Abstract
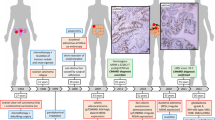
Constitutional mismatch repair deficiency (CMMRD) is an autosomal recessively inherited childhood cancer susceptibility syndrome caused by biallelic germline mutations in one of the mismatch repair (MMR) genes. The spectrum of CMMRD-associated tumours is very broad and many CMMRD patients additionally display signposting non-neoplastic features, most frequently café-au-lait macules and other pigmentation alterations. We report on a 13-month-old girl suspected of having CMMRD due to a desmoplastic medulloblastoma and a striking skin pigmentation that included multiple café-au-lait macules, hypopigmented areas and Mongolian spots. Whole-exome sequencing revealed homozygosity for MSH2 variant p.(Leu92Val) and MSH6 variant p.(Val809del), both variants of uncertain significance (VUS). Immunohistochemical analysis of the tumour tissue showed expression of all four MMR proteins and gMSI testing was negative. However, functional assays demonstrated that the cells of the patient displayed methylation tolerance and ex vivo microsatellite instability, which unequivocally confirmed the diagnosis of CMMRD. Taken together, the results render the MSH2 variant unlikely to be responsible for the phenotype, while they are compatible with MSH6-associated CMMRD. This case illustrates the diagnostic strategy of confirming CMMRD syndrome in patients with VUS.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Wimmer K, Kratz CP, Vasen HF, et al. Diagnostic criteria for constitutional mismatch repair deficiency syndrome: suggestions of the European consortium ‘care for CMMRD’ (C4CMMRD). J Med Genet. 2014;51:355–65.
Wimmer K, Rosenbaum T, Messiaen L. Connections between constitutional mismatch repair deficiency syndrome and neurofibromatosis type 1. Clin Genet. 2017;91:507–19.
Ingham D, Diggle CP, Berry I, et al. Simple detection of germline microsatellite instability for diagnosis of constitutional mismatch repair cancer syndrome. Hum Mutat. 2013;34:847–52.
Bodo S, Colas C, Buhard O, et al. Diagnosis of constitutional mismatch repair-deficiency syndrome based on microsatellite instability and lymphocyte tolerance to methylating agents. Gastroenterology. 2015;149:1017–29. e1013
Mork ME, Borras E, Taggart MW, et al. Identification of a novel PMS2 alteration c.505C>G (R169G) in trans with a PMS2 pathogenic mutation in a patient with constitutional mismatch repair deficiency. Fam Cancer. 2016;15:587–91.
Sjursen W, Bjornevoll I, Engebretsen LF, Fjelland K, Halvorsen T, Myrvold HE. A homozygote splice site PMS2 mutation as cause of Turcot syndrome gives rise to two different abnormal transcripts. Fam Cancer. 2009;8:179–86.
Bougeard G, Charbonnier F, Moerman A, et al. Early onset brain tumor and lymphoma in MSH2-deficient children. Am J Hum Genet. 2003;72:213–6.
Tabori U, Hansford JR, Achatz MI, et al. Clinical management and tumor surveillance recommendations of inherited mismatch repair deficiency in childhood. Clin Cancer Res. 2017;23:e32–e37.
Vasen HF, Ghorbanoghli Z, Bourdeaut F, et al. Guidelines for surveillance of individuals with constitutional mismatch repair-deficiency proposed by the European Consortium “Care for CMMR-D” (C4CMMR-D). J Med Genet. 2014;51:283–93.
Polubothu S, Scott RH, Vabres P, Kinsler VA, Atypical dermal melanocytosis: diagnostic clue in constitutional mismatch repair deficiency syndrome. Br J Dermatol. 2017;177:e185–6.
Scott RH, Mansour S, Pritchard-Jones K, Kumar D, MacSweeney F, Rahman N. Medulloblastoma, acute myelocytic leukemia and colonic carcinomas in a child with biallelic MSH6 mutations. Nat Clin Pract Oncol. 2007;4:130–4.
Carol Durno, C. Richard Boland, Shlomi Cohen, Jason A. Dominitz, Frank M. Giardiello, David A. Johnson, Tonya Kaltenbach, TR. Levin, David Lieberman, Douglas J. Robertson, Douglas K. Rex, Recommendations on Surveillance and Management of Biallelic Mismatch Repair Deficiency (BMMRD) Syndrome: A Consensus Statement by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology 2017:152 (6):1605–14.
Acknowledgements
This work was supported by the Elterninitiative Kinderkrebsklinik e.V. Duesseldorf. The authors wish to thank Dr. Claudia Potthoff and Dr. Stefan Balzer for their contribution in sample acquisition, Mrs. Silke Furlan and Mrs. Katayoun Alemazkour for technical assistance, Mrs. Fanny Bouchlis and Mrs. Brigitte Litra for functional assays and Dr. Joerg Schaper for providing the radiological image. We thank the Biological Resource Center, Hôpital Cochin, APHP-Paris France, for the lymphoid cell line establishment.
Author contributions
J.T. and M.K. performed WES data analysis and first drafted the manuscript. K.W. helped writing the manuscript. K.W., M.M., and C.F. critically revised the manuscript for important intellectual content. J.T. performed Sanger sequencing. K.W. and C.F. performed gMSI testing. M.M., O.L., and C.C. performed methylation tolerance assay and evMSI. T.B. obtained informed consent and asked for the family history. T.B., J.I.H., A.B., and M.K. cared for the child. J.F. and J.R. performed the pathological analysis. M.G. was responsible for library preparation, S.G. for the internal SQL database. A.B. and M.K. designed and supervised the project. All authors approved the final manuscript as submitted.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
The study was approved by the local ethics committee and written informed consent was obtained from the parents.
Additional information
Julia Taeubner and Katharina Wimmer authors contributed equally to this work.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Taeubner, J., Wimmer, K., Muleris, M. et al. Diagnostic challenges in a child with early onset desmoplastic medulloblastoma and homozygous variants in MSH2 and MSH6 . Eur J Hum Genet 26, 440–444 (2018). https://doi.org/10.1038/s41431-017-0071-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-017-0071-5
This article is cited by
-
Medulloblastoma associated with Lynch syndrome: a case report of germline MLH1 variant and tumor molecular characterization
Investigational New Drugs (2025)
-
Using comprehensive genomic and functional analyses for resolving genotype–phenotype mismatches in children with suspected CMMRD in Lebanon: an IRRDC study
Human Genetics (2023)
-
Comprehensive germline-genomic and clinical profiling in 160 unselected children and adolescents with cancer
European Journal of Human Genetics (2021)
-
Medulloblastoma
Nature Reviews Disease Primers (2019)