Abstract
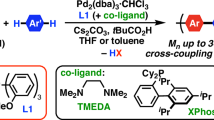
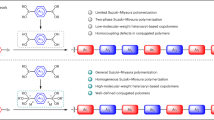
The rise of conjugated polymer (CP)-based modern organic electronics in today’s digital landscape has necessitated the precise synthesis and design of desired CPs and their architectures. This emphasizes the need for constant improvement in synthetic techniques to achieve targeted properties for device applications. In this review, we explore the gradual evolution of various kinds of catalyst transfer polymerization (CTP) methods, including Kumada, Suzuki–Miyaura, Stille, Negishi, Murahashi, and Sonogashira polymerizations, as well as the growing field of direct arylation polymerization (DArP), which exhibit certain aspects of the controlled chain polymerization mechanism, enabling the synthesis of simple CPs to complex architectures with enhanced precision. Our discussion focuses on key advancements within the Kumada and Suzuki–Miyaura catalyst transfer polymerization methods, which have played a significant role in this development. Additionally, we review the ongoing progress in other CTP types. Finally, we address the efforts made by researchers in DArP toward achieving living characteristics, which offer hope for more sustainable and efficient pathways for the precise synthesis of CPs and their structures in the future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout











Similar content being viewed by others
References
Kiriy A, Senkovskyy V, Sommer M. Kumada catalyst-transfer polycondensation: mechanism, opportunities, and challenges. Macromol Rapid Commun. 2011;32:1503–17.
Ding L, Yu Z-D, Wang X-Y, Yao Z-F, Lu Y, Yang C-Y, et al. Polymer semiconductors: synthesis, processing, and applications. Chem Rev. 2023;123:7421–97.
Sheina EE, Liu J, Iovu MC, Laird DW, McCullough RD. Chain growth mechanism for regioregular nickel-initiated cross-coupling polymerizations. Macromolecules. 2004;37:3526–8.
Yokoyama A, Miyakoshi R, Yokozawa T. Chain-growth polymerization for poly(3-hexylthiophene) with a defined molecular weight and a low polydispersity. Macromolecules. 2004;37:1169–71.
Kobayashi M, Chen J, Chung T-C, Moraes F, Heeger AJ, Wudl F. Synthesis and properties of chemically coupled poly(thiophene). Synth Met. 1984;9:77–86.
McCullough RD, Lowe RD. Enhanced electrical conductivity in regioselectively synthesized poly(3-al kylthiophenes). J Chem Soc Chem Commun. 1992;2:70–2.
McCullough RD, Williams SP, Tristram-Nagle S, Jayaraman M, Ewbank PC, Miller L. The first synthesis and new properties of regioregular, head-to-tail coupled polythiophenes. Synth Met. 1995;69:279–82.
Chen T-A, Wu X, Rieke RD. Regiocontrolled synthesis of poly(3-alkylthiophenes) mediated by Rieke zinc: their characterization and solid-state properties. J Am Chem Soc. 1995;117:233–44.
Loewe RS, Khersonsky SM, McCullough RD. A simple method to prepare head-to-tail coupled, regioregular poly(3-alkylthiophenes) using grignard metathesis. Adv Mater. 1999;11:250–3.
Osaka I, Mccullough RD. Advances in molecular design and synthesis of regioregular polythiophenes. Acc Chem Res. 2008;41:1202–14.
Leone AK, Mueller EA, McNeil AJ. The history of palladium-catalyzed cross-couplings should inspire the future of catalyst-transfer polymerization. J Am Chem Soc. 2018;140:15126–39.
Leone AK, McNeil AJ. Matchmaking in catalyst-transfer polycondensation: optimizing catalysts based on mechanistic insight. Acc Chem Res. 2016;49:2822–31.
Verheyen L, Leysen P, Van Den Eede M-P, Ceunen W, Hardeman T, Koeckelberghs G. Advances in the controlled polymerization of conjugated polymers. Polymer. 2017;108:521–46.
