Abstract
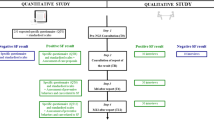
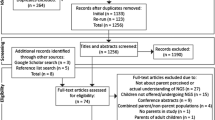
Exome sequencing (ES) has revolutionized diagnostic procedures in medical genetics, particularly for developmental diseases. The variety and complexity of the information produced has raised issues regarding its use in a clinical setting. Of particular interest are patients’ expectations regarding the information disclosed, the accompaniment provided, and the value patients place on these. To explore these issues in parents of children with developmental disorders and no diagnosis with known etiology, a multidisciplinary group of researchers from social and behavioral sciences and patient organizations conducted a mixed-methodology study (quantitative and qualitative) in two centers of expertise for rare diseases in France. The quantitative study aimed to determine the preferences of 513 parents regarding the disclosure of ES results. It showed that parents wished to have exhaustive information, including variants of unknown significance possibly linked to their child’s disorder and secondary findings. This desire for information could be a strategy to maximize the chances of obtaining a diagnosis. The qualitative study aimed to understand the expectations and reactions of 57 parents interviewed just after the return of ES results. In-depth analysis showed that parents had ambivalent feelings about the findings whatever the results returned. The contrasting results from these studies raise questions about the value of the information provided and parents’ high expectations regarding the results. The nature of parental expectations has emerged as an important topic in efforts to optimize accompaniment and support for families during the informed decision-making process and after disclosure of the results in an overall context of uncertainty.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Iglesias A, Anyane-Yeboa K, Wynn J, et al. The usefulness of whole-exome sequencing in routine clinical practice. Genet Med. 2014;16:922–31.
Willemsen MH, Kleefstra T. Genetic diagnostics in intellectual disability: what is the benefit? Ned Tijdschr Geneeskd. 2014;158:A8098.
Yang Y, Muzny DM, Reid JG, et al. Clinical whole-exome sequencing for the diagnosis of mendelian disorders. N Engl J Med. 2013;369:1502–11.
Gilissen C, Hehir-Kwa JY, Thung DT, et al. Genome sequencing identifies major causes of severe intellectual disability. Nature. 2014;511:344–7.
Carmichael N, Tsipis J, Windmueller G, Mandel L, Estrella E. “Is it going to hurt?”: the impact of the diagnostic odyssey on children and their families. J Genet Couns. 2015;24:325–35.
Berg JS, Khoury MJ, Evans JP. Deploying whole genome sequencing in clinical practice and public health: meeting the challenge one bin at a time. Genet Med. 2011;13:499–504.
Ormond KE, Wheeler MT, Hudgins L, et al. Challenges in the clinical application of whole-genome sequencing. Lancet. 2010;375:1749–51.
Pinxten W, Howard HC. Ethical issues raised by whole genome sequencing. Best Pract Res Clin Gastroenterol. 2014;28:269–79.
Borry P, Bentzen HB, Budin-Ljøsne I, et al. The challenges of the expanded availability of genomic information: an agenda-setting paper. J Community Genet. 2018;9:103–16.
Van ElCG, Cornel MC, Borry P, et al. Whole-genome sequencing in health care: recommendations of the European Society of Human Genetics. Eur J Hum Genet. 2013;21:580–4.
Matthijs G, Souche E, Alders M, et al. Guidelines for diagnostic next-generation sequencing. Eur J Hum Genet. 2016;24:2–5.
Clift KE, Halverson CME, Fiksdal AS, Kumbamu A, Sharp RR, McCormick JB. Patients’ views on incidental findings from clinical exome sequencing. Appl Transl Genomics. 2015;4:38–43.
Facio FM, Eidem H, Fisher T, et al. Intentions to receive individual results from whole-genome sequencing among participants in the ClinSeq study. Eur J Hum Genet. 2013;21:261–5.
Fernandez CV, Bouffet E, Malkin D, et al. Attitudes of parents toward the return of targeted and incidental genomic research findings in children. Genet Med. 2014;16:633–40.
Fernandez CV, OʼConnell C, Ferguson, et al. Stability of attitudes to the ethical issues raised by the return of incidental genomic research findings in children: a follow-up study. Public Health Genomics. 2015;18:299–308.
Gray SW, Park ER, Najita J, et al. Oncologists’ and cancer patients’ views on whole-exome sequencing and incidental findings: results from the CanSeq study. Genet Med. 2016;18:1011–9.
Kleiderman E, Knoppers BM, Fernandez CV, et al. Returning incidental findings from genetic research to children: views of parents of children affected by rare diseases. J Med Ethics. 2014;40:691–6.
Sanderson SC, Linderman MD, Suckiel SA, et al. Motivations, concerns and preferences of personal genome sequencing research participants: baseline findings from the HealthSeq project. Eur J Hum Genet. 2016;24:14–20.
Shahmirzadi L, Chao EC, Palmaer E, Parra MC, Tang S, Gonzalez KDF. Patient decisions for disclosure of secondary findings among the first 200 individuals undergoing clinical diagnostic exome sequencing. Genet Med. 2014;16:395–9.
