Abstract
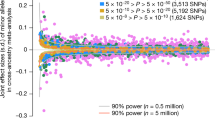
Contour integration, a key visual function to deal with occlusion and discontinuity in natural scenes, is essential to human survival. However, individuals are not equally well equipped with this ability. In particular, contour integration deficiencies are commonly detected in patients with mental disorders, especially schizophrenia. To understand the underlying sources of these individual differences, the current study investigated the genetic basis of contour integration in humans. A total of 2619 normal participants were tested on their ability to detect continuous contours embedded in a cluttered background. Quantitative genomic analysis was performed, involving heritability estimation based on single nucleotide polymorphisms (SNPs) and association testing at SNP, gene, and pathway levels. Heritability estimation showed that common SNPs contributed 49.5% (standard error of the mean = 15.6%) of overall phenotypic variation, indicating moderate heritability of contour integration. Two-stage genome-wide association analysis (GWAS) detected four SNPs reaching genome-wide significance in the discovery test (N = 1931) but not passing the replication test (N = 688). Gene-level analysis further revealed a significant genome-wide association of a microRNA-encoding gene MIR1178 in both the discovery and replication cohorts. Another gene poly(A)-binding protein nuclear 1 like, cytoplasmic (PABPN1L) showed suggestive significance in the discovery cohort (p < 1 × 10−4) and was replicated in the replication cohort (p = 0.009). The pathway analysis did not detect any significant pathway. Taken together, this study identified significant gene associations with contour integration and provided support for a genetic transmission of the ability to perceive continuous contours in the environment.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Field DJ, Hayes A, Hess RF. Contour integration by the human visual-system—evidence for a local association field. Vis Res. 1993;33:173–93.
Chandna A, Pennefather PM, Kovacs I, Norcia AM. Contour integration deficits in anisometropic amblyopia. Invest Ophth Vis Sci. 2001;42:875–8.
Phillips WA, Silverstein SM. Convergence of biological and psychological perspectives on cognitive coordination in schizophrenia. Behav Brain Sci. 2003;26:65–82.
Silverstein SM, Keane BP. Perceptual organization impairment in schizophrenia and associated brain mechanisms: review of research from 2005 to 2010. Schizophr Bull. 2011;37:690–9.
Feigenson KA, Gara MA, Roche MW, Silverstein SM. Is disorganization a feature of schizophrenia or a modifying influence: evidence of covariation of perceptual and cognitive organization in a non-patient sample. Psychiat Res. 2014;217:1–8.
Feigenson KA, Keane BP, Roche MW, Silverstein SM. Contour integration impairment in schizophrenia and first episode psychosis: state or trait? Schizophr Res. 2014;159:515–20.
Kovacs I. Human development of perceptual organization. Vis Res. 2000;40:1301–10.
Silverstein SM, Hatashita-Wong M, Schenkel LS, Wilkniss S, Kovacs I, Feher A, et al. Reduced top-down influences in contour detection in schizophrenia. Cogn Neuropsychiatry. 2006;11:112–32.
Davies G, Tenesa A, Payton A, Yang J, Harris SE, Liewald D, et al. Genome-wide association studies establish that human intelligence is highly heritable and polygenic. Mol Psychiatr. 2011;16:996–1005.
Piffer D. A review of intelligence GWAS hits: Their relationship to country IQ and the issue of spatial autocorrelation. Intelligence. 2015;53:43–50.
Sniekers S, Stringer S, Watanabe K, Jansen PR, Coleman JRI, Krapohl E, et al. Genome-wide association meta-analysis of 78,308 individuals identifies new loci and genes influencing human intelligence (vol 49, pg 1107, 2017). Nat Genet. 2017;49:1558.
Zabaneh D, Krapohl E, Gaspar HA, Curtis C, Lee SH, Patel H, et al. A genome-wide association study for extremely high intelligence. Mol Psychiatr. 2018;23:1226–32.
Debette S, Verbaas CAI, Bressler J, Schuur M, Smith A, Bis JC, et al. Genome-wide studies of verbal declarative memory in nondemented older people: the cohorts for heart and aging research in genomic epidemiology consortium. Biol Psychiat. 2015;77:749–63.
Milnik A, Heck A, Vogler C, Heinze HJ, de Quervain DJF, Papassotiropoulos A. Association of KIBRA with episodic and working memory: a meta-analysis. Am J Med Genet B. 2012;159b:958–69.
Papassotiropoulos A, Henke K, Stefanova E, Aerni A, Muller A, Demougin P, et al. A genome-wide survey of human short-term memory. Mol Psychiatr. 2011;16:184–92.
Papassotiropoulos A, Stefanova E, Vogler C, Gschwind L, Ackermann S, Spalek K, et al. A genome-wide survey and functional brain imaging study identify CTNNBL1 as a memory-related gene. Mol Psychiatr. 2013;18:255–63.
Vogler C, Gschwind L, Coynel D, Freytag V, Milnik A, Egli T, et al. Substantial SNP-based heritability estimates for working memory performance. Transl Psychiatry. 2014;4:e438.
Zhu Z, Chen B, Yan H, Fang W, Zhou Q, Zhou S, et al. Multi-level genomic analyses suggest new genetic variants involved in human memory. Eur J Hum Genet. 2018;26:1668–78.