Mikami K, Nojima M, Masumoto Y, Mizukoshi Y, Takita R, Yokozawa T, et al. Catalyst-dependent intrinsic ring-walking behavior on π-face of conjugated polymers. Polym Chem. 2017;8:1708–13.
Leone AK, Goldberg PK, McNeil AJ. Ring-walking in catalyst-transfer polymerization. J Am Chem Soc. 2018;140:7846–50.
Bryan ZJ, McNeil AJ. Conjugated polymer synthesis via catalyst-transfer polycondensation (CTP): Mechanism, scope, and applications. Macromolecules. 2013;46:8395–405.
Cheng S, Zhao R, Seferos DS. Precision synthesis of conjugated polymers using the Kumada methodology. Acc Chem Res. 2021;54:4203–14.
Tamao K, Sumitani K, Kumada M. Selective carbon-carbon bond formation by cross-coupling of grignard reagents with organic halides. catalysis by nickel-phosphine complexes. J Am Chem Soc. 1972;94:4374–6.
Miyakoshi R, Yokoyama A, Yokozawa T. Catalyst-transfer polycondensation. Mechanism of Ni-catalyzed chain-growth polymerization leading to well-defined poly(3-hexylthiophene). J Am Chem Soc. 2005;127:17542–7.
Senkovskyy V, Khanduyeva N, Komber H, Oertel U, Stamm M, Kuckling D, et al. Conductive polymer brushes of regioregular head-to-tail poly(3-alkylthiophenes) via catalyst-transfer surface-initiated polycondensation. J Am Chem Soc. 2007;129:6626–32.
Khanduyeva N, Senkovskyy V, Beryozkina T, Bocharova V, Simon F, Nitschke M, et al. Grafting of poly(3-hexylthiophene) from poly(4-bromostyrene) films by Kumada catalyst-transfer polycondensation: Revealing of the composite films structure. Macromolecules. 2008;41:7383–9.
Doubina N, Ho A, Jen AK-Y, Luscombe CK. Effect of initiators on the Kumada catalysttransfer polycondensation reaction. Macromolecules. 2009;42:7670–7.
Smeets A, Van Den Bergh K, De Winter J, Gerbaux P, Verbiest T, Koeckelberghs G. Incorporation of different end groups in conjugated polymers using functional nickel initiators. Macromolecules. 2009;42:7638–41.
Doubina N, Stoddard M, Bronstein HA, Jen AK-Y, Luscombe CK. The effects of binding ligand variation on the nickel catalyzed externally initiated polymerization of 2-bromo-3-hexyl-5- iodothiophene. Macromol Chem Phys. 2009;210:1966–72.
Bronstein HA, Luscombe CK. Externally initiated regioregular P3HT with controlled molecular weight and narrow polydispersity. J Am Chem Soc. 2009;131:12894–5.
Kempf CN, Smith KA, Pesek SL, Li X, Verduzco R. Amphiphilic poly(alkylthiophene) block copolymers prepared via externally initiated GRIM and click coupling. Polym Chem. 2013;4:2158–63.
Doubina N, Paniagua SA, Soldatova AV, Jen AK-Y, Marder SR, Luscombe CK. Steric effects of the initiator substituent position on the externally initiated polymerization of 2-bromo-5-iodo-3-hexylthiophene. Macromolecules. 2011;44:512–20.
Kaul E, Senkovskyy V, Tkachov R, Bocharova V, Komber H, Stamm M, et al. Synthesis of a bifunctional initiator for controlled Kumada catalyst-transfer polycondensation/nitroxide-mediated polymerization and preparation of poly(3-hexylthiophene)-polystyrene block copolymer therefrom. Macromolecules. 2010;43:77–81.
