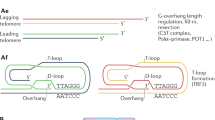
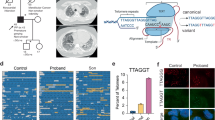
Abstract
Telomere biology disorders (TBDs) are characterized by bone marrow failure (BMF) and dysfunctional telomeres. So far, inherited mutations in 18 genes have been identified in TBDs. Here, we describe a child presenting with early BMF, immunodeficiency, and severely short and defective telomeres, carrying a homozygous splicing mutation (c.409-2 A > G; p.Q136_K138del) in RPA2 – a known replication factor and telomerase accessory factor. The Q136_K138del mutation is predicted to compromise the binding of RPA2 to the single-stranded telomeric DNA. Sequencing of single telomeres revealed that telomere variant repeats (TVRs), present also in healthy individuals, became more prominent in the short telomeres of the patient. Such TVRs may aggravate the telomere dysfunction due to lower affinity to the shelterin proteins. The severe telomere shortening and the activation of DNA damage response at telomeres indicate that this RPA2 mutation causes TBD in a similar manner to other known mutations and should be considered in patients displaying clinical TBD features.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The exome and Sanger sequences (genomic and cDNA) datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
The code used for filtering and processing Nanopore sequencing reads, termed Telomere Analyzer, is available at [Lichtental, D. TelomereAnalyzer. Zenodo (2023). https://doi.org/10.5281/zenodo.8379362].
References
Kam MLW, Nguyen TTT, Ngeow JYY. Telomere biology disorders. NPJ Genom Med. 2021;6:36.
Revy P, Kannengiesser C, Bertuch AA. Genetics of human telomere biology disorders. Nat Rev Genet. 2023; 86–108.
Kvarnung M, Pettersson M, Chun-on P, Rafati M, McReynolds LJ, Norberg A, et al. Identification of biallelic POLA2 variants in two families with an autosomal recessive telomere biology disorder. Eur J Hum Genet. 2024;33:580–7.
Codd V, Wang Q, Allara E, Musicha C, Kaptoge S, Stoma S, et al. Polygenic basis and biomedical consequences of telomere length variation. Nat Genet. 2021;53:1425–33.
Bhat KP, Cortez D. RPA and RAD51: fork reversal, fork protection, and genome stability. Nat Struct Mol Biol. 2018;25:446–53.
Dueva R, Iliakis G. Replication protein A: a multifunctional protein with roles in DNA replication, repair and beyond. NAR Cancer. 2020;2:3.
Agrawal S, Lin X, Susvirkar V, O’Connor MS, Chavez BL, Tholkes VR, et al. Human RPA is an essential telomerase processivity factor for maintaining telomeres. Science. 2025;390:495–502.
Sharma R, Sahoo SS, Honda M, Granger S, Goodings C, Sanchez L, et al. Gain-of-function mutations in RPA1 cause a syndrome with short telomeres and somatic genetic rescue. Blood. 2022;139:1039–51.
Kochman R, Ba I, Yates M, Pirabakaran V, Gourmelon F, Churikov D, et al. Heterozygous RPA2 variant as a novel genetic cause of telomere biology disorders. Genes Dev. 2024;38:755–71.
Abramson J, Adler J, Dunger J, Evans R, Green T, Peitzel A, et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature. 2024;630:493–500.
Cheng TM, Blundell TL, Fernandez-Recio J. pyDock: electrostatics and desolvation for effective scoring of rigid-body protein-protein docking. Proteins. 2007;68:503–15.
Smoom R, May CL, Ortiz V, Tigue M, Kolev HM, Rowe M, et al. Telomouse-a mouse model with human-length telomeres generated by a single amino acid change in RTEL1. Nat Commun. 2023;14:6708.
Stohr BA, Xu L, Blackburn EH. The terminal telomeric DNA sequence determines the mechanism of dysfunctional telomere fusion. Mol Cell. 2012;39:307–14.
Acknowledgements
We thank the patient and his family for their participation in this study. We thank the Jeffrey Modell Foundation, the Israel Ministry of Health, and the Israeli Science Foundation for their support. This work was supported by the Israel Science Foundation grants 3115/19 to R.S. and 1342/23 to Y.T.
Funding
This work was supported by the Jeffrey Modell Foundation, the Israel Ministry of Health, and the Israeli Science Foundation grants 3115/19 and 1342/23 to Y.T.
Author information
Authors and Affiliations
Contributions
Study conceptualization and design, analysis and interpretation of data, and manuscript writing: AJS, YT, and RS; Computational structure modeling and writing original draft: FB, ON, RK; Telomere analysis by NanoTelSeq and FISH, and writing original draft: MNB; Telomere analysis by in-gel hybridization: RS; Sanger sequencing and Western blot analysis: AL, SL; WES and bioinformatics analysis: OB; Patient diagnosis and treatment: AM, MR, EA; Pathology assessment: SN.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
A signed informed consent was obtained from the patient’s parents in accordance with the ethical standards and approved by our institutional ethics committee at Sheba Medical Center (reference number 8842-11-SMC). Clinical data were collected and reviewed from digital hospital-based data.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Simon, A.J., Neustadter-Blackman, M., Lev, A. et al. A homozygote mutation in RPA2 associated with bone marrow failure, immunodeficiency, and telomere biology disorder. Eur J Hum Genet 34, 732–736 (2026). https://doi.org/10.1038/s41431-026-02035-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41431-026-02035-8