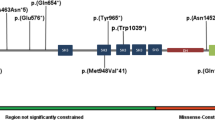
Abstract
Potocki–Shaffer syndrome (PSS) is a contiguous gene syndrome caused by 11p11.2 deletions. PSS is clinically characterized by intellectual disability, craniofacial anomalies, enlarged parietal foramina, and multiple exostoses. PSS occasionally shows autism spectrum disorder, epilepsy, and overgrowth. Some of the clinical features are thought to be associated with haploinsufficiency of two genes in the 11p11.2 region; variants affecting the function of ALX4 cause enlarged parietal foramina and EXT2 lead to multiple exostoses. However, the remaining clinical features were still yet to be linked to specific genetic alterations. In this study, we identified de novo truncating variants in an 11p11.2 gene, PHF21A, in three cases with intellectual disability and craniofacial anomalies. Among these three cases, autism spectrum disorder was recognized in one case, epilepsy in one case, and overgrowth in two cases. This study shows that PHF21A haploinsufficiency results in intellectual disability and craniofacial anomalies and possibly contributes to susceptibility to autism spectrum disorder, epilepsy, and overgrowth, all of which are PSS features.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Romeike BF, Wuyts W. Proximal chromosome 11p contiguous gene deletion syndrome phenotype: case report and review of the literature. Clin Neuropathol. 2007;26:1–11.
Wuyts W, Waeber G, Meinecke P, Schuler H, Goecke TO, Van Hul W, et al. Proximal 11p deletion syndrome (P11pDS): additional evaluation of the clinical and molecular aspects. Eur J Hum Genet. 2004;12:400–6.
Swarr DT, Bloom D, Lewis RA, Elenberg E, Friedman EM, Glotzbach C, et al. Potocki-Shaffer syndrome: comprehensive clinical assessment, review of the literature, and proposals for medical management. Am J Med Genet A. 2010;152a:565–72.
Labonne JD, Vogt J, Reali L, Kong IK, Layman LC, Kim HG. A microdeletion encompassing PHF21A in an individual with global developmental delay and craniofacial anomalies. Am J Med Genet A. 2015;167a:3011–8.
Montgomery ND, Turcott CM, Tepperberg JH, McDonald MT, Aylsworth AS. A 137-kb deletion within the Potocki-Shaffer syndrome interval on chromosome 11p11.2 associated with developmental delay and hypotonia. Am J Med Genet A. 2013;161a:198–202.
Wakui K, Gregato G, Ballif BC, Glotzbach CD, Bailey KA, Kuo PL, et al. Construction of a natural panel of 11p11.2 deletions and further delineation of the critical region involved in Potocki-Shaffer syndrome. Eur J Hum Genet. 2005;13:528–40.
Wu YQ, Badano JL, McCaskill C, Vogel H, Potocki L, Shaffer LG. Haploinsufficiency of ALX4 as a potential cause of parietal foramina in the 11p11.2 contiguous gene-deletion syndrome. Am J Hum Genet. 2000;67:1327–32.
Stickens D, Clines G, Burbee D, Ramos P, Thomas S, Hogue D, et al. The EXT2 multiple exostoses gene defines a family of putative tumour suppressor genes. Nat Genet. 1996;14:25–32.
Kim HG, Kim HT, Leach NT, Lan F, Ullmann R, Silahtaroglu A, et al. Translocations disrupting PHF21A in the Potocki-Shaffer-syndrome region are associated with intellectual disability and craniofacial anomalies. Am J Hum Genet. 2012;91:56–72.
Fujita A, Isidor B, Piloquet H, Corre P, Okamoto N, Nakashima M, et al. De novo MEIS2 mutation causes syndromic developmental delay with persistent gastro-esophageal reflux. J Hum Genet. 2016;61:835–8.
Bredrup C, Johansson S, Bindoff LA, Sztromwasser P, Krakenes J, Mellgren AE, et al. High myopia-excavated optic disc anomaly associated with a frameshift mutation in the MYC-binding protein 2 gene (MYCBP2). Am J Ophthalmol. 2015;159:973–9.e2.
Sohn YB, Yim SY, Cho EH, Kim OH. The first Korean patient with Potocki-Shaffer syndrome: a rare cause of multiple exostoses. J Korean Med Sci. 2015;30:214–7.
