Abstract
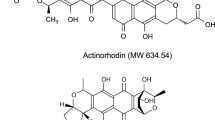
Benzoisochromanequinone (BIQ) polyketides including actinorhodin, medermycin, and granaticin, are biosynthesized via the intermediate 4-dihydro-9-hydroxy-1-methyl-10-oxo-3-H-naphtho[2,3-c]pyran-3-acetic acid (DNPA), which serves as a critical branch point. The configuration at C-3 of DNPA is a key determinant of structural diversity, leading to either (S)- or (R)-DNPA. Actinorhodin and medermycin are generated via (S)-DNPA, whereas granaticin is produced via (R)-DNPA. In contrast to the ketoreductase ActVI–ORF1 involved in (S)-DNPA production, the enzymatic function of Gra-6, presumed to be associated with (R)-DNPA biosynthesis, remains uncharacterized. This study evaluated the DNPA-producing ability of Gra-6 using a recombinant enzyme system expressed in Escherichia coli, revealing that it catalyzes stereoselective (R)-DNPA production. Furthermore, the putative amino acid residues involved in the DNPA-producing ability of Gra-6 were predicted through molecular modeling, including homology modeling and docking simulations with the coenzyme and an acyl carrier protein (ACP)-free bicyclic intermediate (BI) as the substrate. The involvement of these residues was verified by employing Gra-6 mutants. Additionally, the stereoselective mechanisms underlying DNPA production by each enzyme were proposed by comparing the docking models of ActVI–ORF1 and Gra-6 with the coenzyme and ACP-free BI. These findings establish Gra-6 as a pivotal stereospecific branch-point enzyme in BIQ polyketide biosynthetic branching and provide new insights into the stereoselective catalytic mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ōmura S, Ikeda H, Malpartida F, Kieser HM, Hopwood DA. Production of new hybrid antibiotics, mederrhodins A and B, by a genetically engineered strain. Antimicrob Agents Chemother. 1986;29:13–19.
He H, Ding WD, Bernan VS, Richardson AD, Ireland CM, et al. Lomaiviticins A and B, potent antitumor antibiotics from Micromonospora lomaivitiensis. J Am Chem Soc. 2001;123:5362–3.
Nishiyama T, Hashimoto Y, Kusakabe H, Kumano T, Kobayashi M. Natural low-molecular mass organic compounds with oxidase activity as organocatalysts. Proc Natl Acad Sci USA. 2014;111:17152–7.
Mak S, Nodwell JR. Actinorhodin is a redox-active antibiotic with a complex mode of action against Gram-positive cells. Mol Microbiol. 2017;106:597–613.
Naruse N, Goto M, Watanabe Y, Terasawa T, Dobashi K. K1115 A, a new anthraquinone that inhibits the binding of activator Protein-1 (AP-1) to its recognition Sites II. Taxonomy, fermentation, isolation, physico-chemical properties and structure determination. J Antibiot. 1998;51:545–52.
Hashimoto M, Watari S, Taguchi T, Ishikawa K, Kumamoto T, et al. Actinorhodin biosynthesis terminates with an unprecedented biaryl coupling reaction. Angew Chem Int Ed. 2023;62:e202214400.
Zhu HH, Guo J, Yao Q, Yang SZ, Deng MR, et al. Streptomyces vietnamensis sp. nov., a streptomycete with violet blue diffusible pigment isolated from soil in Vietnam. Int J Syst Evol Microbiol. 2007;57:1770–4.
Ichinose K, Bedford DJ, Tornus D, Bechthold A, Bibb MJ, et al. The granaticin biosynthetic gene cluster of Streptomyces violaceoruber Tu22: sequence analysis and expression in a heterologous host. Chem Biol. 1998;5:647–59.
Elson AL, Box SJ, Gilpin ML. New quinone antibiotics of the granaticin type, isolated from Streptomyces lateritius. I. Production, isolation and properties. J Antibiot. 1988;41:570–2.
Gilpin ML, Box SJ, Elson AL. New quinone antibiotics of the granaticin type, isolated from Streptomyces lateritius. II. Structure determination. J Antibiot. 1988;41:512–8.
Jiang B, Li S, Zhao W, Li T, Zuo L, et al. 6-Deoxy-13-hydroxy-8,11-dione-dihydrogranaticin B, an intermediate in granaticin biosynthesis, from Streptomyces sp. CPCC 200532. J Nat Prod. 2014;77:2130–3.
Roy S, Banerjee D. Broad spectrum antibacterial activity of granaticinic acid, isolated from Streptomyces thermoviolaceus NT1; an endophyte in Catharanthus roseus (L.) G. Don. J Appl Pharm Sci. 2015;5:006–11.
Lv Q, Fan Y, Tao G, Fu P, Zhai J, et al. Sekgranaticin, a SEK34b-Granaticin hybrid polyketide from Streptomyces sp. 166#. J Org Chem. 2019;84:9087–92.
Brimble MA, Duncalf LJ, Nairn MR. Pyranonaphthoquinone antibiotics—isolation, structure and biological activity. Nat Prod Rep. 1999;16:267–81.
Nishiyama T, Enomoto N, Nagayasu R, Ueda K. Organocatalytic activity of granaticin and its involvement in bactericidal function. Sci Rep. 2022;12:1–11.
Cole SP, Rudd BAM, Chang CJ, Hopwood DA, Floss HG. Biosynthesis of the antibiotic actinorhodin. Analysis of blocked mutants of Streptomyces coelicolor. J Antibiot. 1987;40:340–7.