Senkovskyy V, Tkachov R, Beryozkina T, Komber H, Oertel U, Horecha M, et al. Hairy’ poly(3-hexylthiophene) particles prepared via surface-initiated Kumada catalyst-transfer polycondensation. J Am Chem Soc. 2009;131:16445–53.
Tkachov R, Senkovskyy V, Komber H, Sommer J-U, Kiriy A. Random catalyst walking along polymerized poly(3-hexylthiophene) chains in Kumada catalyst-transfer polycondensation. J Am Chem Soc. 2010;132:7803–10.
Chavez CA, Choi J, Nesterov EE. One-step simple preparation of catalytic initiators for catalyst-transfer Kumada polymerization: synthesis of defect-free polythiophenes. Macromolecules. 2014;47:506–16.
Johnson JA, Baskin JM, Bertozzi CR, Koberstein JT, Turro NJ. Copper-free click chemistry for the in situ crosslinking of photodegradable star polymers. Chem Commun. 2008; 3064-6.
Yuan M, Okamoto K, Bronstein HA, Luscombe CK. Constructing regioregular star poly(3-hexylthiophene) via externally initiated Kumada catalyst-transfer polycondensation. ACS Macro Lett. 2012;1:392–5.
Smeets A, Willot P, De Winter J, Gerbaux P, Verbiest T, Koeckelberghs G. End group-functionalization and synthesis of block-copolythiophenes by modified nickel initiators. Macromolecules. 2011;44:6017–25.
Monnaie F, Brullot W, Verbiest T, De Winter J, Gerbaux P, Smeets A, et al. Synthesis of end-group functionalized P3HT: General protocol for P3HT/nanoparticle hybrids. Macromolecules. 2013;46:8500–8.
Monnaie F, Verheyen L, De Winter J, Gerbaux P, Brullot W, Verbiest T, et al. Influence of structure of end-group-functionalized poly(3-hexylthiophene) and poly(3-octylselenophene) anchored on au nanoparticles. Macromolecules. 2015;48:8752–9.
Hou W, Zhao NJ, Meng D, Tang J, Zeng Y, Wu Y, et al. Controlled growth of well-defined conjugated polymers from the surfaces of multiwalled carbon nanotubes: photoresponse enhancement via charge separation. ACS Nano. 2016;10:5189–98.
Islam MA, Purkait TK, Mobarok MH, Hoehlein IMD, Sinelnikov R, Iqbal M, et al. Grafting poly(3-hexylthiophene) from silicon nanocrystal surfaces: synthesis and properties of a functional hybrid material with direct interfacial contact. Angew Chem. 2016;128:7519–23.
Leysen P, Teyssandier J, De Feyter S, Koeckelberghs G. Controlled synthesis of a helical conjugated polythiophene. Macromolecules. 2018;51:3504–14.
Delabie J, De Winter J, Deschaume O, Bartic C, Gerbaux P, Verbiest T, et al. Development of a layered hybrid nanocomposite material using α,ω-bifunctionalized polythiophenes. Macromolecules. 2020;53:11098–105.
Okamoto K, Luscombe CK. Simple procedure for mono- and bis-end-functionalization of regioregular poly(3-hexylthiophene)s using chalcogens. Chem Commun. 2014;50:5310–2.
Verbiest T, Sioncke S, Koeckelberghs G, Samyn C, Persoons A, Botek E, et al. Nonlinear optical properties of spincoated films of chiral polythiophenes. Chem Phys Lett. 2005;404:112–5.
Inagaki S, Higashihara T. Synthesis of an ABC triblock copolymer by a bilateral Click reaction using α,ω-bifunctionalized poly(3-hexylthiophene) as an inner segment. Polym Chem. 2022;13:3613–8.
Lotocki V, Grignon E, Mills HA, Ye S, Lough AJ, Seferos DSA. SNAr-active external initiator that enables heterobifunctional clickable polythiophenes. Macromol Chem Phys. 2024;225:2300347.
