group 2 innate lymphoid cells promote beiging of white adipose tissue and limit obesity. Nature. 2015;519:242–6.
Oldenhove G, Boucquey E, Taquin A, Acolty V, Bonetti L, Ryffel B, et al. PD-1 is involved in the dysregulation of type 2 innate lymphoid cells in a murine model of obesity. Cell Rep. 2018;25:2053–60.e4.
Hildreth AD, Ma F, Wong YY, Sun R, Pellegrini M, O’Sullivan TE. Single-cell sequencing of human white adipose tissue identifies new cell states in health and obesity. Nat Immunol. 2021;22:639–53.
Flach M, Diefenbach A. Adipose tissue: ILC2 crank up the heat. Cell Metab. 2015;21:152–3.
Lee M-W, Odegaard JustinI, Mukundan L, Qiu Y, Molofsky AriB, Nussbaum JesseC, et al. Activated type 2 innate lymphoid cells regulate beige fat biogenesis. Cell. 2015;160:74–87.
Ben-Sahra I, Manning BD. mTORC1 signaling and the metabolic control of cell growth. Curr Opin Cell Biol. 2017;45:72–82.
Huang H, Long L, Zhou P, Chapman NM, Chi H. mTOR signaling at the crossroads of environmental signals and T-cell fate decisions. Immunol Rev. 2020;295:15–38.
Tang J, Yang L, Guan F, Miller H, Camara NOS, James LK, et al. The role of Raptor in lymphocytes differentiation and function. Front Immunol. 2023;14:1146628.
Yang K, Shrestha S, Zeng H, Karmaus PWF, Neale G, Vogel P, et al. T-cell exit from quiescence and differentiation into Th2 cells depend on Raptor-mTORC1-mediated metabolic reprogramming. Immunity. 2013;39:1043–56.
Salmond RJ, Mirchandani AS, Besnard AG, Bain CC, Thomson NC, Liew FY. IL-33 induces innate lymphoid cell-mediated airway inflammation by activating mammalian target of rapamycin. J Allergy Clin Immunol. 2012;130:1159–66.e6.
Surace LA-O, Doisne JA-O, Croft CA-O, Thaller A, Escoll PA-OX, Marie S, et al. Dichotomous metabolic networks govern human ILC2 proliferation and function. Nat Immunol. 2021;22:1367–74.
Galbraith LCA, Mui E, Nixon C, Hedley A, Strachan D, MacKay G, et al. PPAR-gamma induced AKT3 expression increases levels of mitochondrial biogenesis driving prostate cancer. Oncogene. 2021;40:2355–66.
Karagiannis F, Masouleh SK, Wunderling K, Surendar J, Schmitt V, Kazakov A, et al. Lipid-droplet formation drives pathogenic group 2 innate lymphoid cells in airway inflammation. Immunity. 2020;52:885.
Xiao Q, He J, Lei A, Xu H, Zhang L, Zhou P, et al. PPARγ enhances ILC2 function during allergic airway inflammation via transcription regulation of ST2. Mucosal Immunol. 2021;14:468–78.
Fali T, Aychek T, Ferhat M, Jouzeau J-Y, Busslinger M, Moulin D, et al. Metabolic regulation by PPARγ is required for IL-33-mediated activation of ILC2s in lung and adipose tissue. Mucosal Immunol. 2021;14:585–93.
Cho HY, Gladwell W, Wang X, Chorley B, Bell D, Reddy SP Wang X, Wang X, Fau -, et al. Nrf2-regulated PPAR{gamma} expression is critical to protection against acute lung injury in mice. Am J Respir Crit Care Med. 2010;182:170–82.
Zhan L, Zhang H, Zhang Q, Woods CG, Chen Y, Xue P, et al. Regulatory role of KEAP1 and NRF2 in PPARγ expression and chemoresistance in human non-small cell lung carcinoma cells. Free Radic Biol Med. 2012;53:758–68.
Li YA-O, Lu YA-O, Lin SH, Li N, Han YA-O, Huang Q, et al. Insulin signaling establishes a developmental trajectory of adipose regulatory T cells. Nat Immunol. 2021;22:1175–85.
Chen Y, He R, Han Z, Wu Y, Wang Q, Zhu X, et al. Cooperation of ATF4 and CTCF promotes adipogenesis through transcriptional regulation. Cell Biol Toxicol. 2022;38:741–63.
Morita M, Gravel S-P, Chénard V, Sikström K, Zheng L, Alain T, et al. mTORC1 controls mitochondrial activity and biogenesis through 4E-BP-dependent translational regulation. Cell Metab. 2013;18:698–711.
Cunningham JT, Rodgers JT, Arlow DH, Vazquez F, Mootha VK, Puigserver P. mTOR controls mitochondrial oxidative function through a YY1-PGC-1alpha transcriptional complex. Nature. 2007;450:736–40.
