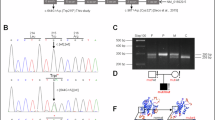
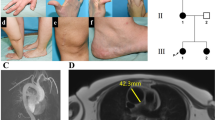
Abstract
Congenital autosomal recessive cataract with unknown genetic etiology is one of the most common Mendelian diseases among the Turkic-speaking Yakut population (Eastern Siberia, Russia). To identify the genetic cause of congenital cataract spread in this population, we performed whole-exome sequencing (Illumina NextSeq 500) in one Yakut family with three affected siblings whose parents had preserved vision. We have revealed the novel homozygous c.1621C>T transition leading to premature stop codon p.(Gln541*) in exon 8 of the FYCO1 gene (NM_024513.4). Subsequent screening of c.1621C>T p.(Gln541*) revealed this variant in a homozygous state in 25 out of 29 Yakut families with congenital cataract (86%). Among 424 healthy individuals from seven populations of Eastern Siberia (Russians, Yakuts, Evenks, Evens, Dolgans, Chukchi, and Yukaghirs), the highest carrier frequency of c.1621C>T p.(Gln541*) was found in the Yakut population (7.9%). DNA samples of 25 homozygous for c.1621C>T p.(Gln541*) patients with congenital cataract and 114 unaffected unrelated individuals without this variant were used for a haplotype analysis based on the genotyping of six STR markers (D3S3512, D3S3685, D3S3582, D3S3561, D3S1289, and D3S3698). The structure of the identified haplotypes indicates a common origin for all of the studied mutant chromosomes bearing c.1621C>T p.(Gln541*). The age of the с.1621C>T p.(Gln541*) founder haplotype was estimated to be approximately 260 ± 65 years (10 generations). These findings characterize Eastern Siberia as the region of the world with the most extensive accumulation of the unique variant c.1621C>T p.(Gln541*) in the FYCO1 gene as a result of the founder effect.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Robinson G, Jan J, Kinnis C. Congenital ocular blindness in children, 1945 to 1984. Am J Dis Child. 1987;141:1321–4. https://doi.org/10.1001/archpedi.1987.04460120087041.
Lambert S, Drack A. Infantile cataracts. Surv Ophthalmol. 1996;40:427–58. https://doi.org/10.1016/S0039-6257(96)82011-X.
Merin S. Inherited cataracts. In: Merin S, editor. Inherited eye diseases. New York: Marcel Dekker, Inc.; 1991.
Shiels A, Bennett T, Hejtmancik J. Cat-Map: putting cataract on the map. Mol Vis. 2010;16:2007–15.
Pras E, Frydman M, Levy-Nissenbaum E, Bakhan T, Raz J, Assia E, et al. A nonsense mutation (W9X) in CRYAA causes autosomal recessive cataract in an inbred Jewish Persian family. Investig Ophthalmol Vis Sci. 2000;41:3511–5.
Pras E, Bakhan T, Levy-Nissenbaum E, Lahat H, Assia E, Garzozi H, et al. A gene causing autosomal recessive cataract maps to the short arm of chromosome 3. Isr Med Assoc J. 2001;3:559–62.
Pras E, Levy-Nissenbaum E, Bakhan T, Lahat H, Assia E, Geffen-Carmi N, et al. A missense mutation in the LIM2 gene is associated with autosomal recessive presenile cataract in an inbred Iraqi Jewish family. Am J Hum Genet. 2002;70:1363–7. https://doi.org/10.1086/340318.
Pras E, Raz J, Yahalom V, Frydman M, Garzozi H, Hejtmancik J, et al. A nonsense mutation in the glucosaminyl (N-acetyl) transferase 2 gene (GCNT2): association with autosomal recessive congenital cataracts. Investig Ophthalmol Vis Sci. 2004;45:1940–5. https://doi.org/10.1167/iovs.03-1117.
Smaoui N, Beltaief O, BenHamed S, M’Rad R, Maazoul F, Ouertani A, et al. A homozygous splice mutation in the HSF4 gene is associated with an autosomal recessive congenital cataract. Investig Ophthalmol Vis Sci. 2004;45:2716–21. https://doi.org/10.1167/iovs.03-1370.
Riazuddin S, Yasmeen A, Yao W, Sergeev Y, Zhang Q, Zulfiqar F, et al. Mutations in beta B3-crystallin associated with autosomal recessive cataract in two Pakistani families. Investig Ophthalmol Vis Sci. 2005;46:2100–6. https://doi.org/10.1167/iovs.04-1481.