Levenseller BL, Soucier DJ, Miller VA, et al. Stakeholders’ opinions on the implementation of pediatric whole exome sequencing: implications for informed consent. J Genet Couns. 2014;23:552–65.
Payne K, Fargher EA, Roberts SA, et al. Valuing pharmacogenetic testing services: a comparison of patients’ and health care professionals’ preferences. Value Health. 2011;14:121–34.
Severin F, Hess W, Schmidtke J, et al. Value judgements for priority setting criteria in genetic testing: a discrete choice experiment. Health Policy. 2015;119:164–73.
Middleton A, Morley KI, Bragin E, et al. Attitudes of nearly 7000 health professionals, genomic researchers and publics toward the return of incidental results from sequencing research. Eur J Hum Genet. 2016;24:21–29.
Krabbenborg L, Vissers LELM, Schieving J, et al. Understanding the psychosocial effects of WES test results on parents of children with rare diseases. J Genet Couns. 2016;25:1207–14.
Sapp JC, Johnston JJ, Driscoll K, et al. Evaluation of recipients of positive and negative secondary findings evaluations in a hybrid CLIA-research sequencing pilot. Am J Hum Genet. 2018;103:358–66.
Biesecker BB, Klein WMP, Lewis KL, et al. How do research participants perceive “uncertainty” in genome sequencing? Genet Med. 2014;16:977–980.
Han PKJ, Umstead KL, Bernhardt BA, et al. A taxonomy of medical uncertainties in clinical genome sequencing. Genet Med. 2017;19:918–25.
Newson AJ, Leonard SJ, Hall A, Gaff CL. Known unknowns: building an ethics of uncertainty into genomic medicine. BMC Med Genomics. 2016;9:57.
Patton, MQ. Qualitative evaluation checklist. Evaluation Checklists Project 2003: 1–13. available at: http://www.wmich.edu/evalctr/checklists/qec.pdf
Miles MB, Huberman AM, Saldana J. Qualitative data analysis. Third edition, Sage, Arizona State University. 2014.
Regier DA, Friedman JM, Makela N, Ryan M, Marra CA. Valuing the benefit of diagnostic testing for genetic causes of idiopathic developmental disability: willingness to pay from families of affected children. Clin Genet. 2009;75:514–21.
Buchannan J, Wordsworth S, Schuh A. Patients’ preferences for genomic diagnostic testing in chronic lymphocytic leukaemia: a discrete choice experiment. Patient. 2016;9:525–36.
Townsend A, Adam S, Birch PH, Lohn Z, Rousseau F, Friedman JM. “I want to know what’s in Pandora’s Box”: comparing stakeholder perspectives on incidental findings in clinical whole genomic sequencing. Am J Med Genet A. 2012;158A:2519–25.
Marshall DA, Gonzales JM, MacDonald KV, Johnson F. Estimating preferences for complex health technologies: lessons learned and implications for personalized medicine. Value Health. 2017;20:32–39.
Regier DA, Peacock SJ, Pataky R, et al. Societal preferences for the return of incidental findings from clinical genomic sequencing: a discrete-choice experiment. CMAJ. 2015;187:E190–197.
Houdayer F, Gargiulo M, Frischmann M, et al. The psychological impact of cryptic chromosomal abnormalities diagnosis announcement. Eur J Med Genet. 2013;56:585–90.
Krabbenborg L, Schieving J, Kleefstra T, et al. Evaluating a counselling strategy for diagnostic WES in pediatric neurology: an exploration of parents’ information and communication needs. Clin Genet. 2016;89:244–50.
Biesecker BB, Woolford SW, Klein WMP, et al. PUGS: a novel scale to assess perceptions of uncertainties in genome sequencing. Clin Genet. 2017;92:172–9.
Acknowledgements
The authors thank the Fondation Maladies Rares for its financial support of the study (APP Human and Social Sciences 2014) as well as the Conseil Régional de Bourgogne through the Plan d’Actions Régional pour l’Innovation (PARI 2015) and the European Union through the PO FEDER-FSE Bourgogne 2014/2020 programs. The authors also thank the parents who participated in the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Chassagne, A., Pélissier, A., Houdayer, F. et al. Exome sequencing in clinical settings: preferences and experiences of parents of children with rare diseases (SEQUAPRE study). Eur J Hum Genet 27, 701–710 (2019). https://doi.org/10.1038/s41431-018-0332-y
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-018-0332-y
This article is cited by
-
Approaches to Incorporation of Preferences into Health Economic Models of Genomic Medicine: A Critical Interpretive Synthesis and Conceptual Framework
Applied Health Economics and Health Policy (2025)
-
ECOLE: Learning to call copy number variants on whole exome sequencing data
Nature Communications (2024)
-
A content analysis of parents’ reflections on pathogenic and uncertain pediatric oncology germline sequencing results
Familial Cancer (2024)
-
Psychological and ethical issues raised by genomic in paediatric care pathway, a qualitative analysis with parents and childhood cancer patients
European Journal of Human Genetics (2024)
-
Expectations, needs and mid-term outcomes in people accessing to secondary findings from ES: 1st French mixed study (FIND Study)
European Journal of Human Genetics (2024)