Abstract
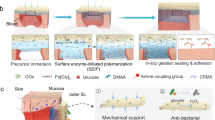
Anastomotic leakage remains a serious complication in gastrointestinal surgery, with unsatisfactory improvement in incidence over recent decades. Conventional sealants, such as fibrin glue, require intraoperative mixing and offer limited ease of handling. Here, we report a ready-to-use, thixotropic hydrogel that is injectable during application and solidifies upon contact with tissue. The hydrogel consists of self-assembled nanofibers of an elastin-like polypeptide crosslinked with oxidized dextran (OD). Rheological step-strain measurements demonstrated reversible sol–gel transitions and shear-responsive behavior, with the most highly oxidized formulation exhibiting the greatest recovery of the storage modulus (44.2%) after high strain. This formulation also showed adhesive strength comparable to fibrin glue in lap-shear tests and a burst pressure (14.8 ± 3.6 kPa) comparable to that of fibrin glue (15.4 ± 4.8 kPa) on porcine small intestine. In vivo experiments in mice demonstrated that the hydrogel remained localized at the injection site up to one week after intraperitoneal administration, indicating favorable physiological stability. Histological analysis revealed only mild serosal thickening without severe inflammation. These findings suggest that this shear-thinning, self-healing hydrogel may serve as a practical and effective sealant for surgical reinforcement and could contribute to reducing the risk of anastomotic leakage in gastrointestinal surgery.
Similar content being viewed by others
References
Fang, A. H., Chao, W. & Ecker, M. Review of colonic anastomotic leakage and prevention methods. J. Clin. Med. 9, 4061 (2020).
van Helsdingen, C. P. M., Jongen, A. C. H. M., de Jonge, W. J., Bouvy, N. D. & Derikx, J. P. M. Consensus on the definition of colorectal anastomotic leakage: a modified Delphi study. World J. Gastroenterol. 26, 3293–3303 (2020).
Rennie, O., Sharma, M. & Helwa, N. Colorectal anastomotic leakage: a narrative review of definitions, grading systems, and consequences of leaks. Front. Surg. 11, 1371567 (2024).
Nordentoft, T., Pommergaard, H.-C., Rosenberg, J. & Achiam, M. P. Fibrin glue does not improve healing of gastrointestinal anastomoses: a systematic review. Eur. Surg. Res. 54, 1–13 (2014).
Nguyen, N. T., Nguyen, C. T., Melinda Stevens, C., Steward, E. & Paya, M. The efficacy of fibrin sealant in prevention of anastomotic leak after laparoscopic gastric bypass. J. Surg. Res. 122, 218–224 (2004).
Anthis, A. H. C. et al. Chemically stable, strongly adhesive sealant patch for intestinal anastomotic leakage prevention. Adv. Funct. Mater. 31, 2007099 (2021).
Pramanik, B. Short peptide-based smart thixotropic hydrogels. Gels 8, 569 (2022).
Ouyang, C. et al. Tough adhesion enhancing strategies for injectable hydrogel adhesives in biomedical applications. Adv. Colloid Interface Sci. 319, 102982 (2023).
Nagahama, K. et al. Thixotropic β-chitin nanofiber hydrogel with a living body-responsive self-hardening property and its application as a sprayable antiadhesion barrier. ACS Appl. Bio Mater. 6, 2636–2643 (2023).
Shin, H. H. & Ryu, J. H. Bio-inspired self-healing, shear-thinning, and adhesive gallic acid-conjugated chitosan/carbon black composite hydrogels as suture support materials. Biomimetics 8, 542 (2023).
Sugioka, Y., Nakamura, J., Ohtsuki, C. & Sugawara-Narutaki, A. Thixotropic hydrogels composed of self-assembled nanofibers of double-hydrophobic elastin-like block polypeptides. Int. J. Mol. Sci. 22, 4104 (2021).
Sugawara-Narutaki, A. Self-assembled nanofibers and hydrogels of double-hydrophobic elastin-like polypeptides formed via coacervation. Polym. J. 57, 863–871 (2025).
Sugawara-Narutaki, A. et al. Rheology of dispersions of high-aspect-ratio nanofibers assembled from elastin-like double-hydrophobic polypeptides. Int. J. Mol. Sci. 20, 6262 (2019).
Natsume, K., Nakamura, J., Sato, K., Ohtsuki, C. & Sugawara-Narutaki, A. Biological properties of self-assembled nanofibers of elastin-like block polypeptides for tissue-engineered vascular grafts: platelet inhibition, endothelial cell activation and smooth muscle cell maintenance. Regen. Biomater. 10, rbac111 (2023).
Le, D. H. T. et al. Double-hydrophobic elastin-like polypeptides with added functional motifs: Self-assembly and cytocompatibility. J. Biomed. Mater. Res. A 105, 2475–2484 (2017).
Le, D. H. T. & Sugawara-Narutaki, A. Elastin-like polypeptides as building motifs toward designing functional nanobiomaterials. Mol. Syst. Des. Eng. 4, 545–565 (2019).
Han, G. -Y. et al. Progress of polysaccharide-based tissue adhesives. Carbohyd. Polym. 327, 121634 (2024).
Du, X. et al. Injectable hydrogel composed of hydrophobically modified chitosan/oxidized-dextran for wound healing. Mater. Sci. Eng. C 104, 109930 (2019).