Pollit AA, Obhi NK, Lough AJ, Seferos DS. Evaluation of an external initiating Ni(II) dIImine catalyst for electron-deficient π-conjugated polymers. Polym Chem. 2017;8:4108–13.
Mao Y, Wang Y, Lucht BL. Regiocontrolled synthesis of poly(3-alkylthiophene)s by Grignard metathesis. J Polym Sci A Polym Chem. 2004;42:5538–47.
Huddleston NE, Sontag SK, Bilbrey JA, Sheppard GR, Locklin J. Palladium-mediated surface-initiated Kumada catalyst polycondensation: A facile route towards oriented conjugated polymers. Macromol Rapid Commun. 2012;33:2115–20.
Bryan ZJ, Smith ML, McNeil AJ. Chain-growth polymerization of aryl grignards initiated by a stabilized NHC-Pd precatalyst. Macromol Rapid Commun. 2012;33:842–7.
Buenaflor J, Sommerville P, Qian H, Luscombe C. Investigation of bimetallic nickel catalysts in catalyst-transfer polymerization of π-conjugated polymers. Macromol Chem Phys. 2020;221:1900363.
King AJ, Wang J, Liu T, Raghavan A, Tomson NC, Zhukhovitskiy AV. Influence of metal identity and complex nuclearity in Kumada cross-coupling polymerizations with a pyridine diimine-based ligand scaffold. ACS Polym Au. 2023;3:475–481.
King AJ, Zhukhovitskiy AV. A chain-growth mechanism for conjugated polymer synthesis facilitated by dinuclear complexes with redox-active ligands. Angew Chem Int Ed. 2022;61:e202206044.
Pollit AA, Lough AJ, Seferos DS. Examining the spin state and redox chemistry of ni(diimine) catalysts during the synthesis of π-conjugated polymers. Macromol Chem Phys. 2020;221:2000321.
Horne WS, Grossmann TN. Proteomimetics as protein-inspired scaffolds with defined tertiary folding patterns. Nat Chem. 2020;12:331–7.
Ter Huurne GM, Palmans ARA, Meijer EW. Supramolecular single-chain polymeric nanoparticles. CCS Chem. 2019;1:64–82.
Hannigan MD, Sampson JA, Damaraju L, Weck M. Homochiral helical poly(thiophene)s accessed via living catalyst-transfer polymerization. Angew Chem Int Ed. 2025;64:e202502104.
Yokoyama A, Suzuki H, Kubota Y, Ohuchi K, Higashimura H, Yokozawa T. Chain-growth polymerization for the synthesis of polyfluorene via Suzuki Miyaura coupling reaction from an externally added initiator unit. J Am Chem Soc. 2007;129:7236–7.
Lee JK, Ko S, Bao Z. In situ hetero end-functionalized polythiophene and subsequent ‘click’ chemistry with DNA. Macromol Rapid Commun. 2012;33:938–42.
Elmalem E, Biedermann F, Johnson K, Friend RH, Huck WTS. Synthesis and photophysics of fully π-conjugated heterobis- functionalized polymeric molecular wires via Suzuki chain-growth polymerization. J Am Chem Soc. 2012;134:17769–77.
Fischer CS, Baier MC, Mecking S. Enhanced brightness emission-tuned nanoparticles from heterodifunctional polyfluorene building blocks. J Am Chem Soc. 2013;135:1148–54.
Zhang HH, Hu QS, Hong K. Accessing conjugated polymers with precisely controlled heterobisfunctional chain ends via post-polymerization modification of the OTf group and controlled Pd(0)/t-Bu3P-catalyzed Suzuki cross-coupling polymerization. Chem Commun. 2015;51:14869–72.
Chen P, Yang G, Liu T, Li T, Wang M, Huang W. Optimization of opto-electronic property and device efficiency of polyfluorenes by tuning structure and morphology. Polym Int. 2006;55:473–90.
