Abstract
In oncology, the expanding use of multi-gene panels to explore familial cancer predisposition and tumor genome analysis has led to increased secondary findings discoveries (SFs) and has given rise to important medical, ethical, and legal issues. The American College of Medical Genetics and Genomics published a policy statement for managing SFs for a list of genes, including 25 cancer-related genes. Currently, there are few recommendations in Europe. From June 2016 to May 2017, the French Society of Predictive and Personalized Medicine (SFMPP) established a working group of 47 experts to elaborate guidelines for managing information given on the SFs for genes related to cancers. A subgroup of ethicists, lawyers, patients’ representatives, and psychologists provided ethical reflection, information guidelines, and materials (written consent form and video). A subgroup with medical expertise, including oncologists and clinical and molecular geneticists, provided independent evaluation and classification of 60 genes. The main criteria were the “actionability” of the genes (available screening or prevention strategies), the risk evaluation (severity, penetrance, and age of disease onset), and the level of evidence from published data. Genes were divided into three classes: for class 1 genes (n = 36), delivering the information on SFs was recommended; for class 2 genes (n = 5), delivering the information remained questionable because of insufficient data from the literature and/or level of evidence; and for class 3 genes (n = 19), delivering the information on SFs was not recommended. These guidelines for managing SFs for cancer-predisposing genes provide new insights for clinicians and laboratories to standardize clinical practices.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Weiner C. Anticipate and communicate: ethical management of incidental and secondary findings in the clinical, research, and direct-to-consumer contexts (December 2013 report of the Presidential Commission for the Study of Bioethical Issues). Am J Epidemiol. 2014;180:562–4.
Kalia SS, Adelman K, Bale SJ, Chung WK, Eng C, Evans JP, et al. Recommendations for reporting of secondary findings in clinical exome and genome sequencing, 2016 update (ACMG SFv2.0): a policy statement of the American College of Medical Genetics and Genomics. Genet Med. 2017;19:249–55.
Tung N, Lin NU, Kidd J, Allen BA, Singh N, Wenstrup RJ, et al. Frequency of germline mutations in 25 cancer susceptibility genes in a sequential series of patients with breast cancer. J Clin Oncol. 2016;34:1460–8.
O’Leary E, Iacoboni D, Holle J, Michalski ST, Esplin ED, Yang S, et al. Expanded gene panel use for women with breast cancer: identification and intervention beyond breast cancer risk. Ann Surg Oncol. 2017;24:3060–6.
Stafford JL, Dyson G, Levin NK, Chaudhry S, Rosati R, Kalpage H, et al. Reanalysis of BRCA1/2 negative high risk ovarian cancer patients reveals novel germline risk loci and insights into missing heritability. PLoS One. 2017;12:e0178450.
Aloraifi F, McDevitt T, Martiniano R, McGreevy J, McLaughlin R, Egan CM, et al. Detection of novel germline mutations for breast cancer in non-BRCA1/2 families. FEBS J. 2015;282:3424–37.
Gieldon L, Masjkur JR, Richter S, Darr R, Lahera M, Aust DE, et al. Next generation panel sequencing identifies NF1 germline mutations in three patients with pheochromocytoma but no clinical diagnosis of Neurofibromatosis Type 1. Eur J Endocrinol. 2017;178:K1–9.
Ring KL, Bruegl AS, Allen BA, Elkin EP, Singh N, Hartman AR, et al. Germline multi-gene hereditary cancer panel testing in an unselected endometrial cancer cohort. Mod Pathol. 2016;29:1381–9.
Amendola LM, Dorschner MO, Robertson PD, Salama JS, Hart R, Shirts BH, et al. Actionable exomic incidental findings in 6503 participants: challenges of variant classification. Genome Res. 2015;25:305–15.
Dorschner MO, Amendola LM, Turner EH, Robertson PD, Shirts BH, Gallego CJ, et al. Actionable, pathogenic incidental findings in 1,000 participants’ exomes. Am J Hum Genet. 2013;93:631–40.
Dewey FE, Murray MF, Overton JD, Habegger L, Leader JB, Fetterolf SN, et al. Distribution and clinical impact of functional variants in 50,726 whole-exome sequences from the DiscovEHR study. Science. 2016;354:aaf6814.
Green RC, Berg JS, Grody WW, Kalia SS, Korf BR, Martin CL, et al. ACMG recommendations for reporting of incidental findings in clinical exome and genome sequencing. Genet Med. 2013;15:565–74.
