Abstract
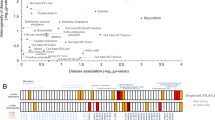
The various malformations of the aerodigestive tract collectively known as esophageal atresia/tracheoesophageal fistula (EA/TEF) constitute a rare group of birth defects of largely unknown etiology. Previous studies have identified a small number of rare genetic variants causing syndromes associated with EA/TEF. We performed a pilot exome sequencing study of 45 unrelated simplex trios (probands and parents) with EA/TEF. Thirteen had isolated and 32 had nonisolated EA/TEF; none had a family history of EA/TEF. We identified de novo variants in protein-coding regions, including 19 missense variants predicted to be deleterious (D-mis) and 3 likely gene-disrupting (LGD) variants. Consistent with previous studies of structural birth defects, there is a trend of increased burden of de novo D-mis in cases (1.57-fold increase over the background mutation rate), and the burden is greater in constrained genes (2.55-fold, p = 0.003). There is a frameshift de novo variant in EFTUD2, a known EA/TEF risk gene involved in mRNA splicing. Strikingly, 15 out of 19 de novo D-mis variants are located in genes that are putative target genes of EFTUD2 or SOX2 (another known EA/TEF gene), much greater than expected by chance (3.34-fold, p value = 7.20e−5). We estimated that 33% of patients can be attributed to de novo deleterious variants in known and novel genes. We identified APC2, AMER3, PCDH1, GTF3C1, POLR2B, RAB3GAP2, and ITSN1 as plausible candidate genes in the etiology of EA/TEF. We conclude that further genomic analysis to identify de novo variants will likely identify previously undescribed genetic causes of EA/TEF.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
All likely pathogenic variants are in ClinVar submission number SUB7053346. Accession numbers of submitted variants can be found in Supplemental Table 1.
References
Pinheiro PFM, e Silva ACS, Pereira RM. Current knowledge on esophageal atresia. World J Gastroenterol. 2012;18:3662.
Krishnan U, Mousa H, Dall’Oglio L, Homaira N, Rosen R, Faure C, et al. ESPGHAN-NASPGHAN guidelines for the evaluation and treatment of gastrointestinal and nutritional complications in children with esophageal atresia-tracheoesophageal fistula. J Pediatr Gastroenterol Nutr. 2016;63:550–70.
Stoll C, Alembik Y, Dott B, Roth M-P. Associated malformations in patients with esophageal atresia. Eur J Med Genet. 2009;52:287–90.
Shaw-Smith C. Genetic factors in esophageal atresia, tracheo-esophageal fistula and the VACTERL association: roles for FOXF1 and the 16q24. 1 FOX transcription factor gene cluster, and review of the literature. Eur J Med Genet. 2010;53:6–13.
Geneviève D, de Pontual L, Amiel J, Lyonnet S. Genetic factors in isolated and syndromic esophageal atresia. J Pediatr Gastroenterol Nutr. 2011;52:S6–8.
Felix JF, Tibboel D, de Klein A. Chromosomal anomalies in the aetiology of oesophageal atresia and tracheo-oesophageal fistula. Eur J Med Genet. 2007;50:163–75.
Murphy AJ, Li Y, Pietsch JB, Chiang C, Lovvorn HN. Mutational analysis of NOG in esophageal atresia and tracheoesophageal fistula patients. Pediatr Surg Int. 2012;28:335–40.
Que J, Okubo T, Goldenring JR, Nam K-T, Kurotani R, Morrisey EE, et al. Multiple dose-dependent roles for Sox2 in the patterning and differentiation of anterior foregut endoderm. Development. 2007;134:2521–31.
Kormish JD, Sinner D, Zorn AM. Interactions between SOX factors and Wnt/β-catenin signaling in development and disease. Dev Dyn. 2010;239:56–68.
Morrisey EE, Hogan BL. Preparing for the first breath: genetic and cellular mechanisms in lung development. Dev Cell. 2010;18:8–23.
Gordon CT, Petit F, Oufadem M, Decaestecker C, Jourdain AS, Andrieux J, et al. EFTUD2 haploinsufficiency leads to syndromic oesophageal atresia. J Med Genet. 2012;49:737–46.
Voigt C, Mégarbané A, Neveling K, Czeschik JC, Albrecht B, Callewaert B, et al. Oto-facial syndrome and esophageal atresia, intellectual disability and zygomatic anomalies-expanding the phenotypes associated with EFTUD2 mutations. Orphanet J Rare Dis. 2013;8:110.
Lines MA, Huang L, Schwartzentruber J, Douglas SL, Lynch DC, Beaulieu C, et al. Haploinsufficiency of a spliceosomal GTPase encoded by EFTUD2 causes mandibulofacial dysostosis with microcephaly. Am J Hum Genet. 2012;90:369–77.