Cohen D, Bar-Yosef U, Levy J, Gradstein L, Belfair N, Ofir R, et al. Homozygous CRYBB1 deletion mutation underlies autosomal recessive congenital cataract. Investig Ophthalmol Vis Sci. 2007;48:2208–13. https://doi.org/10.1167/iovs.06-1019.
Ponnam S, Ramesha K, Tejwani S, Matalia J, Kannabiran C. A missense mutation in LIM2 causes autosomal recessive congenital cataract. Mol Vis. 2008;14:1204–8.
Ponnam S, Ramesha K, Tejwani S, Ramamurthy B, Kannabiran C. Mutation of the gap junction protein alpha 8 (GJA8) gene causes autosomal recessive cataract. J Med Genet. 2007;44:e85. https://doi.org/10.1136/jmg.2007.050138.
Ramachandran R, Perumalsamy V, Hejtmancik J. Autosomal recessive juvenile onset cataract associated with mutation in BFSP1. Hum Genet. 2007;121:475–82. https://doi.org/10.1007/s00439-006-0319-6.
Kaul H, Riazuddin S, Shahid M, Kousar S, Butt N, Zafar A, et al. Autosomal recessive congenital cataract linked to EPHA2 in a consanguineous Pakistani family. Mol Vis. 2010;16:511–7.
Pankiv S, Johansen T. FYCO1 linking autophagosomes to microtubule plus end-directing molecular motors. Autophagy. 2010;6:550–2. https://doi.org/10.4161/auto.6.4.11670.
Chen J, Ma Z, Jiao X, Fariss R, Kantorow W, Kantorow M, et al. Mutations in FYCO1 cause autosomal-recessive congenital cataracts. Am J Hum Genet. 2011;88:827–38. https://doi.org/10.1016/j.ajhg.2011.05.008.
Aldahmesh M, Khan A, Mohamed J, Alghamdi M, Alkuraya F. Identification of a truncation mutation of acylglycerol kinase (AGK) gene in a novel autosomal recessive cataract locus. Hum Mutat. 2012;33:960–2. https://doi.org/10.1002/humu.22071.
Evers C, Paramasivam N, Hinderhofer K, Fischer C, Granzow M, Schmidt-Bacher A, et al. SIPA1L3 identified by linkage analysis and whole-exome sequencing as a novel gene for autosomal recessive congenital cataract. Eur J Hum Genet. 2015;23:1627–33.
Ansar M, Chung H, Taylor RL, Nazir A, Imtiaz S, Sarwar MT, et al. Bi-allelic loss-of-function variants in DNMBP cause infantile cataracts. Am J Hum Genet. 2018;103:568–78. https://doi.org/10.1016/j.ajhg.2018.09.004.
Jiao X, Khan SY, Irum B, Khan AO, Wang Q, Kabir F, et al. Missense mutations in CRYAB are liable for recessive congenital cataracts. PLoS ONE. 2015;10:e0137973. https://doi.org/10.1371/journal.pone.0137973.
Khan SY, Vasanth S, Kabir F, Gottsch JD, Khan AO, Chaerkady R, et al. FOXE3 contributes to Peters anomaly through transcriptional regulation of an autophagy-associated protein termed DNAJB1. Nat Commun. 2016;7:10953. https://doi.org/10.1038/ncomms10953.
Lachke SA, Alkuraya FS, Kneeland SC, Ohn T, Aboukhalil A, Howell GR, et al. Mutations in the RNA granule component TDRD7 cause cataract and glaucoma. Science. 2011;331:1571–6. https://doi.org/10.1126/science.1195970.
Yasmeen A, Riazuddin SA, Kaul H, Mohsin S, Khan M, Qazi ZA, et al. Autosomal recessive congenital cataract in consanguineous Pakistani families is associated with mutations in GALK1. Mol Vis. 2010;16:682–8.
Zhao L, Chen XJ, Zhu J, Xi YB, Yang X, Hu LD, et al. Lanosterol reverses protein aggregation in cataracts. Nature. 2015;523:607–11. https://doi.org/10.1038/nature14650.
Micheal S, Niewold ITSGl, Siddiqui SN, Zafar SN, Khan MI, Bergen AA. Delineation of novel autosomal recessive mutation in GJA3 and autosomal dominant mutations in GJA8 in Pakistani congenital cataract families. Genes. 2018;9:112. https://doi.org/10.3390/genes9020112.