Directors ABo. ACMG policy statement: updated recommendations regarding analysis and reporting of secondary findings in clinical genome-scale sequencing. Genet Med. 2015;17:68–9.
van El CG, Cornel MC, Borry P, Hastings RJ, Fellmann F, Hodgson SV, et al. Whole-genome sequencing in health care: recommendations of the European Society of Human Genetics. Eur J Hum Genet. 2013;21:580–4.
Vears DF, Senecal K, Borry P. Reporting practices for unsolicited and secondary findings from next-generation sequencing technologies: perspectives of laboratory personnel. Hum Mutat. 2017;38:905–11.
Hehir-Kwa JY, Claustres M, Hastings RJ, van Ravenswaaij-Arts C, Christenhusz G, Genuardi M, et al. Towards a European consensus for reporting incidental findings during clinical NGS testing. Eur J Hum Genet. 2015;23:1601–6.
Claustres M, Kozich V, Dequeker E, Fowler B, Hehir-Kwa JY, Miller K, et al. Recommendations for reporting results of diagnostic genetic testing (biochemical, cytogenetic and molecular genetic). Eur J Hum Genet. 2014;22:160–70.
Yang P, Cornejo KM, Sadow PM, Cheng L, Wang M, Xiao Y, et al. Renal cell carcinoma in tuberous sclerosis complex. Am J Surg Pathol. 2014;38:895–909.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24.
Plon SE, Eccles DM, Easton D, Foulkes WD, Genuardi M, Greenblatt MS, et al. Sequence variant classification and reporting: recommendations for improving the interpretation of cancer susceptibility genetic test results. Hum Mutat. 2008;29:1282–91.
Cascon A, Comino-Mendez I, Curras-Freixes M, de Cubas AA, Contreras L, Richter S, et al. Whole-exome sequencing identifies MDH2 as a new familial paraganglioma gene. J Natl Cancer Inst. 2015;107.
Bausch B, Schiavi F, Ni Y, Welander J, Patocs A, Ngeow J, et al. Clinical characterization of the pheochromocytoma and paraganglioma susceptibility genes SDHA, TMEM127, MAX, and SDHAF2 for gene-informed prevention. JAMA Oncol. 2017;3:1204–12.
Bayley JP, Kunst HP, Cascon A, Sampietro ML, Gaal J, Korpershoek E, et al. SDHAF2 mutations in familial and sporadic paraganglioma and phaeochromocytoma. Lancet Oncol. 2010;11:366–72.
Touraine M. Décret no. 2013-527 relatif aux conditions de mise en œuvre de l’information de la parentèle dans le cadre d’un examen des caractéristiques génétiques à finalité médicale; in: santéMdasedl (ed), 20 juin 2013.
Abdel-Rahman MH, Rai K, Pilarski R, Davidorf FH, Cebulla CM. Germline BAP1 mutations misreported as somatic based on tumor-only testing. Fam Cancer. 2016;15:327–30.
Bijlsma RM, Bredenoord AL, Gadellaa-Hooijdonk CG, Lolkema MP, Sleijfer S, Voest EE, et al. Unsolicited findings of next-generation sequencing for tumor analysis within a Dutch consortium: clinical daily practice reconsidered. Eur J Hum Genet. 2016;24:1496–500.
Peplow M. The 100,000 Genomes Project. BMJ. 2016;353:i1757.
Samuel GN, Farsides B. Public trust and ‘ethics review’ as a commodity: the case of Genomics England Limited and the UK’s 100,000 genomes project. Med Health Care Philos. 2018;21:159–68.
Greely HT. Iceland’s plan for genomics research: facts and implications. Jurimetrics. 2000;40:153–91.
Gudbjartsson DF, Helgason H, Gudjonsson SA, Zink F, Oddson A, Gylfason A, et al. Large-scale whole-genome sequencing of the Icelandic population. Nat Genet. 2015;47:435–44.
Palsson G, Rabinow P. Iceland: the case of a national human genome project. Anthropol Today. 1999;15:14–8.
Sutrop M, Simm K. The Estonian healthcare system and the genetic database project: from limited resources to big hopes. Camb Q Healthc Ethics. 2004;13:254–62.
Metspalu A, Estonian Genome Project—before the take-off and take-off. Bioinformatics. 2002;18(Suppl 2):S152
Metspalu A,Kohler F,Laschinski G,Ganten D,Roots I, [The Estonian Genome Project in the context of European genome research]. Dtsch Med Wochenschr. 2004;129(Suppl 1):S25–8.
Levy Y. Genomic medicine 2025: France in the race for precision medicine. Lancet. 2016;388:2872.
Boomsma DI, Wijmenga C, Slagboom EP, Swertz MA, Karssen LC, Abdellaoui A, et al. The Genome of the Netherlands: design, and project goals. Eur J Hum Genet. 2014;22:221–7.