Zhang X, Yan C, Hang J, Finci LI, Lei J, Shi Y. An atomic structure of the human spliceosome. Cell. 2017;169:918–29e.14.
Bertram K, Agafonov DE, Dybkov O, Haselbach D, Leelaram MN, Will CL, et al. Cryo-EM structure of a pre-catalytic human spliceosome primed for activation. Cell. 2017;170:701–13.e11.
Schulz AC, Bartels E, Stressig R, Ritgen J, Schmiedeke E, Mattheisen M, et al. Nine new twin pairs with esophageal atresia: a review of the literature and performance of a twin study of the disorder. Birth Defects Res Part A: Clin Mol Teratol. 2012;94:182–6.
Maroszyńska I, Fortecka-Piestrzeniewicz K, Niedźwiecka M, Żarkowska-Szaniawska A. Isolated esophageal atresia in both premature twins. Pediatr Pol. 2015;90:91–3.
Shaw-Smith C. Oesophageal atresia, tracheo-oesophageal fistula, and the VACTERL association: review of genetics and epidemiology. J Med Genet. 2006;43:545–54.
Zhang Y, Jiang M, Kim E, Lin S, Liu K, Que J, et al. Development and stem cells of the esophagus. Semin Cell Dev Biol. 2017;66:25–35.
Al-Salem AH, Kothari M, Oquaish M, Khogeer S, Desouky MS. Morbidity and mortality in esophageal atresia and tracheoesophageal fistula: a 20-year review. Ann Pediatr Surg. 2013;9:93–8.
Li H. Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. arXiv preprint arXiv:13033997. 2013. https://arxiv.org/abs/1303.3997.
DePristo MA, Banks E, Poplin R, Garimella KV, Maguire JR, Hartl C, et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet. 2011;43:491.
Homsy J, Zaidi S, Shen Y, Ware JS, Samocha KE, Karczewski KJ, et al. De novo mutations in congenital heart disease with neurodevelopmental and other congenital anomalies. Science. 2015;350:1262–6.
Qi H, Yu L, Zhou X, Wynn J, Zhao H, Guo Y, et al. De novo variants in congenital diaphragmatic hernia identify MYRF as a new syndrome and reveal genetic overlaps with other developmental disorders. PLoS Genet. 2018;14:e1007822.
Thorvaldsdóttir H, Robinson JT, Mesirov JP. Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform. 2013;14:178–92.
Fromer M, Moran JL, Chambert K, Banks E, Bergen SE, Ruderfer DM, et al. Discovery and statistical genotyping of copy-number variation from whole-exome sequencing depth. Am J Hum Genet. 2012;91:597–607.
Wang K, Li M, Hakonarson H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010;38:e164.
Lek M, Karczewski KJ, Minikel EV, Samocha KE, Banks E, Fennell T, et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature. 2016;536:285.
Kircher M, Witten DM, Jain P, O’roak BJ, Cooper GM, Shendure J. A general framework for estimating the relative pathogenicity of human genetic variants. Nat Genet. 2014;46:310.
Ioannidis NM, Rothstein JH, Pejaver V, Middha S, McDonnell SK, Baheti S, et al. REVEL: an ensemble method for predicting the pathogenicity of rare missense variants. Am J Hum Genet. 2016;99:877–85.
Van Nostrand EL, Freese P, Pratt GA, Wang X, Wei X, Blue SM, et al. A large-scale binding and functional map of human RNA binding proteins. bioRxiv. 2018. https://doi.org/10.1101/179648.
Feng H, Bao S, Rahman MA, Weyn-Vanhentenryck SM, Khan A, Wong J, et al. Modeling RNA-binding protein specificity in vivo by precisely registering protein-RNA crosslink sites. Mol Cell. 2019;74:1189–1204.e6.
Sarkar A, Huebner AJ, Sulahian R, Anselmo A, Xu X, Flattery K, et al. Sox2 suppresses gastric tumorigenesis in mice. Cell Rep. 2016;16:1929–41.
Lachmann A, Xu H, Krishnan J, Berger SI, Mazloom AR, Ma’ayan A. ChEA: transcription factor regulation inferred from integrating genome-wide ChIP-X experiments. Bioinformatics. 2010;26:2438–44.
Han X, Chen S, Flynn E, Wu S, Wintner D, Shen Y. Distinct epigenomic patterns are associated with haploinsufficiency and predict risk genes of developmental disorders. Nat Commun. 2018;9:2138.
Samocha KE, Robinson EB, Sanders SJ, Stevens C, Sabo A, McGrath LM, et al. A framework for the interpretation of de novo mutation in human disease. Nat Genet. 2014;46:944.
