Abstract
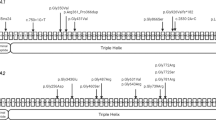
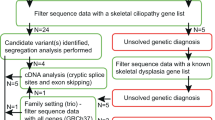
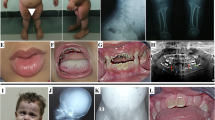
Skeletal dysplasias are a clinically and genetically heterogeneous group of rare disorders. Studies from large cohorts are essential to provide insights into the disease epidemiology, phenotypic spectrum, and mutational profiles. Here we enumerate additional 248 Indians from 197 families with a skeletal dysplasia, following a similar study earlier. We achieved a clinical-molecular diagnosis in 145 families by targeted analysis in 37 and next generation sequencing (exomes and genomes) in 108 families that resulted in a diagnostic yield of 73.6% (145 of 197 families). We identified 149 causal variants, of which 85 were novel, across 73 genes. Eighty-one distinct monogenic forms of skeletal dysplasia were observed with a high proportion of autosomal recessive skeletal dysplasias (60%, 84 families). We observed consanguinity in 35% of the families. Lysosomal storage diseases with skeletal involvement, FGFR3-related skeletal dysplasia and disorders of bone mineralisation were most frequent in this cohort. We expand the phenotypic and genotypic spectrum of rarely reported conditions (RAB33B, TRIP11, NEPRO, RPL13, COL27A1, PTHR1, EXOC6B, PRKACA, FUZ and RSPRY1) and noted novel gene-disease relationships for PISD, BNIP1, TONSL, CCN2 and SCUBE3 related skeletal dysplasia. We successfully implemented genomic testing for skeletal dysplasia in clinical and research settings. Our study provides valuable information on the spectrum of skeletal dysplasia and disease-causing variants for Asian Indians.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
Most of the data relevant to this study are included here. However, any additional information about the study is available from the corresponding authors upon reasonable request.
References
Unger S., Ferreira C. R., Mortier G. R., Ali H., Bertola D. R., Calder A. et al. Nosology of genetic skeletal disorders: 2023 revision. Am J Med Genet A. 2023;191:1164–1209.
Spranger J. Pattern recognition in bone dysplasias. Prog Clin Biol Res. 1985;200:315–42.
Uttarilli A, Shah H, Bhavani GS, Upadhyai P, Shukla A, Girisha KM. Phenotyping and genotyping of skeletal dysplasias: evolution of a center and a decade of experience in India. Bone. 2019;120:204–11.
Nampoothiri S, Yesodharan D, Sainulabdin G, Narayanan D, Padmanabhan L, Girisha KM, et al. Eight years experience from a skeletal dysplasia referral center in a tertiary hospital in Southern India: a model for the diagnosis and treatment of rare diseases in a developing country. Am J Med Genet A. 2014;164A(9):2317–23.
Krakow D, Rimoin DL. The skeletal dysplasias. Genet Med. 2010;12(6):327–41.
Nakatsuka N, Moorjani P, Rai N, Sarkar B, Tandon A, Patterson N, et al. The promise of discovering population-specific disease-associated genes in South Asia. Nat Genet. 2017;49(9):1403–7.
Bhasin MK, Nag S. Consanguinity and its effects on fertility, mortality and morbidity in the Indian region: a review. Int J Human Genet. 2012;12(4):197–301.
Kulkarni ML, Samuel K, Bhagyavathi M, Sureshkumar C. Skeletal dysplasias in a hospital in southern India. Ind Pediatr. 1995;32(6):657–65.
Sheth J, Bhavsar R, Mistri M, Pancholi D, Bavdekar A, Dalal A, et al. Gaucher disease: single gene molecular characterization of one-hundred Indian patients reveals novel variants and the most prevalent mutation. BMC Med Genet. 2019;20(1):31.
Holling T, Bhavani GS, von, Elsner L, Shah H, Kausthubham N, Bhattacharyya SS, et al. A homozygous hypomorphic BNIP1 variant causes an increase in autophagosomes and reduced autophagic flux and results in a spondylo-epiphyseal dysplasia. Hum Mutat. 2022;43(5):625–42.
Lin YC, Niceta M, Muto V, Vona B, Pagnamenta AT, Maroofian R, et al. SCUBE3 loss-of-function causes a recognizable recessive developmental disorder due to defective bone morphogenetic protein signaling. Am J Hum Genet. 2021;108(1):115–33.
