Abstract
Introduction
A large number of genes involved in autosomal recessive forms of intellectual disability (ID) were identified over the past few years through whole-exome sequencing (WES) or whole-genome sequencing in consanguineous families. Disease-associated variants in TRAPPC9 were reported in eight multiplex consanguineous sibships from different ethnic backgrounds, and led to the delineation of the phenotype. Affected patients have microcephaly, obesity, normal motor development, severe ID, and language impairment and brain anomalies.
Patients
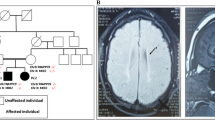
We report six new patients recruited through a national collaborative network.
Results
In the two patients heterozygous for a copy-number variation (CNV), the phenotype was clinically relevant with regard to the literature, which prompted to sequence the second allele, leading to identification of disease-associated variants in both. The third patient was homozygote for an intragenic TRAPPC9 CNV. The phenotype of the patients reported was concordant with the literature. Recent reports emphasized the role of CNVs in the etiology of rare recessive disorders.
Conclusion
This study demonstrates that CNVs significantly contribute to the mutational spectrum of TRAPPC9 gene, and also confirms the interest of combining WES with CNV analysis to provide a molecular diagnosis to patients with rare Mendelian disorders.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Mochida GH, Mahajnah M, Hill AD, et al. A truncating mutation of TRAPPC9 is associated with autosomal-recessive intellectual disability and postnatal microcephaly. Am J Hum Genet. 2009;85:897–902.
Philippe O, Rio M, Carioux A, et al. Combination of linkage mapping and microarray-expression analysis identifies NF-κB signaling defect as a cause of autosomal-recessive mental retardation. Am J Hum Genet. 2009;85:903–8.
Mir A, Kaufman L, Noor A, et al. Identification of mutations in TRAPPC9, which encodes the NIK- and IKK-β-binding protein, in nonsyndromic autosomal-recessive mental retardation. Am J Hum Genet. 2009;85:909–15.
Najmabadi H, Motazacker MM, Garshasbi M, et al. Homozygosity mapping in consanguineous families reveals extreme heterogeneity of non-syndromic autosomal recessive mental retardation and identifies 8 novel gene loci. Hum Genet. 2007;121:43–8.
Abou Jamra R, Wohlfart S, Zweier M, et al. Homozygosity mapping in 64 Syrian consanguineous families with non-specific intellectual disability reveals 11 novel loci and high heterogeneity. Eur J Hum Genet. 2011;19:1161–6.
Kakar N, Goebel I, Daud S, et al. A homozygous splice site mutation in TRAPPC9 causes intellectual disability and microcephaly. Eur J Med Genet. 2012;55:727–31.
Marangi G, Leuzzi V, Manti F, et al. TRAPPC9-related autosomal recessive intellectual disability: report of a new mutation and clinical phenotype. Eur J Hum Genet. 2013;21:229–32.
Giorgio E, Ciolfi A, Biamino E, et al. Whole exome sequencing is necessary to clarify ID/DD cases with de novo copy number variants of uncertain significance: two proof-of-concept examples. Am J Med Genet A. 2016;170:1772–9.
Koifman A, Feigenbaum A, Bi W, et al. A homozygous deletion of 8q24.3 including the NIBP gene associated with severe developmental delay, dysgenesis of the corpus callosum, and dysmorphic facial features. Am J Med Genet A. 2010;152A:1268–72.
Boone PM, Campbell IM, Baggett BC, et al. Deletions of recessive disease genes: CNV contribution to carrier states and disease-causing alleles. Genome Res. 2013;23:1383–94.
Dad S, Rendtorff ND, Kann E, et al. Partial USH2A deletions contribute to Usher syndrome in Denmark. Eur J Hum Genet. 2015;23:1646–51.
Lindstrand A, Frangakis S, Carvalho CMB, et al. Copy-number variation contributes to the mutational load of Bardet-Biedl syndrome. Am J Hum Genet. 2016;99:318–36.
Rivera-Brugués N, Albrecht B, Wieczorek D, et al. Cohen syndrome diagnosis using whole genome arrays. J Med Genet. 2011;48:136–40.
Miller DT, Adam MP, Aradhya S, et al. Consensus statement: chromosomal microarray is a first-tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet. 2010;86:749–64.
Amos JS, Huang L, Thevenon J, et al. Autosomal recessive mutations in THOC6 cause intellectual disability: syndrome delineation requiring forward and reverse phenotyping. Clin Genet. 2017;91:92–9.
Pfundt, R, Del Rosario M, Vissers LELM, et al. Detection of clinically relevant copy-number variants by exome sequencing in a large cohort of genetic disorders. Genet Med Off J Am Coll Med Genet. 2016. https://doi.org/10.1038/gim.2016.163.
Acknowledgements
We thank the families for their cooperation. We also thank all the lab technicians involved in this research. Affymetrix microarrays were processed in the Microarray Core Facility of the Institute in Regenerative Medicine and Biotherapy, CHRU-INSERM-UM Montpellier, http://irmb.chu-montpellier.fr.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing financial interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Mortreux, J., Busa, T., Germain, D.P. et al. The role of CNVs in the etiology of rare autosomal recessive disorders: the example of TRAPPC9-associated intellectual disability. Eur J Hum Genet 26, 143–148 (2018). https://doi.org/10.1038/s41431-017-0018-x
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41431-017-0018-x
This article is cited by
-
Expanding the genetic and phenotypic spectrum of TRAPPC9 and MID2-related neurodevelopmental disabilities: report of two novel mutations, 3D-modelling, and molecular docking studies
Journal of Human Genetics (2024)
-
A novel homozygous mutation in TRAPPC9 gene causing autosomal recessive non-syndromic intellectual disability
BMC Medical Genomics (2022)
-
Biallelic loss of TRAPPC9 function links vesicle trafficking pathway to autosomal recessive intellectual disability
Journal of Human Genetics (2022)
-
Whole exome sequencing revealed novel variants in consanguineous Pakistani families with intellectual disability
Genes & Genomics (2021)