Abstract
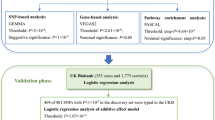
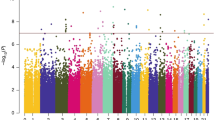
Development of high-throughput genotyping platforms provides an opportunity to identify new genetic elements related to complex cognitive functions. Taking advantage of multi-level genomic analysis, here we studied the genetic basis of human short-term (STM, n = 1623) and long-term (LTM, n = 1522) memory functions. Heritability estimation based on single nucleotide polymorphism showed moderate (61%, standard error 35%) heritability of short-term memory but almost zero heritability of long-term memory. We further performed a two-step genome-wide association study, but failed to find any SNPs that could pass genome-wide significance and survive replication at the same time. However, suggestive significance for rs7011450 was found in the shared component of the two STM tasks. Further inspections on its nearby gene zinc finger and at-hook domain containing and SNPs around this gene showed suggestive association with STM. In LTM, a polymorphism within branched chain amino acid transaminase 2 showed suggestive significance in the discovery cohort and has been replicated in another independent population of 1862. Furthermore, we performed a pathway analysis based on the current genomic data and found pathways including mTOR signaling and axon guidance significantly associated with STM capacity. These findings warrant further replication in other larger populations.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Skodzik T, Holling H, Pedersen A. Long-term memory performance in adult ADHD: a meta-analysis. J Atten Disord. 2017;21:267–83.
Wang Y, Zhang YB, Liu LL, et al. A meta-analysis of working memory impairments in autism spectrum disorders. Neuropsychol Rev. 2017;27:46–61.
Heck A, Fastenrath M, Ackermann S, et al. Converging genetic and functional brain imaging evidence links neuronal excitability to working memory, psychiatric disease, and brain activity. Neuron. 2014;81:1203–13.
Bettencourt K, Xu Y. Decoding the content of visual short-term memory under distraction in occipital and parietal areas. Nat Neurosci. 2016;19:150–7.
Muller NG, Knight RT. The functional neuroanatomy of working memory: contributions of human brain lesion studies. Neuroscience. 2006;139:51–58.
Tang Y, Shimizu E, Dube GR, et al. Genetic enhancement of learning and memory in mice. Nature. 1999;401:63–69.
Voss JL, Paller KA. An electrophysiological signature of unconscious recognition memory. Nat Neurosci. 2009;12:349–55.
McClearn GE, Johansson B, Berg S, et al. Substantial genetic influence on cognitive abilities in twins 80 or more years old. Science. 1997;276:1560–3.
Goldberg HX, Lemos GS, Fananas SL. A systematic review of the complex organization of human cognitive domains and their heritability. Psicothema. 2014;26:1–9.
Jensen AR, Marisi DQ. Note on the heritability of memory span. Behav Genet. 1979;9:379–87.
Volk HE, McDermott KB, Roediger HL III, Todd RD. Genetic influences on free and cued recall in long-term memory tasks. Twin Res Hum Genet. 2006;9:623–31.
Vogler C, Gschwind L, Coynel D, et al. Substantial SNP-based heritability estimates for working memory performance. Transl Psychiatry. 2014;4:e438.
Papassotiropoulos A, Stephan DA, Huentelman MJ, et al. Common KIBRA alleles are associated with human memory performance. Science. 2006;314:475–8.
Papassotiropoulos A, Henke K, Stefanova E, et al. A genome-wide survey of human short-term memory. Mol Psychiatry. 2011;16:184–92.
Papassotiropoulos A, Stefanova E, Vogler C, et al. A genome-wide survey and functional brain imaging study identify CTNNBL1 as a memory-related gene. Mol Psychiatry. 2013;18:255–63.
Price AL, Patterson N, Plenge RM, Weinblatt ME, Shadick NA, Reich D. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet. 2006;38:904–9.
Yang J, Lee SH, Goddard ME, Visscher PM. GCTA: a tool for genome-wide complex trait analysis. Am J Hum Genet. 2011;88:76–82.
Yang J, Benyamin B, McEvoy BP, et al. Common SNPs explain a large proportion of the heritability for human height. Nat Genet. 2010;42:565–U131.
Visscher PM, Hemani G, Vinkhuyzen AA, et al. Statistical power to detect genetic (co)variance of complex traits using SNP data in unrelated samples. PLoS Genet. 2014;10:e1004269.
Purcell S, Neale B, Todd-Brown K, et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559–75.
Barrett JC, Fry B, Maller J, Daly MJ. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics. 2005;21:263–5.
Delaneau O, Marchini J, Zagury J. A linear complexity phasing method for thousands of genomes. Nat Methods. 2012;9:179–81.
Howie B, Fuchsberger C, Stephens M, Marchini J, Abecasis GR. Fast and accurate genotype imputation in genome-wide association studies through pre-phasing. Nat Genet. 2012;44:955–9.
Howie BN, Donnelly P, Marchini J. A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet. 2009;5:e1000529.
Marchini J, Howie B, Myers S, Mcvean G, Donnelly P. A new multipoint method for genome-wide association studies via imputation of genotypes. Nat Genet. 2007;39:906–13.
Pruim RJ, Welch RP, Sanna S, et al. LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics. 2010;26:2336–7.
Liu JZ, McRae AF, Nyholt DR, et al. A versatile gene-based test for genome-wide association studies. Am J Hum Genet. 2010;87:139–45.
Segrè AV, DIAGRAM Consortium, MAGIC investigators. et al. Common inherited variation in mitochondrial genes is not enriched for associations with Type 2 diabetes or related glycemic traits. PLoS Genet. 2010;6:e1001058.