Girisha KM, von Elsner L, Neethukrishna K, Muranjan M, Shukla A, Bhavani GS, et al. The homozygous variant c.797G>A/p.(Cys266Tyr) in PISD is associated with a Spondyloepimetaphyseal dysplasia with large epiphyses and disturbed mitochondrial function. Hum Mutat. 2019;40(3):299–309.
Girisha KM, Pande S, Dalal A, Phadke SR. Untapped opportunities for rare disease gene discovery in India. Am J Med Genet A. 2020;182(12):3056–9.
Kausthubham N, Shukla A, Gupta N, Bhavani GS, Kulshrestha S, Das Bhowmik A, et al. A dataset of variants derived from 1455 clinical and research exomes is efficient in variant prioritization for early-onset monogenic disorders in Indians. Hum Mutat. 2021;42(4):e15–61.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17(5):405–24.
Girisha KM, Kortüm F, Shah H, Alawi M, Dalal A, Bhavani GS, et al. A novel multiple joint dislocation syndrome associated with a homozygous nonsense variant in the EXOC6B gene. Eur J Human Genet. 2016;24(8):1206–10.
Girisha KM, Bhavani GS, Shah H, Moirangthem A, Shukla A, Kim OH, et al. Biallelic variants p.Arg1133Cys and p.Arg1379Cys in COL2A1: Further delineation of phenotypic spectrum of recessive Type 2 collagenopathies. Am J Med Genet A. 2020;182(2):338–47.
Chang HR, Cho SY, Lee JH, Lee E, Seo J, Lee HR, et al. Hypomorphic mutations in TONSL Cause SPONASTRIME dysplasia. Am J Hum Genet. 2019;104(3):439–53.
Singh S, Danda S, Sharma N, Shah H, Madhuri V, Mir TA, et al. Biallelic variants in CCN2 underlie an autosomal recessive kyphomelic dysplasia. Eur J Hum Genet. 2024 Nov 6; Published online
Singh S., Shah H., Dalal A., Shukla A., Bhavani G. S., Girisha K. M. Two sisters with RSPRY1-related spondyloepimetaphyseal dysplasia. Am J Med Genet A. 2024, 2;e63601.
Singh S, Nampoothiri S, Narayanan DL, Chaudhry C, Salvankar S, Girisha KM. Biallelic loss of function variants in FUZ result in an orofaciodigital syndrome. Eur J Hum Genet. 2024;32:1022–1026.
Jacob P, Lindelöf H, Rustad CF, Sutton VR, Moosa S, Udupa P, et al. Clinical, genetic and structural delineation of RPL13-related spondyloepimetaphyseal dysplasia suggest extra-ribosomal functions of eL13. NPJ Genom Med. 2023;8(1):39.
Simsek-Kiper PO, Jacob P, Upadhyai P, Taşkıran ZE, Guleria VS, Karaosmanoglu B, et al. Biallelic loss-of-function variants in EXOC6B are associated with impaired primary ciliogenesis and cause spondylo-epi-metaphyseal dysplasia with joint laxity type 3. Hum Mutat. 2022;43(12):2116–29.
Girisha KM, Jacob P, SriLakshmi Bhavani G, Shah H, Mortier GR. Steel syndrome: Report of three patients, including monozygotic twins and review of clinical and mutation profiles. Eur J Med Genet. 2022;65(6):104521.
Upadhyai P, Radhakrishnan P, Guleria VS, Kausthubham N, Nayak SS, Superti-Furga A, et al. Biallelic deep intronic variant c.5457+81T>A in TRIP11 causes loss of function and results in achondrogenesis 1A. Hum Mutat. 2021;42(8):1005–14.
Narayanan DL, Shukla A, Kausthubham N, Bhavani GS, Shah H, Mortier G, et al. An emerging ribosomopathy affecting the skeleton due to biallelic variations in NEPRO. Am J Med Genet A. 2019;179(9):1709–17.
Salian S, Cho TJ, Phadke SR, Gowrishankar K, Bhavani GS, Shukla A, et al. Additional three patients with Smith-McCort dysplasia due to novel RAB33B mutations. Am J Med Genet A. 2017;173(3):588–95.