Kobayashi S, Fujiwara K, Jiang D-H, Yamamoto T, Tajima K, Yamamoto Y, et al. Suzuki-Miyaura catalyst-transfer polycondensation of triolborate-type fluorene monomer: Toward rapid access to polyfluorene-containing block and graft copolymers from various macroinitiators. Polym Chem. 2020;11:6832–9.
Jiang D-H, Ree BJ, Isono T, Xia X-C, Hsu L-C, Kobayashi S, et al. Facile one-pot synthesis of rod-coil bio-block copolymers and uncovering their role in forming the efficient stretchable touch-responsive light emitting diodes. Chem Eng J. 2021;418:129421.
Seo K-B, Lee I-H, Lee J, Choi I, Choi T-L. A rational design of highly controlled Suzuki-Miyaura catalyst-transfer polycondensation for precision synthesis of polythiophenes and their block copolymers: marriage of palladacycle precatalysts with MIDA-boronates. J Am Chem Soc. 2018;140:4335–43.
Javier AE, Patel SN, Hallinan DT, Srinivasan V, Balsara NP. Simultaneous electronic and ionic conduction in a block copolymer: application in lithium battery electrodes. Angew Chem Int Ed. 2011;50:9848–51.
De Cuendias A, Hiorns RC, Cloutet E, Vignau L, Cramail H. Conjugated rod-coil block copolymers and optoelectronic applications. Polym Int. 2010;59:1452–76.
Sary BN, Richard F, Brochon C, Leclerc N, Lévêque P, Audinot J-N, et al. A new supramolecular route for using rod-coil block copolymers in photovoltaic applications. Adv Mater. 2010;22:763–8.
Nguyen HQ, Bhatt MP, Rainbolt EA, Stefan MC. Synthesis and characterization of a polyisoprene-b-polystyrene-b-poly(3- hexylthiophene) triblock copolymer. Polym Chem. 2013;4:462–5.
Higashihara T, Ito S, Fukuta S, Miyane S, Ochiai Y, Ishizone T. Synthesis and characterization of multicomponent ABC- and ABCD-type miktoarm star-branched polymers containing a poly(3-hexylthiophene) segment. ACS Macro Lett. 2016;5:631–5.
Choi H-N, Yang H-S, Chae J-H, Choi T-L, Lee I-H. Synthesis of conjugated rod-coil block copolymers by RuPhos Pd-Catalyzed Suzuki-Miyaura catalyst-transfer polycondensation: initiation from coil-type polymers. Macromolecules. 2020;53:5497–503.
Choi H-N, Yang H-S, Park S, Choi T-L, Lee I-H. Combining two powerful living polymerizations using rationally designed bifunctional initiators for versatile synthesis of conjugated rod-coil block copolymers. Macromolecules. 2024;57:8050–8.
Choi H-N, Lee I-H. Protection-free one-pot synthesis of alcohol end-functionalized poly(3-hexylthiophene). Polym J. 2021;53:1205–11.
Lee J, Kim H, Park H, Kim T, Hwang SH, Seo D, et al. Universal Suzuki-Miyaura catalyst-transfer polymerization for precision synthesis of strong donor/acceptor-based conjugated polymers and their sequence engineering. J Am Chem Soc. 2021;143:11180–90.
Bautista MV, Varni AJ, Ayuso-Carrillo J, Tsai C-H, Noonan KJT. Chain-growth polymerization of benzotriazole using Suzuki-Miyaura cross-coupling and dialkylbiarylphosphine palladium catalysts. ACS Macro Lett. 2020;9:1357–62.
Kim H, Lee J, Kim T, Cho M, Choi T-L. Precision synthesis of various low-bandgap donor–acceptor alternating conjugated polymers via living Suzuki–Miyaura catalyst-transfer polymerization. Angew Chem Int Ed. 2022;61:e202205828.
Kleine A, Schubert US, Jager M. Exploiting α-/ω-reactivities during polymerization for controlled heterotelechelic poly(carbazole)s. Macromolecules. 2022;55:3688–98.
