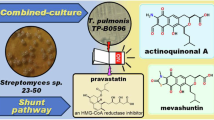
Abstract
A series of novel sulfur-containing cyclic lipopeptides named thioamycolamides F–I (1–4), with thiazoline, sulfoxide/sulfide, and fatty acid moieties, was discovered by applying GNPS molecular networking technology against combined-culture broth of Amycolatopsis sp. 26-4 and Tsukamurella pulmonis TP-B0596. Planar structural elucidation was accomplished by HRMS and 1D/2D NMR spectroscopic data analyses. The absolute configuration was determined by the highly sensitive advanced Marfey’s method developed by our group, electronic circular dichroism (ECD) spectroscopy, chemical synthesis, and Gaussian calculation. Moreover, compound 2 exhibited cytotoxicity against human cervix adenocarcinoma HeLa S3 cells with an IC50 of 24.0 μM.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Genilloud O. Actinomycetes: still a source of novel antibiotics. Nat Prod Rep. 2017;34:1203–32.
Rutledge PJ, Challis GL. Discovery of microbial natural products by activation of silent biosynthetic gene clusters. Nat Rev Microbiol. 2015;13:509–23.
Mao D, Okada BK, Wu Y, Xu F, Seyedsayamdost MR. Recent advances in activating silent biosynthetic gene clusters in bacteria. Curr Opin Microbiol. 2018;45:156–63.
Onaka H, Mori Y, Igarashi Y, Furumai T. Mycolic acid-containing bacteria induce natural-product biosynthesis in Streptomyces species. Appl Environ Microbiol. 2011;77:400–6.
Onaka H. Unlocking hidden bioactive compounds: from indolocarbazole and RiPP biosynthesis to the activation of cryptic secondary metabolism via microbial interactions. J Antibiot. 2025;78:395–407.
Kakeya H. Natural products-prompted chemical biology: phenotypic screening and a new platform for target identification. Nat Prod Rep. 2016;33:648–54.
Yanagisawa K, Kaneko K, Ikeda H, Iwata S, Muranaka A, Koshino H, et al. A new pyranonaphthoquinone, actinoquinonal A, and its congeners from the combined-culture of Streptomyces sp. 23–50 and Tsukamurella pulmonis TP-B0596. J Antibiot. 2025;78:350–8.
Sugiyama R, Nishimura S, Ozaki T, Asamizu S, Onaka H, Kakeya H. 5-Alkyl-1, 2, 3, 4-tetrahydroquinolines, new membrane-interacting lipophilic metabolites produced by combined culture of Streptomyces nigrescens and Tsukamurella pulmonis. Org Lett. 2015;17:1918–21.
Sugiyama R, Nishimura S, Ozaki T, Asamizu S, Onaka H, Kakeya H. Discovery and total synthesis of streptoaminals: antimicrobial [5, 5]-spirohemiaminals from the combined-culture of Streptomyces nigrescens and Tsukamurella pulmonis. Angew Chem. 2016;128:10434–8.
Pan C, Ikeda H, Minote M, Tokuda T, Kuranaga T, Taniguchi T, et al. Amoxetamide A, a new anoikis inducer, produced by combined-culture of Amycolatopsis sp. and Tsukamurella pulmonis. J Antibiot. 2024;77:66–70.
Pan C, Kuranaga T, Cao X, Suzuki T, Dohmae N, Shinzato N, et al. Amycolapeptins A and B, cyclic nonadepsipeptides produced by combined-culture of Amycolatopsis sp. and Tsukamurella pulmonis. J Org Chem. 2021;86:1843–9.
Wang M, et al. Sharing and community curation of mass spectrometry data with Global Natural Products Social Molecular Networking. Nat Biotechnol. 2016;34:828–37.
Nothias LF, et al. Feature-based molecular networking in the GNPS analysis environment. Nat Methods. 2020;17:905–8.
Pan C, Kuranaga T, Liu C, Lu S, Shinzato N, Kakeya H. Thioamycolamides A–E, sulfur-containing cycliclipopeptides produced by the rare Actinomycete Amycolatopsis sp. Org Lett. 2020;22:3014–7.
Kuranaga T, Minote M, Morimoto R, Pan C, Ogawa H, Kakeya H. Highly sensitive labeling reagents for scarce natural products. ACS Chem Biol. 2020;15:2499–506.
Wipf P, Fritch PC. Total synthesis and assignment of configuration of lissoclinamide 7. J Am Chem Soc. 1996;118:12358–67.
Fernandez I, Khiar N. Recent developments in the synthesis and utilization of chiral sulfoxides. Chem Rev. 2003;103:3651–706.
Pitchen P, Kagan HB. An efficient asymmetric oxidation of sulfides to sulfoxides. Tetrahedron Lett. 1984;25:1049–52.
Pitchen P, Dunach E, Deshmukh MN, Kagan HB. An efficient asymmetric oxidation of sulfides to sulfoxides. J Am Chem Soc. 1984;106:8188–93.
Frisch, M. J. et al. Gaussian 16. Gaussian, Inc.: 2016.
Acknowledgements
This work was supported in part by the National Natural Science Foundation of China (grant number 82404465), the Senior Talent Foundation of Jiangsu University (grant number 5501290012), the Jiangsu Provincial Double-Innovation Doctor Program (grant number JSSCBS0623), the Chugai Foundation for Innovative Drug Discovery Science: C-FINDs (grant number 2025-CF-01 to HK), a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan (17H06401, 23H04882, and 24H00493, all awarded to HK), the Project for Promotion of Cancer Research and Therapeutic Evolution (24ama221540 and 25ama221540 to HK), and the Platform Project for Supporting Drug Discovery and Life Science Research (24ama121034 and 25ama121034 to HK) from the Japan Agency for Medical Research and Development (AMED), Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, C., Zhang, L., Kuranaga, T. et al. Combination of combined-culture of Amycolatopsis sp. with Tsukamurella pulmonis and GNPS molecular networking reveals novel sulfur-containing cyclic lipopeptides thioamycolamides F–I. J Antibiot 79, 293–300 (2026). https://doi.org/10.1038/s41429-026-00909-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41429-026-00909-z