Walsh R, Mazzarotto F, Whiffin N, Buchan R, Midwinter W, Wilk A, et al. Quantitative approaches to variant classification increase the yield and precision of genetic testing in Mendelian diseases: the case of hypertrophic cardiomyopathy. Genome Med. 2019;11:5.
Jin SC, Homsy J, Zaidi S, Lu Q, Morton S, DePalma SR, et al. Contribution of rare inherited and de novo variants in 2871 congenital heart disease probands. Nat Genet. 2017;49:1593–601.
Deciphering Developmental Disorders S. Prevalence and architecture of de novo mutations in developmental disorders. Nature. 2017;542:433–8.
Feliciano P, Zhou X, Astrovskaya I, Turner T, Wang T, Brueggeman L, et al. Exome sequencing of 457 autism families recruited online provides evidence for novel ASD genes. bioRxiv. 2019: 516625. https://www.biorxiv.org/content/10.1101/516625v1.
He X, Sanders SJ, Liu L, De Rubeis S, Lim ET, Sutcliffe JS, et al. Integrated model of de novo and inherited genetic variants yields greater power to identify risk genes. PLoS Genet. 2013;9:e1003671.
Hussain NK, Jenna S, Glogauer M, Quinn CC, Wasiak S, Guipponi M, et al. Endocytic protein intersectin-l regulates actin assembly via Cdc42 and N-WASP. Nat Cell Biol. 2001;3:927–32.
Takatsu H, Sakurai M, Shin HW, Murakami K, Nakayama K. Identification and characterization of novel clathrin adaptor-related proteins. J Biol Chem. 1998;273:24693–700.
Ogawa M, Yoshikawa Y, Kobayashi T, Mimuro H, Fukumatsu M, Kiga K, et al. A Tecpr1-dependent selective autophagy pathway targets bacterial pathogens. Cell Host Microbe. 2011;9:376–89.
Spang N, Feldmann A, Huesmann H, Bekbulat F, Schmitt V, Hiebel C, et al. RAB3GAP1 and RAB3GAP2 modulate basal and rapamycin-induced autophagy. Autophagy. 2014;10:2297–309.
Gaudet P, Livstone MS, Lewis SE, Thomas PD. Phylogenetic-based propagation of functional annotations within the Gene Ontology consortium. Brief Bioinform. 2011;12:449–62.
Mische SM, Mooseker MS, Morrow JS. Erythrocyte adducin: a calmodulin-regulated actin-bundling protein that stimulates spectrin-actin binding. J Cell Biol. 1987;105:2837–45.
Brauburger K, Akyildiz S, Ruppert JG, Graeb M, Bernkopf DB, Hadjihannas MV, et al. Adenomatous polyposis coli (APC) membrane recruitment 3, a member of the APC membrane recruitment family of APC-binding proteins, is a positive regulator of Wnt-beta-catenin signalling. FEBS J. 2014;281:787–801.
Zhang X, Zhang J, Bauer A, Zhang L, Selinger DW, Lu CX, et al. Fine-tuning BMP7 signalling in adipogenesis by UBE2O/E2-230K-mediated monoubiquitination of SMAD6. EMBO J. 2013;32:996–1007.
Au AC, Hernandez PA, Lieber E, Nadroo AM, Shen YM, Kelley KA, et al. Protein tyrosine phosphatase PTPN14 is a regulator of lymphatic function and choanal development in humans. Am J Hum Genet. 2010;87:436–44.
Acknowledgements
We would like to acknowledge the patients and their families who participated in the study and are grateful for their tremendous contribution. We thank Steve Wyles and Sue Paul from EA for adults, as well as the TOFS UK, OARA, Bridging the Gap of EA/TEF and the Canadian EA network organizations for publicizing the study and assisting with recruitment. We are also appreciative for the technical assistance provided by Patricia Lanzano, Jiangyuan Hu, Liyong Deng, Nikita Chintalapudi, and Charles LeDuc from Columbia University and the study team at Cairo University General Hospital. We thank Na Zhu for help with the calculation of background mutation rate. Funding support provided by P01HD093363 (JW, YS, and WKC) and R01GM120609 (YS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, J., Ahimaz, P.R., Hashemifar, S. et al. Novel candidate genes in esophageal atresia/tracheoesophageal fistula identified by exome sequencing. Eur J Hum Genet 29, 122–130 (2021). https://doi.org/10.1038/s41431-020-0680-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-020-0680-2
This article is cited by
-
Single-cell analysis reveals the spatial-temporal expression of genes associated with esophageal malformations
Scientific Reports (2024)
-
Ätiologische Konzepte von gastrointestinalen Fehlbildungen/Atresien
Monatsschrift Kinderheilkunde (2023)