Romeike BF, Shen Y, Nishimoto HK, Morton CC, Layman LC, Kim HG. Spectrum of genes involved in a unique case of Potocki Schaffer syndrome with a large chromosome 11 deletion. Clin Neuropathol. 2014;33:238–44.
Ferrarini A, Gaillard M, Guerry F, Ramelli G, Heidi F, Keddache CV, et al. Potocki-Shaffer deletion encompassing ALX4 in a patient with frontonasal dysplasia phenotype. Am J Med Genet A. 2014;164a:346–52.
Palka C, Alfonsi M, Mohn A, Guanciali Franchi P, Chiarelli F, Calabrese G. Delayed diagnosis of Potocki-Shaffer syndrome in a woman with multiple exostoses and mental retardation. Mol Syndromol. 2012;2:259–61.
Chuang L, Wakui K, Sue WC, Su MH, Shaffer LG, Kuo PL. Interstitial deletion 11(p11.12p11.2) and analphoid marker formation results in inherited Potocki-Shaffer syndrome. Am J Med Genet A. 2005;133a:180–3.
McCool C, Spinks-Franklin A, Noroski LM, Potocki L. Potocki-Shaffer syndrome in a child without intellectual disability-The role of PHF21A in cognitive function. Am J Med Genet A. 2017;173:716–20.
Ansari M, Rainger J, Hanson IM, Williamson KA, Sharkey F, Harewood L, et al. Genetic analysis of ‘PAX6-Negative’ individuals with Aniridia or Gillespie Syndrome. PLoS ONE. 2016;11:e0153757.
Dollfus H, Joanny-Flinois O, Doco-Fenzy M, Veyre L, Joanny-Flinois L, Khoury M, et al. Gillespie syndrome phenotype with a t(X;11)(p22.32; p12) de novo translocation. Am J Ophthalmol. 1998;125:397–9.
Roberson MS, Ban M, Zhang T, Mulvaney JM. Role of the cyclic AMP response element binding complex and activation of mitogen-activated protein kinases in synergistic activation of the glycoprotein hormone alpha subunit gene by epidermal growth factor and forskolin. Mol Cell Biol. 2000;20:3331–44.
Acknowledgements
We thank all the participants for their cooperation in this research. We also thank Ms. K. Takabe, Mr. T. Miyama, Ms. N. Watanabe, Ms. M. Sato, Mr. S. Nakamura, and Ms. S. Sugimoto at the Department of Human Genetics, Yokohama City University Graduate School of Medicine, for their technical assistance. This work was supported by AMED under grant numbers, JP18ek0109280, JP18dm0107090, JP18ek0109301, JP18ek0109348, and JP18kk020500; JSPS KAKENHI Grant Numbers, JP17H01539, JP16H05160, JP16H05357, JP16H06254, JP15K10367, JP17K10080, JP 17K15630, and JP17H06994; the fund for Creation of Innovation Centers for Advanced Interdisciplinary Research Areas Program in the Project for Developing Innovation Systems (N Matsumoto) from the Japanese Science and Technology Agency; grants from the Ministry of Health, Labor and Welfare (N Matsumoto); the Takeda Science Foundation (N Miyake, HS, N. Matsumoto); and Kawano Masanori Memorial Public Interest Incorporated Foundation for Promotion of Pediatrics (S Miyatake). This study makes use of data generated by the DECIPHER community. A full list of centers who contributed to the generation of the data is available from http://decipher.sanger.ac.uk and via email from decipher@sanger.ac.uk.
Funding
Funding for the decipher project was provided by the Wellcome Trust. We thank Jeremy Allen, PhD, from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hamanaka, K., Sugawara, Y., Shimoji, T. et al. De novo truncating variants in PHF21A cause intellectual disability and craniofacial anomalies. Eur J Hum Genet 27, 378–383 (2019). https://doi.org/10.1038/s41431-018-0289-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-018-0289-x
This article is cited by
-
Molecular Consequences of a Missense PHF21A Variant, c.1285G > A, Associated With Syndromic Neurodevelopmental Disorder
Cellular and Molecular Neurobiology (2025)
-
11p11.12p12 duplication in a family with intellectual disability and craniofacial anomalies
BMC Medical Genomics (2021)
-
Disruption of PHF21A causes syndromic intellectual disability with craniofacial anomalies, epilepsy, hypotonia, and neurobehavioral problems including autism
Molecular Autism (2019)