Chen Z, Narita A, Müllen K. Graphene nanoribbons: on-surface synthesis and integration into electronic devices. Adv Mater. 2020;32:2001893.
Lee J, Ryu H, Park S, Cho M, Choi T-L. Living Suzuki-Miyaura catalyst-transfer polymerization for precision synthesis of length-controlled armchair graphene nanoribbons and their block copolymers. J Am Chem Soc. 2023;145:15488–95.
Yano Y, Mitoma N, Ito H, Itami K. A quest for structurally uniform graphene nanoribbons: synthesis, properties, and applications. J Org Chem. 2020;85:4–33.
Luo H, Yu G. Preparation, bandgap engineering, and performance control of graphene nanoribbons. Chem Mater. 2022;34:3588–615.
Yin J, Jacobse PH, Pyle D, Wang Z, Crommie MF, Dong G. Programmable fabrication of monodisperse graphene nanoribbons via deterministic iterative synthesis. J Am Chem Soc. 2022;144:16012–9.
von Kugelgen S, Piskun I, Griffin JH, Eckdahl CT, Jarenwattananon NN, Fischer FR. Templated synthesis of end-functionalized graphene nanoribbons through living ring-opening alkyne metathesis polymerization. J Am Chem Soc. 2019;141:11050–8.
Takahashi A, Lin C-J, Ohshimizu K, Higashihara T, Chen W-C, Ueda M. Synthesis and characterization of novel polythiophenes with graphene-like structures via intramolecular oxidative coupling. Polym Chem. 2012;3:479–85.
Pun SH, Delgado A, Dadich C, Cronin A, Fischer FR. Controlled catalyst-transfer polymerization in graphene nanoribbon synthesis. Chem. 2024;10:675–85.
Zhang J-J, Liu K, Xiao Y, Yu X, Huang L, Gao H-J, et al. Precision graphene nanoribbon heterojunctions by chain-growth polymerization. Angew Chem Int Ed. 2023;62:e202310880.
Cai J, Pignedoli CA, Talirz L, Ruffieux P, Söde H, Liang L, et al. Graphene nanoribbon heterojunctions. Nat Nanotechnol. 2014;9:896–900.
Nguyen GD, Tsai H-Z, Omrani AA, Marangoni T, Wu M, Rizzo DJ, et al. Atomically precise graphene nanoribbon heterojunctions from a single molecular precursor. Nat Nanotechnol. 2017;12:1077–82.
Rizzo DJ, Veber G, Cao T, Bronner C, Chen T, Zhao F, et al. Topological band engineering of graphene nanoribbons. Nature. 2018;560:204–8.
Gröning O, Wang S, Yao X, Pignedoli CA, Borin Barin G, Daniels C, et al. Engineering of robust topological quantum phases in graphene nanoribbons. Nature. 2018;560:209–13.
Carsten B, He F, Son HJ, Xu T, Yu L. Stille polycondensation for synthesis of functional materials. Chem Rev. 2011;111:1493–528.
Zheng T, Schneider AM, Yu L. Stille polycondensation: a versatile synthetic approach to functional polymers; Wiley-VCH Verlag GmbH & Co. KgaA, 2017.
Bao Z, Chan W, Yu L. Synthesis of conjugated polymer by the Stille coupling reaction. Chem Mater. 1993;5:2–3.
Bao Z, Chan WK, Yu L. Exploration of the Stille coupling reaction for the syntheses of functional polymers. J Am Chem Soc. 1995;117:12426–35.
Kang S, Ono RJ, Bielawski CW. Controlled catalyst transfer polycondensation and surface-initiated polymerization of a p-phenyleneethynylene-based monomer. J Am Chem Soc. 2013;135:4984–7.
Qiu Y, Mohin J, Tsai C-H, Tristram-Nagle S, Gil RR, Kowalewski T, et al. Stille catalyst-transfer polycondensation using Pd-PEPPSI-IPr for high-molecular-weight regioregular poly(3-hexylthiophene). Macromol Rapid Commun. 2015;36:840–4.
