Abstract
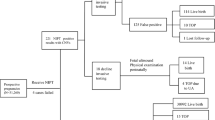
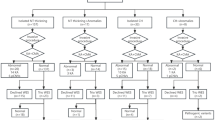
This study aimed to examine the choice pregnant women make about the amount of fetal genetic information they want from chromosome microarray. Women having invasive prenatal testing in the absence of fetal structural abnormality were recruited in Victoria, Australia. A decision aid for women described ‘targeted’ analysis as reporting only copy number variants implicated in a highly penetrant and well-described phenotype and ‘extended’ as additionally reporting variants of uncertain or unknown significance. Participant’s choice and demographics were collected by survey before chorionic villus sampling or amniocentesis; psychological data were also collected then and again about 10 days after receiving results. High-resolution single-nucleotide polymorphism array analysis was performed, and a clinical review committee assessed variants for reporting before returning results to participants. Sixty-six participants (59.5%) chose extended analysis and 45 (40.5%) targeted. Choosing extended information was associated with (1) indication for prenatal diagnosis: maternal age alone (adjusted odds ratio (adjOR) 9.6, 95% confidence interval (CI): 1.4–66.0, p= 0.02), or ‘other’ indication (adjOR 7.1, 95% CI: 1.5–33.1, p= 0.01)); (2) >12 months to conceive (adjOR 4.1, 95% CI: 1.0–17.7, p= 0.05); and (3) Asian background (adjOR 4.67, 95% CI: 1.0–21.0, p= 0.04). No adverse psychological impact occurred in either group. We conclude that offering pregnant women different levels of fetal genetic analysis is warranted, alongside decision support.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Le Caignec C, Boceno M, Saugier-Veber P, Jacquemont S, Joubert M, David A, et al. Detection of genomic imbalances by array based comparative genomic hybridisation in fetuses with multiple malformations. J Med Genet. 2005;42:121–8.
Wapner RJ, Martin CL, Levy B, Ballif BC, Eng CM, Zachary JM, et al. Chromosomal microarray versus karyotyping for prenatal diagnosis. N Engl J Med. 2012;367: 2175–84.
Srebniak MI, Joosten M, Knapen M, Arends LR, Polak M, van Veen S, et al. Frequency of submicroscopic chromosome aberrations in pregnancies without increased risk for structural chromosome aberrations: a systematic review of literature and meta-analysis. Ultrasound Obstet Gynecol. 2017. https://doi.org/10.1002/uog.17533
McGillivray G, Rosenfeld JA, McKinlay Gardner RJ, Gillam LH. Genetic counselling and ethical issues with chromosome microarray analysis in prenatal testing. Prenat Diagn. 2012;32:389–95.
Bernhardt BA, Soucier D, Hanson K, Savage M, Jackson LG, Wapner RJ. Women’s experiences receiving abnormal prenatal chromosomal microarray testing results. Genet Med. 2012;15:139–45.
Stark Z, Gillam L, Walker SP, McGillivray G. Ethical controversies in prenatal microarray. Curr Opin Obstet Gynecol. 2013;25:133–7.
Crolla JA, Wapner R, Van Lith JMM. Controversies in prenatal diagnosis 3: should everyone undergoing invasive testing have a microarray? Prenat Diagn. 2014;34:18–22.
American College of Obstetrics and Gynecologists. Committee on Genetics Society for Maternal–Fetal Medicine. Committee Opinion no 682: microarrays and next generation sequencing technology: the use of advanced genetic diagnostic tools in obstetrics and gynecology. Obstet Gynecol. 2016;128:e262–e268.
Vanakker O, Vilain C, Janssens K, Van der Aa N, Smits G, Bandelier C, et al. Implementation of genomic arrays in prenatal diagnosis: The Belgian approach to meet the challenges. Eur J Med Genet. 2014;57:151–6.
Gardiner C, Wellesley D, Kilby M, Kerr B. Recommendations for the use of chromosome microarray in pregnancy. London: The Royal College of Pathologists, PUB 290615; 2015.
Royal Australian and New Zealand College of Obstetricians and Gynaecologists. Prenatal screening and diagnosis of chromosomal and genetic conditions in the fetus in pregnancy. C-Obs 59. Royal Australian and New Zealand College of Obstetricians and Gynaecologists; 2016.
Ganesamoorthy D, Bruno DL, McGillivray G, Norris F, White S. M., Adroub S, et al. Meeting the challenge of interpreting high-resolution single nucleotide polymorphism array data in prenatal diagnosis: does increased diagnostic power outweigh the dilemma of rare variants? BJOG. 2013;120:594–606.
O’Connor AM. Validation of a decisional conflict scale. Med Decis Making. 1995;15:25–30.
Spielberger CD, Van der Aa N., Smits, G, Bandelier C, Manual for the State-Trait Anxiety Inventory (STAI). Palo Alto, CA: Consulting Psychologists Press; 1983.
Marteau TM, Bekker H. The development of a six-item short-form of the state scale of the Spielberger State-Trait Anxiety Inventory (STAI). Br J Clin Psychol. 1992;31:301–6.
Braithwaite D, Sutton S, Steggles N. Intention to participate in predictive genetic testing for hereditary cancer: the role of attitude toward uncertainty. Psychol Health. 2002;17:761–72.
Carleton RN, Norton MAPJ, Asmundson GJG. Fearing the unknown: a short version of the Intolerance of Uncertainty Scale. J Anxiety Disord. 2007;21:105–17.
Scheier MF, Carver CS, Bridges MW. Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): a reevaluation of the Life Orientation Test. J Pers Soc Psychol. 1994;67:1063–78.
Muller C, Cameron LD. Trait anxiety, information modality, and responses to communications about prenatal genetic testing. J Behav Med. 2014;37:988–99.