Sun L, Ji S, Xie X, Si L, Liu S, Lin Y, et al. Deciphering the interaction between Twist1 and PPARγ during adipocyte differentiation. Cell Death Dis. 2023;14:764.
Aprile M, Cataldi S, Ambrosio MR, D’Esposito V, Lim K, Dietrich A, et al. PPARγΔ5, a naturally, occurring dominant-negative splice isoform, impairs PPARγ function and adipocyte differentiation. Cell Rep. 2018;25:1577–92.e6.
Kamon J, Yamauchi T, Terauchi Y, Kubota N, Kadowaki T. The mechanisms by which PPARgamma and adiponectin regulate glucose and lipid metabolism]. Nihon Yakurigaku Zasshi. 2003;122:294–300.
Festuccia WT, Blanchard P-G & Deshaies Y. Control of brown adipose tissue glucose and lipid metabolism by PPARγ. Front Endocrinol. 2011;2:84.
Ahmadian M, Suh JM, Hah N, Liddle C, Atkins AR, Downes M, et al. PPARγ signaling and metabolism: the good, the bad and the future. Nat Med. 2013;19:557–66.
Honda K, Marquillies P, Capron M, Dombrowicz D. Peroxisome proliferator-activated receptor gamma is expressed in airways and inhibits features of airway remodeling in a mouse asthma model. J Allergy Clin Immunol. 2004;113:882–8.
Benayoun L, Letuve S, Druilhe A, Boczkowski J, Dombret MC, Mechighel P, et al. Regulation of peroxisome proliferator-activated receptor gamma expression in human asthmatic airways: relationship with proliferation, apoptosis, and airway remodeling. Am J Respir Crit Care Med. 2001;164:1487–94.
Nobs SP, Natali S, Pohlmeier L, Okreglicka K, Schneider C, Kurrer M, et al. PPARγ in dendritic cells and T cells drives pathogenic type-2 effector responses in lung inflammation. J Exp Med. 2017;214:3015–35.
Chen T, Tibbitt CA, Feng X, Stark JM, Rohrbeck L, Rausch L, et al. PPAR-γ promotes type 2 immune responses in allergy and nematode infection. Sci Immunol. 2017;2:eaal5196.
Steiner CA, Janez A, Jensterle M, Reisinger K, Forst T, Pfützner A. Impact of treatment with rosiglitazone or metformin on biomarkers for insulin resistance and metabolic syndrome in patients with polycystic ovary syndrome. J Diab Sci Technol. 2007;1:211–7.
Tfayli H, Ulnach JW, Lee S, Sutton-Tyrrell K, Arslanian S. Drospirenone/ethinyl estradiol versus rosiglitazone treatment in overweight adolescents with polycystic ovary syndrome: comparison of metabolic, hormonal, and cardiovascular risk factors. J Clin Endocrinol Metab. 2011;96:1311–9.
Wagstaff AJ, Goa KL. Rosiglitazone: a review of its use in the management of type 2 diabetes mellitus. Drugs. 2002;62:1805–37.
Deeks ED, Keam SJ. Rosiglitazone : a review of its use in type 2 diabetes mellitus. Drugs. 2007;67:2747–79.
Buettner R, Schölmerich J, Bollheimer LC. High-fat diets: modeling the metabolic disorders of human obesity in rodents. Obesity. 2007;15:798–808.
de Moura EDM, Dos Reis SA, da Conceição LL, Sediyama C, Pereira SS, de Oliveira LL, et al. Diet-induced obesity in animal models: points to consider and influence on metabolic markers. Diabetol Metab Syndr. 2021;13:32.
Acknowledgements
We thank all the members from the Shen laboratory for their help and suggestions on this project. We thank the Flow Cytometry Facility at the Shanghai Institute of Immunology and Animal Facility at Shanghai Jiao Tong University School of Medicine for their service and assistance. This study was supported by grants 32250710147 and 81971487 (to L.S.) from the National Natural Science Foundation of China, grant 2020YFA0509200 (to L.S.) from the Ministry of Science and Technology of China, and grant 23ZR1455100 (to L.S.) from the Science and Technology Commission of Shanghai Municipality.
Author information
Authors and Affiliations
Contributions
LH and DW designed and performed the experiments, analyzed the data, and drafted the manuscript. YC, JQ, and MZ performed some of the experiments. H. contributed to human sample collection. YZ and JQ contributed to the discussion. LS and JS wrote the manuscript. LS conceived, designed, and supervised the project. All the authors discussed, revised, and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no conflicting interests. J.Q. is an editorial board member of Cellular & Molecular Immunology, but she has not been involved in peer review or decision-making related to the article.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, L., Wang, D., Chen, Y. et al. Obesity disrupts ILC2 metabolic and functional homeostasis by inhibiting mTORC1 signaling. Cell Mol Immunol 23, 301–314 (2026). https://doi.org/10.1038/s41423-026-01389-9
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41423-026-01389-9