Chen, Z. et al. An injectable anti-microbial and adhesive hydrogel for the effective noncompressible visceral hemostasis and wound repair. Mater. Sci. Eng. C 129, 112422 (2021).
Li, Z. et al. Ultrafast in-situ forming halloysite nanotube-doped chitosan/oxidized dextran hydrogels for hemostasis and wound repair. Carbohyd. Polym. 267, 118155 (2021).
Muñoz Taboada, G., Dahis, D., Dosta, P., Edelman, E. & Artzi, N. Sprayable hydrogel sealant for gastrointestinal wound shielding. Adv. Mater. 36, 2311798 (2024).
Chen, X. et al. Injectable dopamine–polysaccharide in situ composite hydrogels with enhanced adhesiveness. ACS Biomater. Sci. Eng. 9, 427–436 (2023).
Lin, X., Lv, J., Wang, D. & Liu, K. Injectable adhesive carboxymethyl chitosan-based hydrogels with self-mending and antimicrobial features for the potential management of periodontal diseases. RSC Adv. 13, 11903–11911 (2023).
Chen, Z. et al. A triple-network carboxymethyl chitosan-based hydrogel for hemostasis of incompressible bleeding on wet wound surfaces. Carbohyd. Polym. 303, 120434 (2023).
Zhu, Z. et al. A choline phosphoryl-conjugated chitosan/oxidized dextran injectable self-healing hydrogel for improved hemostatic efficacy. Biomacromolecules 24, 690–703 (2023).
Yin, Y. et al. Rosmarinic acid-grafted dextran/gelatin hydrogel as a wound dressing with improved properties: strong tissue adhesion, antibacterial, antioxidant and anti-inflammatory. Molecules 28, 4034 (2023).
Echavarría, A. P., Pagán, J. & Ibarz, A. Melanoidins formed by Maillard reaction in food and their biological activity. Food Eng. Rev. 4, 203–223 (2012).
Wang, H. -Y., Qian, H. & Yao, W. -R. Melanoidins produced by the Maillard reaction: structure and biological activity. Food Chem. 128, 573–584 (2011).
Bundi, A. & Wüthrich, K. 1H-NMR parameters of the common amino acid residues measured in aqueous solutions of the linear tetrapeptides H-Gly-Gly-X-L-Ala-OH. Biopolymers 18, 285–297 (1979).
Navarro, R. S. et al. Tuning polymer hydrophilicity to regulate gel mechanics and encapsulated cell morphology. Adv. Healthc. Mater. 11, 2200011 (2022).
Hofmann, T. 4-Alkylidene-2-imino-5-[4-alkylidene-5-oxo-1,3-imidazol-2-inyl]aza- methylidene-1,3-imidazolidine −A novel colored substructure in melanoidins formed by Maillard reactions of bound arginine with glyoxal and furan-2-carboxaldehyde. J. Agric. Food Chem. 58, 2470–2478 (2010).
Shakoor, A., Zhang, C., Xie, J. & Yang, X. Maillard reaction chemistry in formation of critical intermediates and flavour compounds and their antioxidant properties. Food Chem. 393, 133416 (2022).
Ma, Y. -J. et al. Model studies on the formation of 2-vinylpyrazine and 2-vinyl-6-methylpyrazine in Maillard-type reactions. Food Chem. 374, 131652 (2022).
Lin, Y. -J., Chuang, W. -T. & Hsu, S. Gelation mechanism and structural dynamics of chitosan self-healing hydrogels by in situ SAXS and coherent X-ray scattering. ACS Macro Lett. 8, 1449–1455 (2019).
Wood-Adams, P. M., Dealy, J. M., deGroot, A. W. & Redwine, O. D. Effect of molecular structure on the linear viscoelastic behavior of polyethylene. Macromolecules 33, 7489–7499 (2000).
Basson, M. D. Paradigms for mechanical signal transdution in the intestinal epithelium. Digention 68, 217–225 (2003).
Kim, H. J., Huh, D., Hamilton, G. & Ingber, D. E. Human gut-on-a-chip inhabited by microbial flora that experiences intestinal peristalsis-like motions and flow. Lab. Chip 12, 2165–2174 (2012).
Acknowledgements
This work was supported by JSPS KAKENHI Grant Numbers 23K18312 and 23H04934, the MEXT Program: Data Creation and Utilization-Type Material Research and Development Project (Grant Number JPMXP1122714694), and the MEXT Program: Advanced Research Infrastructure for Materials and Nanotechnology in Japan (ARIM, Grant Number JPMXP1222NU0266). We thank Yoshiharu Sawada (Nagoya University) for assistance in 1H NMR measurements.
Author information
Authors and Affiliations
Contributions
Y.A., M.K., and A.S.-N. conceived and designed the study. Y.A., under the supervision of K.S., C.O., and A.S.-N., performed material preparation, structural analysis, rheological measurements, lap shear tests, and drafted the manuscript. Y.N. was responsible for material preparation and burst pressure measurements. T.S. and K.A. conducted the animal experiments. A.S.-N. wrote the manuscript. M.K., K.S., and C.O. reviewed and edited the manuscript. All authors contributed to the finalization of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aoyama, Y., Nakano, Y., Shinozuka, T. et al. Injectable thixotropic hydrogel composed of elastin-like polypeptide and oxidized dextran for anastomotic support. NPG Asia Mater (2026). https://doi.org/10.1038/s41427-026-00646-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41427-026-00646-7