Bickeboller H. [The National Genome Research Network. Genome research in Germany]. Bundesgesundheitsblatt Gesundh Gesundh. 2007;50:168–73.
Rehm HL, Berg JS, Brooks LD, Bustamante CD, Evans JP, Landrum MJ, et al. ClinGen—the clinical genome resource. N Engl J Med. 2015;372:2235–42.
Strande NT, Riggs ER, Buchanan AH, Ceyhan-Birsoy O, DiStefano M, Dwight SS, et al. Evaluating the clinical validity of gene-disease associations: an evidence-based framework developed by the clinical genome resource. Am J Hum Genet. 2017;100:895–906.
Van Allen EM, Robinson D, Morrissey C, Pritchard C, Imamovic A, Carter S, et al. A comparative assessment of clinical whole exome and transcriptome profiling across sequencing centers: implications for precision cancer medicine. Oncotarget. 2016;7:52888–99.
Nirsimloo R, Gourley C. The safety and efficacy of olaparib therapy in patients with relapsed ovarian cancer. Expert Rev Anticancer Ther. 2016;16:597–603.
Tewari KS, Eskander RN, Monk BJ. Development of olaparib for BRCA-deficient recurrent epithelial ovarian cancer. Clin Cancer Res. 2015;21:3829–35.
Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357:409–13.
Gong J, Cho M, Sy M, Salgia R, Fakih M. Molecular profiling of metastatic colorectal tumors using next-generation sequencing: a single-institution experience. Oncotarget. 2017;8:42198–213.
Gong J, Wang C, Lee PP, Chu P, Fakih M. Response to PD-1 blockade in microsatellite stable metastatic colorectal cancer harboring a POLE mutation. J Natl Compr Cancer Netw. 2017;15:142–7.
Budin-Ljosne I, Teare HJ, Kaye J, Beck S, Bentzen HB, Caenazzo L, et al. Dynamic Consent: a potential solution to some of the challenges of modern biomedical research. BMC Med Ethics. 2017;18:4.
Directors Abo. Laboratory and clinical genomic data sharing is crucial to improving genetic health care: a position statement of the American College of Medical Genetics and Genomics. Genet Med. 2017;19:721–2.
Acknowledgements
We thank the patient associations BRCA France, Vaincre les Maladies Lysosomales, and APTEF Familial polyposis for their involvement. No industry or corporate sponsors contributed to the development of these recommendations. SFMPP is not funded by industrial or commercial companies but by grants from public. We thank public institutions such as the French National Cancer Institute (“Institut National du Cancer”, INCa) and, the French Foundation for Cancer Research (“Association pour la Recherche sur le Cancer”, ARC), and territorial community support (by the “Metropole of Montpellier” and the “Region Occitanie”). for donations supporting SFMPP annual meeting. Communication of the recommendations to the medical and scientific society took place in an independent framework of industrial actors, at the annual 4th SFMPP congress in Paris (June 2017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest with industry or firms commercializing genetic tests. No industry or corporate sponsors contributed to the development of these recommendations. SFMPP is not funded by industrial or commercial companies but by grants from public institutions and partially self-funded by SFMPP annual congresses. Dr. Pujol discloses attending Advisory Board Membership meetings for AstraZeneca, Pfizer, and Genomic Health.
Electronic supplementary material
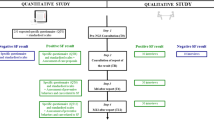
41431_2018_224_MOESM3_ESM.png (download PNG )
Flowchart representing the evaluation process and ranking of the initial cancer related gene list by the medical workgroup of the SFMPP
Rights and permissions
About this article
Cite this article
Pujol, P., Vande Perre, P., Faivre, L. et al. Guidelines for reporting secondary findings of genome sequencing in cancer genes: the SFMPP recommendations. Eur J Hum Genet 26, 1732–1742 (2018). https://doi.org/10.1038/s41431-018-0224-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-018-0224-1
This article is cited by
-
Cascade counselling and testing. Recommendations of the European Society of Human Genetics
European Journal of Human Genetics (2026)
-
Returning of individual genetic findings to biobank participants: a mixed methods study results in Lithuania
BMC Medical Ethics (2025)
-
Population-based genetic carrier screening. A consensus statement from the Spanish societies: AEGH, AEDP, ASEBIR, SEAGEN, SEF and SEGCD
European Journal of Human Genetics (2025)
-
Secondary findings in hereditary cancer genes after germline genetic testing – systematic review of literature
Human Genetics (2025)
-
Evaluation of a two-step model of opportunistic genomic screening
European Journal of Human Genetics (2024)