Chacon-Camacho O, Buentello-Volante B, Velazquez-Montoya R, Ayala-Ramirez R, Zenteno J. Homozygosity mapping identifies a GALK1 mutation as the cause of autosomal recessive congenital cataracts in 4 adult siblings. Gene. 2014;534:218–21. https://doi.org/10.1016/j.gene.2013.10.057.
Patel N, Anand D, Monies D, Maddirevula S, Khan A, Algoufi T, et al. Novel phenotypes and loci identified through clinical genomics approaches to pediatric cataract. Hum Genet. 2017;136:205–25. https://doi.org/10.1007/s00439-016-1747-6.
Polyakov A, Shagina I, Khlebnikova O, Evgrafov O. Mutation in the connexin 50 gene (GJA8) in a Russian family with zonular pulverulent cataract. Clin Genet. 2001;60:476–8. https://doi.org/10.1034/j.1399-0004.2001.600614.x.
Aznabaev MT, Khidiyatova II, Khidiyatova IM, Avkhadeeva SR, Dzhemileva LU, Khusnutdinova EK. [Mutatsii v gene CRYAA - odna iz prichin razvitiya nasledstvennoy vrozhdennoy katarakty]. Meditsinskiy Vestn Bashkortostana. 2014;9:7–9.
Khidiyatova II, Aznabaev MT, Khidiyatova IM, Avkhadeeva SR, Dzhemileva LU, Zinchenko RA, et al. [Analiz gena Konneksina 50 (GJA8) u bol’nykh s nasledstvennoy vrozhdonnoy kataraktoy iz respubliki Bashkortostan]. Meditsinskaya genetika 2014;13:37–42.
Khidiyatova II, Aznabaev MT, Khidiyatova IM, Avkhadeeva SR, Dzhemileva LU, Khusnutdinova EK. [Analiz gena konneksina 46 (GJA3) u bol’nykh nasledstvennoy vrozhdennoy kataraktoy respubliki Bashkortostan]. Meditsinskiy Vestn Bashkortostana. 2016;11:16–20.
Tarskaia L, Zinchenko R, Elchinova G, Egorova A, Korotov M, Basova E, et al. The structure and diversity of hereditary pathology in Sakha Republic (Yakutia). Russ J Genet. 2004;40:1264–72. https://doi.org/10.1023/B:RUGE.0000048669.22362.1c.
Fedorova S, Reidla M, Metspalu E, Metspalu M, Rootsi S, Tambets K et al. Autosomal and uniparental portraits of the native populations of Sakha (Yakutia): implications for the peopling of Northeast Eurasia. BMC Evol Biol. 2013;13. https://doi.org/10.1186/1471-2148-13-127.
Galeeva N, Voevoda M, Spiridonova M, Stepanov V, Polyakov A. Population frequency and age of c.806C>T mutation in CYB5R3 gene as cause of recessive congenital methemoglobinemia in Yakutia. Russ J Genet. 2013;49:457–63. https://doi.org/10.1134/S102279541303006X.
Puzyrev V, Maximova N. Hereditary diseases among Yakuts. Russ J Genet. 2008;44:1141–7. https://doi.org/10.1134/S1022795408100037.
Maksimova N, Hara K, Nikolaeva I, Chun-Feng T, Usui T, Takagi M, et al. Neuroblastoma amplified sequence gene is associated with a novel short stature syndrome characterised by optic nerve atrophy and Pelger-Huet anomaly. J Med Genet. 2010;47:538–48. https://doi.org/10.1136/jmg.2009.074815.
Maksimova N, Hara K, Miyashia A, Nikolaeva I, Shiga A, Nogovicina A, et al. Clinical, molecular and histopathological features of short stature syndrome with novel CUL7 mutation in Yakuts: new population isolate in Asia. J Med Genet. 2007;44:772–8. https://doi.org/10.1136/jmg.2007.051979.
Barashkov NA, Dzhemileva LU, Fedorova SA, Teryutin FM, Posukh OL, Fedotova EE, et al. Autosomal recessive deafness 1A (DFNB1A) in Yakut population isolate in Eastern Siberia: extensive accumulation of the splice site mutation IVS1+1G>A in GJB2 gene as a result of founder effect. J Hum Genet. 2011;56:631–9. https://doi.org/10.1038/jhg.2011.72.