Moirangthem A, Narayanan DL, Jacob P, Nishimura G, Mortier G, Girisha KM. Report of second case and clinical and molecular characterization of Eiken syndrome. Clin Genet. 2018;94(5):457–60.
Jacob P, Soni JP, Mortier G, Girisha KM. The third family with Eiken syndrome. Clin Genet. 2019;96(4):378–9.
Li S, Lyu S, Fu W, Hu Y, Yue H, Chen L, et al. Clinical and genetic profiles of 985 Chinese families with skeletal dysplasia. Chin Med J. 2023;136(12):1485–7.
Scocchia A, Kangas-Kontio T, Irving M, Hero M, Saarinen I, Pelttari L, et al. Diagnostic utility of next-generation sequencing-based panel testing in 543 patients with suspected skeletal dysplasia. Orphanet J Rare Dis. 2021;16(1):412.
Kurt-Sukur ED, Simsek-Kiper PO, Utine GE, Boduroglu K, Alanay Y. Experience of a skeletal dysplasia registry in Turkey: a five-years retrospective analysis. Am J Med Genet A. 2015;167A(9):2065–74.
Mortier GR, Cohn DH, Cormier‐Daire V, Hall C, Krakow D, Mundlos S, et al. Nosology and classification of genetic skeletal disorders: 2019 revision. Am J Med Genet. 2019;179(12):2393–419.
Lv S, Zhao J, Xi L, Lin X, Wang C, Yue H, et al. Genetics evaluation of targeted exome sequencing in 223 Chinese probands with genetic skeletal dysplasias. Front Cell Dev Biol. 2021;9:715042.
Bae JS, Kim NKD, Lee C, Kim SC, Lee HR, Song HR, et al. Comprehensive genetic exploration of skeletal dysplasia using targeted exome sequencing. Genet Med. 2016;18(6):563–9.
Zhang H, Yang R, Wang Y, Ye J, Han L, Qiu W, et al. A pilot study of gene testing of genetic bone dysplasia using targeted next-generation sequencing. J Hum Genet. 2015;60(12):769–76.
Retterer K, Juusola J, Cho MT, Vitazka P, Millan F, Gibellini F, et al. Clinical application of whole-exome sequencing across clinical indications. Genet Med. 2016;18(7):696–704.
Silveira KC, Kanazawa TY, Silveira C, Lacarrubba-Flores MDJ, Carvalho BS, Cavalcanti DP. Molecular diagnosis in a cohort of 114 patients with rare skeletal dysplasias. Am J Med Genet C Semin Med Genet. 2021;187(3):396–408.
Hammarsjö A, Pettersson M, Chitayat D, Handa A, Anderlid BM, Bartocci M, et al. High diagnostic yield in skeletal ciliopathies using massively parallel genome sequencing, structural variant screening and RNA analyses. J Hum Genet. 2021;66(10):995–1008.
Maddirevula S, Alsahli S, Alhabeeb L, Patel N, Alzahrani F, Shamseldin HE, et al. Expanding the phenome and variome of skeletal dysplasia. Genet Med. 2018;20(12):1609–16.
Sithambaram S., Jacob P., Neethukrishna K., Bhavani G. S., Dalal A., Shah H., et al. PRKACA-related, atrial defects-polydactyly-multiple congenital malformation syndrome in an Indian patient. Am J Med Genet A. 2024;194:e63566.
Singh S, Jacob P, Patil SJ, Muranjan M, Shah H, Girisha KM, et al. Indian patients with CHST3-related chondrodysplasia with congenital joint dislocations. Am J Med Genet A. 2024;194(3):e63422.
Bhavani GS, Shah H, Shukla A, Gupta N, Gowrishankar K, Rao AP, et al. Clinical and mutation profile of multicentric osteolysis nodulosis and arthropathy. Am J Med Genet Part A. 2016;170(2):410–7.
Jacob P, Bhavani GSL, Shah H, Galada C, Nampoothiri S, Kamath N, et al. Pseudoachondroplasia: phenotype and genotype in 11 Indian patients. Am J Med Genet A. 2022;188(3):751–9.
Bhavani GS, Shah H, Dalal AB, Shukla A, Danda S, Aggarwal S, et al. Novel and recurrent mutations in WISP3 and an atypical phenotype. Am J Med Genet A. 2015;167A(10):2481–4.