Bartel PL, Zhu CB, Lampel JS, Dosch DC, Connors NC, et al. Biosynthesis of anthraquinones by interspecies cloning of actinorhodin biosynthesis genes in streptomycetes: clarification of actinorhodin gene functions. J Bacteriol. 1990;172:4816–26.
Staunton J, Weissman KJ. Polyketide biosynthesis: a millennium review. Nat Prod Rep. 2001;18:380–416.
Ichinose K, Ozawa M, Itou K, Kunieda K, Ebizuka Y. Cloning, sequencing and heterologous expression of the medermycin biosynthetic gene cluster of Streptomyces sp. AM-7161: towards comparative analysis of the benzoisochromanequinone gene clusters. Microbiology. 2003;149:1633–45.
Risdian C, Mozef T, Wink J. Biosynthesis of polyketides in Streptomyces. Microorganisms. 2019;7:124.
Bentley SD, Chater KF, Cerdeño-Tárraga AM, Challis GL, Thomson NR, et al. Complete genome sequence of the model actinomycete Streptomyces coelicolor A3(2). Nature. 2002;417:141–7.
Taguchi T, Awakawa T, Nishihara Y, Kawamura M, Ohnishi Y, et al. Bifunctionality of ActIV as a cyclase-thioesterase revealed by in vitro reconstitution of actinorhodin biosynthesis in Streptomyces coelicolor A3(2). ChemBioChem. 2017;18:316–23.
Hashimoto M, Taguchi T, Ishikawa K, Mori R, Hotta A, et al. Unveiling two consecutive hydroxylations: mechanisms of aromatic hydroxylations catalyzed by flavin-dependent monooxygenases for the biosynthesis of actinorhodin and related antibiotics. ChemBioChem. 2020;21:623–7.
Ishikawa K, Hashimoto M, Komatsu K, Taguchi T, Okamoto S, et al. Characterization of stereospecific enoyl reductase ActVI-ORF2 for pyran ring formation in the actinorhodin biosynthesis of Streptomyces coelicolor A3(2). Bioorg Med Chem Lett. 2022;66:128727.
Ichinose K, Taguchi T, Bedford DJ, Ebizuka Y, Hopwood DA. Functional complementation of Pyran ring formation in actinorhodin biosynthesis in Streptomyces coelicolor A3(2) by ketoreductase genes for granaticin biosynthesis. J Bacteriol. 2001;183:3247–50.
Taguchi T, Itou K, Ebizuka Y, Malpartida F, Hopwood DA, et al. Chemical characterisation of disruptants of the Streptomyces coelicolor A3(2) actVI genes involved in actinorhodin biosynthesis. J Antibiot. 2000;53:144–52.
Taguchi T, Kunieda K, Takeda-Shitaka M, Takaya D, Kawano N, et al. Remarkably different structures and reaction mechanisms of ketoreductases for the opposite stereochemical control in the biosynthesis of BIQ antibiotics. Bioorg Med Chem. 2004;12:5917–27.
Taguchi T, Ebizuka Y, Hopwood DA, Ichinose K. A new mode of stereochemical control revealed by analysis of the biosynthesis of dihydrogranaticin in Streptomyces violaceoruber Tü22. J Am Chem Soc. 2001;123:11376–80.
Javidpour P, Pereira JH, Goh EB, McAndrew RP, Ma SM, et al. Biochemical and structural studies of NADH-dependent FabG used to increase the bacterial production of fatty acids under anaerobic conditions. Appl Environ Microbiol. 2014;80:497–505.
Nakajima K, Yamashita A, Akama H, Nakatsu T, Kato H, et al. Crystal structures of two tropinone reductases: different reaction stereospecificities in the same protein fold. Proc Natl Acad Sci USA. 1998;95:4876–81.
Ziegler J, Facchini PJ, Geißler R, Schmidt J, Ammer C, et al. Evolution of morphine biosynthesis in opium poppy. Phytochemistry. 2009;70:1696–707.
Serapian SA, Crosby J, Crump MP, Van Der Kamp MW. Path to actinorhodin: regio- and stereoselective ketone reduction by a type II polyketide ketoreductase revealed in atomistic detail. JACS Au. 2022;2:972–84.
Narcross L, Fossati E, Bourgeois L, Dueber JE, Martin VJJ. Microbial factories for the production of benzylisoquinoline alkaloids. Trends Biotechnol. 2016;34:228–41.
Cao L, Teo D, Wang Y, Ye Q, Liu C, et al. Advancements in microbial cell engineering for benzylisoquinoline alkaloid production. ACS Synth Biol. 2024;13:3842–56.
Mugo AN, Kobayashi J, Mikami B, Yoshikane Y, Yagi T, et al. Crystal structure of 5-formyl-3-hydroxy-2-methylpyridine 4-carboxylic acid 5-dehydrogenase, an NAD+-dependent dismutase from Mesorhizobium loti. Biochem Biophys Res Commun. 2015;456:35–40.
Acknowledgements
This work was supported by JSPS KAKENHI grants No. JP23K06200 (to K. Ishikawa) and JP 24K09883 (to K. Ichinose).
Author information
Authors and Affiliations
Contributions
K. Ishikawa, Conceptualization, Data curation, Funding acquisition, Investigation, Methodology, Writing – original draft, Writing – review & editing. M. Hashimoto, N. Kusuoku and C. Nozaki, investigation. M. Hashimoto and K. Ichinose, Funding acquisition and Writing – review & editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ishikawa, K., Hashimoto, M., Kusuoku, N. et al. Functional characterization of Gra-6 as a branch-point enzyme directing (R)-DNPA formation in BIQ polyketide biosynthesis. J Antibiot (2026). https://doi.org/10.1038/s41429-026-00912-4
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41429-026-00912-4