Chen BQ, Zhu ZJ, Wang YY, Ding XH, Guo XB, He MG, et al. Nature vs. nurture in human sociality: multi-level genomic analyses of social conformity. J Hum Genet. 2018;63:605–19.
Chen BQ, Zhu ZJ, Na R, Fang W, Zhang WX, Zhou Q, et al. Genomic analyses of visual cognition: perceptual rivalry and top-down control. J Neurosci. 2018;38:9668–78.
Yang JA, Lee SH, Goddard ME, Visscher PM. GCTA: a tool for genome-wide complex trait analysis. Am J Hum Genet. 2011;88:76–82.
Bach M. The Freiburg visual acuity test—automatic measurement of visual acuity. Optom Vis Sci. 1996;73:49–53.
Li W, Piech V, Gilbert CD. Learning to link visual contours. Neuron. 2008;57:442–51.
Brainard DH. The psychophysics toolbox. Spat Vis. 1997;10:433–6.
Li W, Gilbert CD. Global contour saliency and local colinear interactions. J Neurophysiol. 2002;88:2846–56.
Li W, Piech V, Gilbert CD. Contour saliency in primary visual cortex. Neuron. 2006;50:951–62.
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet. 2006;38:904.
Yang JA, Benyamin B, McEvoy BP, Gordon S, Henders AK, Nyholt DR, et al. Common SNPs explain a large proportion of the heritability for human height. Nat Genet. 2010;42:565–U131.
Visscher PM, Hemani G, Vinkhuyzen AAE, Chen GB, Lee SH, Wray NR, et al. Statistical power to detect genetic (Co)variance of complex traits using SNP data in unrelated samples. PLoS Genet. 2014;10:e1004269.
Delaneau O, Marchini J, Zagury JF. A linear complexity phasing method for thousands of genomes. Nat Methods. 2012;9:179–81.
Howie B, Fuchsberger C, Stephens M, Marchini J, Abecasis GR. Fast and accurate genotype imputation in genome-wide association studies through pre-phasing. Nat Genet. 2012;44:955–9.
Howie B, Donnelly P, Marchini J. A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet. 2009;5:e1000529.
Marchini J, Howie B, Myers S, McVean G, Donnelly P. A new multipoint method for genome-wide association studies by imputation of genotypes. Nat Genet. 2007;39:906–13.
Barrett JC, Fry B, Maller J, Daly MJ. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics. 2005;21:263–5.
Gauderman WJ, Morrison JM. QUANTO 1.1: a computer program for power and sample size calculations for genetic-epidemiology studies. http://hydrauscedu/gxe. 2006.
Liu JZ, Mcrae AF, Nyholt DR, Medland SE, Wray NR, Brown KM, et al. A versatile gene-based test for genome-wide association studies. Am J Hum Genet. 2010;87:139–45.
Miller SM, Hansell NK, Ngo TT, Liu GB, Pettigrew JD, Martin NG, et al. Genetic contribution to individual variation in binocular rivalry rate. Proc Natl Acad Sci USA. 2010;107:2664–8.
Shannon RW, Patrick CJ, Jiang Y, Bernat E, He S. Genes contribute to the switching dynamics of bistable perception. J Vis. 2011;11:1–7.
Lau JYF, Burt M, Leibenluft E, Pine DS, Rijsdijk F, Shiffrin N, et al. Individual differences in children’s facial expression recognition ability: the role of nature and nurture. Dev Neuropsychol. 2009;34:37–51.
Wang Y, Wang L, Xu Q, Liu D, Chen LH, Troje NF, et al. Heritable aspects of biological motion perception and its covariation with autistic traits. Proc Natl Acad Sci USA. 2018;115:1937–42.
Robinson EB, Kirby A, Ruparel K, Yang J, McGrath L, Anttila V, et al. The genetic architecture of pediatric cognitive abilities in the Philadelphia Neurodevelopmental Cohort. Mol Psychiatr. 2015;20:454–8.
Coleman JRI, Lester KJ, Keers R, Munafo MR, Breen G, Eley TC. Genome-wide association study of facial emotion recognition in children and association with polygenic risk for mental health disorders. Am J Med Genet B. 2017;174:701–11.
Cao Z, Xu J, Huang H, Shen P, You L, Zhou L, et al. MiR-1178 promotes the proliferation, G1/S transition, migration and invasion of pancreatic cancer cells by targeting CHIP. PLoS ONE. 2015;10:e0116934.
Beecham GW, Dickson DW, Scott WK, Martin ER, Schellenberg G, Nuytemans K, et al. PARK10 is a major locus for sporadic neuropathologically confirmed Parkinson disease. Neurology. 2015;84:972–80.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Projects 31421003 and 31671168). We are grateful to Zhangyan Guan and Huizhen Yang for help with DNA preparation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhu, Z., Chen, B., Na, R. et al. Heritability of human visual contour integration—an integrated genomic study. Eur J Hum Genet 27, 1867–1875 (2019). https://doi.org/10.1038/s41431-019-0478-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-019-0478-2
This article is cited by
-
A genome-wide association study reveals a substantial genetic basis underlying the Ebbinghaus illusion
Journal of Human Genetics (2021)