Goto E, Ando S, Ueda M, Higashihara T. Nonstoichiometric Stille coupling polycondensation for synthesizing naphthalene-diimide-based π-conjugated polymers. ACS Macro Lett. 2015;4:1004–7.
Terayama K, Goto E, Higashihara T. Nonstoichiometric Stille coupling polycondensation via an intramolecular Pd(0) catalyst transfer using excess phthalimide monomer. Macromol Chem Phys. 2018;219:1800175.
Terayama K, Liu CW, Higashihara T. Catalyst-transfer system in stoichiometry-independent AA+BB-type Migita–Kosugi–Stille coupling polycondensation using ester-functionalized dibromo monomer. Polym J. 2022;54:143–50.
Yokawa K, Takamura M, Higashihara T. Direct synthesis of π-conjugated polymers with bromoaryl groups in side chains via a chemoselective intramolecular catalyst transfer system. Polym Chem. 2024;15:1591–7.
Higashihara T, Goto E, Ueda M. Purification-free and protection-free synthesis of regioregular poly(3-hexylthiophene) and poly(3-(6-hydroxyhexyl)thiophene) using a zincate complex of tBu4ZnLi2. ACS Macro Lett. 2012;1:167–70.
Goto E, Nakamura S, Kawauchi S, Mori H, Ueda M, Higashihara T. Precision synthesis of regioregular poly(3-hexylthiophene) with low dispersity using a zincate complex catalyzed by nickel with the ligand of 1,2-bis(dicyclohexylphosphino)ethane. J Polym Sci A Polym Chem. 2014;52:2287–96.
Higashihara T, Goto E. Controlled synthesis of low-polydisperse regioregular poly(3-hexylthiophene) and related materials by zincate-complex metathesis polymerization. Polym J. 2014;46:381–90.
Goto E, Mori H, Ueda M, Higashihara T. Controlled polymerization of electron-deficient naphthalene-diimide containing monomer by Negishi-type catalyst-transfer polymerization. J Photopolym Sci Technol. 2015;28:279–83.
Goto E, Higashihara T. Investigation of stoichiometry in reactants for atom-economical synthesis of regioregular poly(3-hexylthiophene) with low dispersity using zincate complex of tBu4ZnLi2. Microsyst Technol. 2016;22:39–44.
Goto E, Ochiai Y, Lo C-T, Koganezawa T, Ueda M, Higashihara T. Synthesis of regioblock copolythiophene by Negishi catalyst-Transfer polycondensation using tBu2Zn.2LiCl. Polym Chem. 2017;8:6143–9.
Ochiai Y, Goto E, Higashihara T. Controlled Synthesis of Poly(p-phenylene) Using a Zincate Complex, tBu4ZnLi2. Macromol Rapid Commun. 2017;38:1700155.
Inagaki S, Yamamoto T, Higashihara T. Direct synthesis of chain-end-functionalized poly(3-hexylthiophene) without protecting groups using a zincate complex. Macromol Rapid Commun. 2020;41:2000148.
Uchiyama M, Furuyama T, Kobayashi M, Matsumoto Y, Tanaka K. Toward a protecting-group-free halogen-metal exchange reaction: Practical, chemoselective metalation of functionalized aromatic halides using dianion-type zincate,tBu4ZnLi2. J Am Chem Soc. 2006;128:8404–5.
Tkachov R, Senkovskyy V, Beryozkina T, Boyko K, Bakulev V, Lederer A, et al. Palladium-catalyzed chain-growth polycondensation of AB-type monomers: High catalyst turnover and polymerization rates. Angew Chem Int Ed. 2014;53:2402–7.
Erdmann T, Back J, Tkachov R, Ruff A, Voit B, Ludwigs S, et al. Dithienosilole-based all-conjugated block copolymers synthesized by a combination of quasi-living Kumada and Negishi catalyst-transfer polycondensations. Polym Chem. 2014;5:5383–90.