Risch N, de Leon D, Ozelius L, Kramer P, Almasy L, Singer B, et al. Genetic analysis of idiopathic torsion dystonia in Ashkenazi Jews and their recent descent from a small founder population. Nat Genet. 1995;9:152–9. https://doi.org/10.1038/ng0295-152.
Bengtsson B, Thomson G. Measuring the strength of associations between HLA antigens and diseases. Tissue Antigens. 1981;18:356–63. https://doi.org/10.1111/j.1399-0039.1981.tb01404.x.
Khan AO, Aldahmesh MA, Alkuraya FS. Phenotypes of recessive pediatric cataract in a cohort of children with identified homozygous gene mutations (An American Ophthalmological Society Thesis). Trans Am Ophthalmol Soc. 2015;113:T7.
Chen J, Wang Q, Cabrera P, Zhong Z, Sun W, Jiao X, et al. Molecular genetic analysis of Pakistani families with autosomal recessive congenital cataracts by homozygosity screening. Investig Ophthalmol Vis Sci. 2017;58:2207–17. https://doi.org/10.1167/iovs.17-21469.
Li J, Leng Y, Han S, Yan L, Lu C, Luo Y, et al. Clinical and genetic characteristics of Chinese patients with familial or sporadic pediatric cataract. Orphanet J Rare Dis. 2018;13. https://doi.org/10.1186/s13023-018-0828-0.
Iqbal H, Khan SY, Zhou L, Irum B, Ali M, Ahmed MR, et al. Mutations in FYCO1 identified in families with congenital cataracts. Mol Vis. 2020;26:334–44.
Gillespie RL, O’Sullivan J, Ashworth J, Bhaskar S, Williams S, Biswas S, et al. Personalized diagnosis and management of congenital cataract by next-generation sequencing. Ophthalmology. 2014;121:2124–37. https://doi.org/10.1016/j.ophtha.2014.06.006.
Gogolev AI [Etnicheskaya istoriya narodov Yakutii (do nachala XX veka)]. 103. Yakutsk: Izd-vo YAGU; 2004
Marusin A, Kurtanov H, Maksimova N, Swarovsakaja M, Stepanov V. Haplotype analysis of oculopharyngeal muscular dystrophy (OPMD) locus in Yakutia. Russ J Genet. 2016;52:331–8. https://doi.org/10.1134/S1022795416030091.
Swarovskaya M, Stepanova S, Marussin A, Sukhomyasova A, Maximova N, Stepanov V. Genetic variability and structure of SNP haplotypes in the DMPK gene in Yakuts and other ethnic groups of northern Eurasia in relation to myotonic dystrophy. Russ J Genet. 2015;51:619–26. https://doi.org/10.1134/S1022795415060150.
Osakovskiy VL, Goldfarb LG, Platonov FA. [K voprosu proiskhozhdeniya SCA1 mutatsii v yakutskoy populyatsii]. Bulletin’ SO RAMN. 2004;1:103–4.
Acknowledgements
We thank all the patients and blood sample donors who contributed to this study. Special thanks to the physicians and tutors of the special boarding school for blind children (Yakutsk, Russia).
Funding
The study was supported by the Project of the Ministry of Science and Higher Education of the Russian Federation (basic part of funding to M.K. Ammosov North-Eastern Federal University #FSRG-2020–0016) and by the Russian Foundation for Basic Research (grants #18–05–600035_Arctika, #19–34–60023_Perspektiva).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments and wrote the paper: NAB, FAK, TVB, and FMT; performed the experiments: AVS, VGP, NVS, and LSV; analyzed the data: GPR, NNG, IVM, and AAB; collected the samples: FMT, VGP, FAP, and TEB; discussed and proofed the paper: EKK, OLP, and SAF.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Barashkov, N.A., Konovalov, F.A., Borisova, T.V. et al. Autosomal recessive cataract (CTRCT18) in the Yakut population isolate of Eastern Siberia: a novel founder variant in the FYCO1 gene. Eur J Hum Genet 29, 965–976 (2021). https://doi.org/10.1038/s41431-021-00833-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-021-00833-w
This article is cited by
-
Exploiting autophagy and related pathways: pioneering new horizons in cataract therapy
Apoptosis (2025)
-
Cataracts in Havanese: genome wide association study reveals two loci associated with posterior polar cataract
Canine Medicine and Genetics (2023)
-
Detection of an FYCO1 nonsense mutation in an affected patient with autosomal recessive cataract (CTRCT18): a case report
Egyptian Journal of Medical Human Genetics (2022)