Smith BW, Dalen J, Wiggins K, Tooley E, Christopher P, Bernard J. The brief resilience scale: assessing the ability to bounce back. Int J Behav Med. 2008;15:194–200.
Brehaut JC, O’Connor AM, Wood TJ, Hack TF, Siminoff L, Gordon E, et al. Validation of a decision regret scale. Med Decis Making. 2003;23:281–92.
Shkedi-Rafid S, Fenwick A, Dheensa S, Wellesley D, Lucassen AM. What results to disclose, when, and who decides? Healthcare professionals’ views on prenatal chromosomal microarray analysis. Prenat Diagn. 2016;36:252–9.
van der Steen SL, Diderich KE, Riedijk SR, Verhagen-Visser J, Govaerts LC, Joosten M, et al. Pregnant couples at increased risk for common aneuploidies choose maximal information from invasive genetic testing. Clin Genet. 2015;88: 25–31.
Gil M, Revello R, Poon L, Akolekar R, Nicolaides K. Clinical implementation of routine screening for fetal trisomies in the UK NHS: cell-free DNA test contingent on results from first-trimester combined test. Ultra Obstet Gynecol. 2016;47:45–52.
Chitty L, Wright D, Hill M, Verhoef T, Daley R, Lewis C, et al. Uptake, outcomes, and costs of implementing non-invasive prenatal testing for Down’s syndrome into NHS maternity care: prospective cohort study in eight diverse maternity units. BMJ. 2016;354:i3426.
Wapner RJ, Babiarz JE, Levy B, Stosic M, Zimmermann B, Sigurjonsson S, et al. Expanding the scope of noninvasive prenatal testing: detection of fetal microdeletion syndromes. Am J Obstet Gynecol. 2015;212:332.e1–339.
Fiallos K, Applegate C, Mathews DJ, Bollinger J, Bergner AL, James CA. Choices for return of primary and secondary genomic research results of 790 members of families with Mendelian disease. Eur J Hum Genet. 2017;25:530–7.
Kalia SS, Adelman K, Bale SJ, Chung WK, Eng C, Evans JP, et al. Recommendations for reporting of secondary findings in clinical exome and genome sequencing, 2016 update (ACMG SFv2.0): a policy statement of the American College of Medical Genetics and Genomics. Genet Med. 2017;19:249–55.
Chan YM, Leung TN, Leung TY, Fung TY, Chan LW, Lau TK, The utility assessment of Chinese pregnant women towards the birth of a baby with Down syndrome compared to a procedure-related miscarriage. Prenat Diagn. 2006;26:819–24.
Calonico E, Blumenfeld YJ, Hudgins L, Taylor J. Patient preferences for prenatal testing of microdeletion and microduplication syndromes. Prenat Diagn. 2016;36:244–51.
Hui L, Hutchinson B, Poulton A, Halliday J, Population-based impact of noninvasive prenatal screening on screening and diagnostic testing for fetal aneuploidy. Genet Med. 2017;19:1338–45. doi:https://doi.org/10.1038/gim.2017.55.
Westerfield L, Darilek S, van den Veyver IB. Counselling challenges with variants of uncertain significance and incidental findings in prenatal genetic screening and diagnosis. J Clin Med. 2014;3:1018–32.
Scientific American Editors. Beware the destiny test. Sci Am Online. 2013;308:12.
Dolan SM. Personalized genomic medicine and prenatal genetic testing. JAMA. 2014;312:1203–5.
Kuppermann M, Pena S, Bishop JT, Nakagawa S, Gregorich SE, Sit A, et al. Effect of enhanced information, values clarification, and removal of financial barriers on use of prenatal genetic testing: a randomized clinical trial. JAMA. 2014;312:1210–7.
Beulen L, van den Berg M, Faas BH, Feenstra I, Hageman M, van Vugt JM, et al. The effect of a decision aid on informed decision-making in the era of non-invasive prenatal testing: a randomised controlled trial. Eur J Hum Genet. 2016;24:1409–16.
Walser SA, Kellom KS, Palmer SC, Bernhardt BA. Comparing genetic counselor’s and patient’s perceptions of needs in prenatal chromosomal microarray testing. Prenat Diagn. 2015;35:870–8.
Acknowledgements
We would like to acknowledge all the research genetic counsellors who recruited participants and contributed to the design and implementation of the research, particularly Susan Fawcett. All practice managers and medical staff at sites involved are also thanked. VCGS laboratory personnel involved in review and reporting of results included David Francis and the laboratory head at the time, Howard Slater, who facilitated the two-tier reporting approach.
Funding
Jane Halliday and Bettina Meiser were supported through National Health and Medical Research Council (NHMRC) Senior Fellowships, Level B, numbers 1021252 and 1078523 respectively. The research project was also funded by NHMRC (grant number 1059993). This work was made possible through the Victorian State Government Operational Infrastructure Support and Australian Government NHMRC IRIISS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Halliday, J.L., Muller, C., Charles, T. et al. Offering pregnant women different levels of genetic information from prenatal chromosome microarray: a prospective study. Eur J Hum Genet 26, 485–494 (2018). https://doi.org/10.1038/s41431-017-0084-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-017-0084-0
This article is cited by
-
Impact of a digital platform on genetic counselling encounters in the screening context
European Journal of Human Genetics (2026)
-
Exploring attitudes and experiences with reproductive genetic carrier screening among couples seeking medically assisted reproduction: a longitudinal survey study
Journal of Assisted Reproduction and Genetics (2024)
-
Development and use of the Australian reproductive genetic carrier screening decision aid
European Journal of Human Genetics (2022)
-
Study protocol: childhood outcomes of fetal genomic variants: the PrenatAL Microarray (PALM) cohort study
BMC Pediatrics (2021)