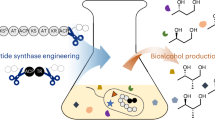
Abstract
Thioester reductase domain in nonribosomal peptide synthetases catalyzes reductive offloading to produce aldehyde- or alcohol-containing peptides. By genome mining focusing on thioester reductase domains, we isolated aquimarinols A-D, four novel polyketide-peptide hybrid metabolites isolated from the sponge-derived strain Aquimarina muelleri LMG 22569. Structural elucidation of aquimarinols revealed a β-formamidated fatty acid (FA) moiety and a threoninol unit linked by a peptide bond or an ester bond. The biosynthesis of aquimarinols is proposed to proceed via a hybrid trans-AT polyketide synthase-nonribosomal peptide synthetase pathway coupled with modification enzymes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hertweck C. The biosynthetic logic of polyketide diversity. Angew Chem Int Ed. 2009;48:4688–716.
Fischbach MA, Walsh CT. Assembly-Line Enzymology for Polyketide and Nonribosomal Peptide Antibiotics: Logic, Machinery, and Mechanisms. Chem Rev. 2006;106:3468–96.
Sussmuth RD, Mainz A. Nonribosomal Peptide Synthesis-Principles and Prospects. Angew Chem Int Ed. 2017;56:3770–821.
Bauman KD, Butler KS, Moore BS, Chekan JR. Genome mining methods to discover bioactive natural products. Nat Prod Rep. 2021;38:2100–29.
Xu M, Wang W, Waglechner N, Culp EJ, Guitor AK, Wright GD. Phylogeny-Informed Synthetic Biology Reveals Unprecedented Structural Novelty in Type V Glycopeptide Antibiotics. ACS Cent Sci. 2022;8:615–26.
Hashimoto T, Hashimoto J, Kozone I, Amagai K, Kawahara T, Takahashi S, et al. Biosynthesis of Quinolidomicin, the Largest Known Macrolide of Terrestrial Origin: Identification and Heterologous Expression of a Biosynthetic Gene Cluster over 200 kb. Org Lett. 2018;20:7996–9.
Little RF, Hertweck C. Chain release mechanisms in polyketide and non-ribosomal peptide biosynthesis. Nat Prod Rep. 2022;39:163–205.
Mullowney MW, McClure RA, Robey MT, Kelleher NL, Thomson RJ. Natural products from thioester reductase containing biosynthetic pathways. Nat Prod Rep. 2018;35:847–78.
Schneider BA, Balskus EP. Discovery of small molecule protease inhibitors by investigating a widespread human gut bacterial biosynthetic pathway. Tetrahedron. 2018;74:3215–30.
Yao S, Xie S, Liu R-Z, Huang Z, Zhang L. Expanding catalytic versatility of modular polyketide synthases for alcohol biosynthesis. Nat Chem Biol. 2025;21:1368–75.
Dan Q, Chiu Y, Lee N, Pereira JH, Rad B, Zhao X, et al. A polyketide-based biosynthetic platform for diols, amino alcohols and hydroxy acids. Nat Catal. 2025;8:147–61.
Zhang Z, Zhou Y, Xie S, Liu R-Z, Huang Z, Kumar PS, et al. NRPStransformer, an Accurate Adenylation Domain Specificity Prediction Algorithm for Genome Mining of Nonribosomal Peptides. J Am Chem Soc. 2025;147:32662–70.
Wang M, Carver JJ, Phelan VV, Sanchez LM, Garg N, Peng Y, et al. Sharing and community curation of mass spectrometry data with Global Natural Products Social Molecular Networking. Nat Biotechnol. 2016;34:828–37.
Flack H. On enantiomorph-polarity estimation. Acta Crystallogr Sect A. 1983;39:876–81.
Harris NC, Sato M, Herman NA, Twigg F, Cai W, Liu J, et al. Biosynthesis of isonitrile lipopeptides by conserved nonribosomal peptide synthetase gene cluster in Actinobacteria. Proc Natl Acad Sci USA. 2017;114:7025–30.
He H-Y, Tang M-C, Zhang F, Tang G-L. Cis-Double Bond Formation by Thioesterase and Transfer by Ketosynthase in FR901464 Biosynthesis. J Am Chem Soc. 2014;136:4488–91.
Ittiamornkul K, Zhu Q, Gkotsi DS, Smith DRM, Hillwig ML, Nightingale N, et al. Promiscuous indolyl vinyl isonitrile synthases in the biogenesis and diversification of hapaindole-type alkaloids. Chem Sci. 2015;6:6836–40.
Jia K, Sun H, Zhou Y, Zhang W. Biosynthesis of isonitrile lipopeptides. Curr Opin Chem Biol. 2024;81:102470.
Acknowledgements
This work was supported by the Zhejiang Key Laboratory Construction Project and the National Natural Science Foundation of China General Program (22177092) to L.Z.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Z., Zhang, L. Genome mining discovery of aquimarinols, threoninol-containing acylpeptides from Aquimarina muelleri. J Antibiot 79, 221–226 (2026). https://doi.org/10.1038/s41429-026-00894-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41429-026-00894-3