Fujimoto T, Doi K, Koyanagi M, et al. ZFAT is an antiapoptotic molecule and critical for cell survival in MOLT-4 cells. FEBS Lett. 2009;583:568–72.
Ishikura S, Tsunoda T, Nakabayashi K, et al. Molecular mechanisms of transcriptional regulation by the nuclear zinc-finger protein Zfat in T cells. Biochim Biophys Acta. 2016;1859:1398–410.
You L, Zou J, Zhao H, et al. Deficiency of the chromatin regulator BRPF1 causes abnormal brain development. J Biol Chem. 2015;290:7114–29.
Kushima I, Aleksic B, Ikeda M, et al. Association study of bromodomain-containing 1 gene with schizophrenia in Japanese population. Am J Med Genet B Neuropsychiatr Genet. 2010;153B:786–91.
Shirasawa S, Harada H, Furugaki K, et al. SNPs in the promoter of a B cell-specific antisense transcript, SAS-ZFAT, determine susceptibility to autoimmune thyroid disease. Hum Mol Genet. 2004;13:2221–31.
Bourguiba-Hachemi S, Ashkanani TK, Kadhem FJ, Almawi WY, Alroughani R, Fathallah MD. ZFAT gene variant association with multiple sclerosis in the Arabian Gulf population: A genetic basis for gender-associated susceptibility. Mol Med Rep. 2016;14:3543–50.
Hull J, Hindy ME, Kehoe PG, Chalmers K, Love S, Conway ME. Distribution of the branched chain aminotransferase proteins in the human brain and their role in glutamate regulation. J Neurochem. 2012;123:997–1009.
Arndt S, Poser I, Schubert T, Moser M, Bosserhoff AK. Cloning and functional characterization of a new Ski homolog, Fussel-18, specifically expressed in neuronal tissues. Lab Invest. 2005;85:1330–41.
Minaki Y, Nakatani T, Mizuhara E, Inoue T, Ono Y. Identification of a novel transcriptional corepressor, Corl2, as a cerebellar Purkinje cell-selective marker. Gene Expr Patterns. 2008;8:418–23.
Rietveld CA, Esko T, Davies G, et al. Common genetic variants associated with cognitive performance identified using the proxy-phenotype method. Proc Natl Acad Sci USA. 2014;111:13790–4.
Mu YG, Huang LJ, Li SY, et al. Working memory and the identification of facial expression in patients with left frontal glioma. Neuro Oncol. 2012;14:81–89.
Bekinschtein P, Katche C, Slipczuk LN, et al. mTOR signaling in the hippocampus is necessary for memory formation. Neurobiol Learn Mem. 2007;87:303–7.
Nakahara S, Miyake S, Tajinda K, Ito H. Mossy fiber mis-pathfinding and semaphorin reduction in the hippocampus of α-CaMKII hKO mice. Neurosci Lett. 2015;598:47–51.
Dines M, Grinberg S, Vassiliev M, Ram A, Tamir T, Lamprecht R. The roles of Eph receptors in contextual fear conditioning memory formation. Neurobiol Learn Mem. 2015;124:62–70.
Zhang X, Yu JT, Li J, et al. Bridging Integrator 1 (BIN1) genotype effects on working memory, hippocampal volume, and functional connectivity in young healthy individuals. Neuropsychopharmacology. 2015;40:1794–803.
Davies G, Marioni RE, Liewald DC, et al. Genome-wide association study of cognitive functions and educational attainment in UK Biobank (N = 112 151). Mol Psychiatry. 2016;21:758–67.
Xiang B, Wu JY, Ma XH, et al. Genome-wide association study with memory measures as a quantitative trait locus for schizophrenia. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 2012;29:255–9.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Project 31421003); the Beijing Innovation Center for Genomics at Peking University; the Applied Development Program from the Science and Technology Committee of Chongqing (cstc2014yykfB10003 and cstc2015shms-ztzx10006); and the Program of Mass Creativities Workshops from the Science and Technology Committee of Chongqing. We are grateful to Zhangyan Guan and Huizhen Yang for help with DNA preparation. Zijian Zhu thanks the Chinese Scholarship Council (CSC, No. 201709920075) and the German Academic Research Foundation (DAAD, No. 91658524) for financial support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
41431_2018_201_MOESM2_ESM.pdf (download PDF )
Supplementary Figure 1 Quantile-Quantile (Q-Q) plots of memory-related phenotypes. (a) Q-Q plot for STM measured in Digit Span task; (b) Q-Q plot for STM measured in Visuospatial memory task; (c) Q-Q plot for the shared component of STM measured in Digit Span task and in Visuospatial memory task; (d) Q-Q plot for LTM measurements. The red line represents null hypothesis. Blue dots represent results assuming an additive genetic model; orange dots represent results assuming a dominant/recessive genetic model. Plots were generated in R
Rights and permissions
About this article
Cite this article
Zhu, Z., Chen, B., Yan, H. et al. Multi-level genomic analyses suggest new genetic variants involved in human memory. Eur J Hum Genet 26, 1668–1678 (2018). https://doi.org/10.1038/s41431-018-0201-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-018-0201-8
This article is cited by
-
Beyond Single Variants: A Pathway-Based Approach to Explore the Genetic Basis of Memory
Molecular Neurobiology (2026)
-
Genome-wide association study of population-standardised cognitive performance phenotypes in a rural South African community
Communications Biology (2023)
-
Heritability of human visual contour integration—an integrated genomic study
European Journal of Human Genetics (2019)