Kubo T, Young MS, Souther KD, Hannigan MD, McNeil AJ. Air-tolerant poly(3-hexylthiophene) synthesis via catalyst-transfer polymerization. J Polym Sci. 2021;59:268–73.
Fuji K, Tamba S, Shono K, Sugie A, Mori A. Murahashi coupling polymerization: Nickel(II)-N-HETEROCYCLIC CARBENE complex-catalyzed polycondensation of organolithium species of (hetero)arenes. J Am Chem Soc. 2013;135:12208–11.
Yamamura M, Moritani I, Murahashi S-I. The reaction of σ-vinylpalladium complexes with alkyllithiums. stereospecific syntheses of olefins from vinyl halides and alkyllithiums. J Organomet Chem. 1975;91:C39–C42.
Schnürch M, Spina M, Khan AF, Mihovilovic MD, Stanetty P. Halogen dance reactions—a review. Chem Soc Rev. 2007;36:1046–57.
McKeown GR, Ye S, Cheng S, Seferos DS. Homogenous synthesis of monodisperse high oligomers of 3-hexylthiophene by temperature cycling. J Am Chem Soc. 2019;141:17053–6.
Xu H, Ye S, Zhao R, Seferos DS. Homogeneous synthesis of monodisperse sequence-defined conjugated oligomers by temperature cycling. Angew Chem Int Ed. 2022;61:e202210340.
Xu H, Mills HA, Ye S, Seferos DS. Recyclable terthiophenes for synthesizing precision conjugated oligomers. Polym Chem. 2024;15:3814–22.
Amna B, Ateş A, Ozturk T. Pd/Cu-catalyzed Sonogashira cross-coupling polycondensation: a promising approach for synthesizing conjugated polymers with useful applications. Eur Polym J. 2023;196:112275.
Su M, Liu N, Wang Q, Wang H, Yin J, Wu Z-Q. Facile synthesis of poly(phenyleneethynylene)-block-polyisocyanide copolymers via two mechanistically distinct, sequential living polymerizations using a single catalyst. Macromolecules. 2016;49:110–9.
Lee JA, Luscombe CK. Dual-catalytic Ag-Pd system for direct arylation polymerization to synthesize poly(3-hexylthiophene). ACS Macro Lett. 2018;7:767–71.
Xing L, Luscombe CK. Advances in applying C-H functionalization and naturally sourced building blocks in organic semiconductor synthesis. J Mater Chem C Mater. 2021;9:16391–409.
Ye S, Lotocki V, Xu H, Seferos DS. Group 16 conjugated polymers based on furan, thiophene, selenophene, and tellurophene. Chem Soc Rev. 2022;51:6442–74.
Takimoto L, Goto T, Chen J, Kuwabara J, Kanbara T. Nonstoichiometric direct arylation polymerization of octafluorobiphenyl with 2,7-diiodofluorene for regulating C-H terminals of π-conjugated polymer. Macromol Rapid Commun. 2024;45:2300225.
Huang S-L, Ye C-C, Pan Y-N, Liu Q-Y, Wang C, Xu L. Living direct arylation polymerization via C-H activation for the precision synthesis of polythiophenes and their block copolymers. Macromolecules. 2025;58:2357–65.
Yokawa K, Higashihara T. Nonstoichiometric direct arylation polymerization for synthesis of naphthalenediimide-based π-conjugated polymer. Macromol Rapid Commun. 2025;46:2500254.
Acknowledgements
Financial support from the Okinawa Institute of Science and Technology Graduate University and JSPS KAKENHI Grant Number JP24K08518 is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sanskriti, I., Luscombe, C.K. Evolution of catalyst transfer polymerization toward complex conjugated polymer architectures. Polym J (2026). https://doi.org/10.1038/s41428-026-01146-9
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41428-026-01146-9