Bidchol AM, Dalal A, Shah H, S S, Nampoothiri S, Kabra M, et al. GALNS mutations in Indian patients with mucopolysaccharidosis IVA. Am J Med Genet A. 2014;164A(11):2793–801.
Bittles AH. Endogamy, consanguinity and community genetics. J Genet. 2002;81(3):91–8.
Mrosk J, Bhavani GS, Shah H, Hecht J, Krüger U, Shukla A, et al. Diagnostic strategies and genotype-phenotype correlation in a large Indian cohort of osteogenesis imperfecta. Bone. 2018;110:368–77.
Stephen J, Shukla A, Dalal A, Girisha KM, Shah H, Gupta N, et al. Mutation spectrum of COL1A1 and COL1A2 genes in Indian patients with osteogenesis imperfecta. Am J Med Genet Part A. 2014;164(6):1482–9.
Mamadapur M, Mahadevan S, ArulRajamurugan PS, Gandham S, Singh S. Multicentric Osteolysis Nodulosis and Arthropathy (MONA): A Case Series and Review of the Literature. Mediterr J Rheumatol. 2024;35:486–9.
Acknowledgements
We would like to thank the participants and their families for their consent and participation in the study. We are grateful to all the referring physicians who made this work possible. We thank the “SG10K_Pilot Investigators” for providing the SG10K_Pilot data (EGAD00001005337). The data from the “SG10K_Pilot Study” reported here were obtained from EGA. This manuscript was not prepared in collaboration with the “SG10K_Pilot Study” and does not necessarily reflect the opinions or views of the “SG10K_Pilot Study”. Additionally, we acknowledge that Figure 1 was created using BioRender.
Funding
This work was supported by the following research projects awarded to Katta M Girisha: Department of Biotechnology/Wellcome Trust India Alliance project titled “Centre for Rare Disease Diagnosis, Research and Training” (Reference number: IA/CRC/20/1/600002), Department of Science and Technology, Government of India project entitled ‘Application of Autozygosity Mapping and Exome Sequencing to Identify Genetic Basis of Disorders of Skeletal Development’ (SB/SO/HS/005/2014), and Indian Council of Medical Research project entitled “Clinical and molecular evaluation of inherited arthropathies and multiple vertebral segmentation defects” (Project ID: BMS 54/2/2013). Ashwin Dalal is supported by the Department of Biotechnology, Government of India project entitled ‘Development of Genomic Technologies for Predictive Genetic Health and Forensic Profiling’ (Grant No. BTI/AAQ/01/CDFD-Flagship/2019). Swati Singh is supported by Joint CSIR-UGC NET Junior Research Fellowship awarded by Human Resource Development Group under Council of Scientific and Industrial Research (CSIR), Government of India: (08/028(0002)/2019-EMR-I).
Author information
Authors and Affiliations
Contributions
Conceptualisation: KMG, HS, GSB; Data curation: PJ, SS, GSB; Formal analysis: PJ, SS, GSB, KMG; Funding acquisition: KMG, AD; Investigation: PJ, SS GSB, GN, GM, HS, KMG; Resources: KG, DLN, SN, SJP, JPS, MM, SK, BD, BSB, VB, SB, AB, MM, SVH, NK., RDS, DS, AS, SRP; Supervision: GSB, HS, KMG; Writing original draft: PJ, SS, KMG; Writing final draft: PJ, SS, GSB, KG, DLN, SN, SJP, JPS, MM, SK, BD, BVB, SB, AB, MM, SVH, NK, RDS, DS, AS, AD, SRP, GN, GM, HS, KMG All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
KMG is the director of Suma Genomics Private Limited and holds shares of the company that has interests in genetic testing.
Ethical approval
We obtained the informed consents from the families for genetic testing, publication of data and clinical photographs. This study has the approvals from the Institutional Ethics Committee at the Kasturba Medical College and Kasturba Hospital, Manipal (IEC:921/2018; IEC:363/2020, IEC: 430/2013, IEC: 570/2015, IEC: 302/2013).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jacob, P., Singh, S., Bhavani, G.S. et al. Genetic and allelic heterogeneity in 248 Indians with skeletal dysplasia. Eur J Hum Genet 33, 607–613 (2025). https://doi.org/10.1038/s41431-024-01776-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-024-01776-8
This article is cited by
-
Editorial Déjà Vu: This time, it’s actually ‘what’s new in EJHG in May 2025?’
European Journal of Human Genetics (2025)