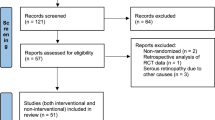
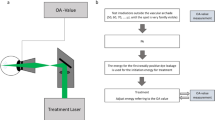
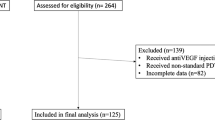
Abstract
Central serous chorioretinopathy is one of the most frequent causes of vision reduction among middle-aged men. This disease usually has a self-limiting course, but sometimes it lasts more than 4–6 months or a second episode follows a complete resolution of the first one. Nevertheless, to date no consensus exists about the duration threshold and therapy protocols for these non-resolving central serous chorioretinopathy. Treatment as half-dose and half-fluence photodynamic therapy, subthreshold micropulse laser treatment, mineralocorticoid receptor antagonists, intravitreal anti-angiogenic drugs, transpupillary thermal therapy, anti-androgenic drugs, methotrexate, Rifampicin and melatonin are described in this review. Complications are very uncommon but end-point results like central macular thickness reduction and best-corrected visual acuity improvement are difficult to compare among different therapeutic modalities due to different duration of follow-up and lack of homogeneity in patient recruitment. The aim of this review is focusing on treatment modalities for these chronic forms with comprehensive recent management updates according to latest clinical trial results.
摘要
中心性浆液性脉络膜视网膜病变是导致中年男性视力下降最常见的原因之一。这种疾病通常具有自限性, 但有时它会持续4-6个月以上, 或在第一次治愈后复发。然而, 迄今为止, 人们对于这种难治性中心性浆液性脉络膜视网膜病变的时间阈值和治疗方案尚未达成共识。本文综述了半剂量、半效应光动力疗法、阈下微脉冲激光光凝治疗、盐皮质激素受体阻断剂、玻璃体内注入抗新生血管药物、经瞳孔温热疗法、抗雄激素药物、甲氨蝶呤、利福平、褪黑激素等治疗方法的疗效。以上几种治疗方法的并发症少见, 但由于随访时间不同和招募的患者缺乏同质性, 导致难以比较不同治疗方法下患者的黄斑中心凹厚度的减少和最佳矫正视力的提高等疗效。本综述的目的是根据最新的临床试验结果, 对这种慢性疾病的治疗方案的管理进行全面的更新。
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Daruich A, Matet A, Dirani A, Bousquet E, Zhao M, Farman N, et al. Central serous chorioretinopathy: recent findings and new physiopathology hypothesis. Prog Retin Eye Res. 2015;48:82–118.
Wang M, Munch IC, Hasler PW, Prunte C, Larsen M. Central serous chorioretinopathy. Acta Ophthalmol. 2008;86:126–45.
Kitzmann AS, Pulido JS, Diehl NN, Hodge DO, Burke JP. The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980-2002. Ophthalmology. 2008;115:169–73.
Tsai DC, Chen SJ, Huang CC, Chou P, Chung CM, Huang PH, et al. Epidemiology of idiopathic central serous chorioretinopathy in Taiwan, 2001-2006: a population-based study. PLoS ONE. 2013;8:e66858.
Chan WM, Lai TY, Tano Y, Liu DT, Li KK, Lam DS. Photodynamic therapy in macular diseases of asian populations: when East meets West. Jpn J Ophthalmol. 2006;50:161–9.
Lehmann M, Bousquet E, Beydoun T, Behar-Cohen F. PACHYCHOROID: an inherited condition? Retina. 2015;35:10–6.
Weenink AC, Borsje RA, Oosterhuis JA. Familial chronic central serous chorioretinopathy. Ophthalmologica. 2001;215:183–7.
Gilbert CM, Owens SL, Smith PD, Fine SL. Long-term follow-up of central serous chorioretinopathy. Br J Ophthalmol. 1984;68:815–20.
Gemenetzi M, De Salvo G, Lotery AJ. Central serous chorioretinopathy: an update on pathogenesis and treatment. Eye (Lond). 2010;24:1743–56.
Ross A, Ross AH, Mohamed Q. Review and update of central serous chorioretinopathy. Curr Opin Ophthalmol. 2011;22:166–73.
Miki A, Kondo N, Yanagisawa S, Bessho H, Honda S, Negi A. Common variants in the complement factor H gene confer genetic susceptibility to central serous chorioretinopathy. Ophthalmology. 2014;121:1067–72.
Schubert C, Pryds A, Zeng S, Xie Y, Freund KB, Spaide RF, et al. Cadherin 5 is regulated by corticosteroids and associated with central serous chorioretinopathy. Hum Mutat. 2014;35:859–67.
Tsai DC, Huang CC, Chen SJ, Chou P, Chung CM, Chan WL, et al. Central serous chorioretinopathy and risk of ischaemic stroke: a population-based cohort study. Br J Ophthalmol. 2012;96:1484–8.
Carvalho-Recchia CA, Yannuzzi LA, Negrao S, Spaide RF, Freund KB, Rodriguez-Coleman H, et al. Corticosteroids and central serous chorioretinopathy. Ophthalmology. 2002;109:1834–7.
Yannuzzi LA. Type A behavior and central serous chorioretinopathy. Retina. 2012;32(Suppl 1):709.
Mansuetta CC, Mason JO 3rd, Swanner J, Feist RM, White MF Jr., Thomley ML, et al. An association between central serous chorioretinopathy and gastroesophageal reflux disease. Am J Ophthalmol. 2004;137:1096–100.
Bousquet E, Dhundass M, Lehmann M, Rothschild PR, Bayon V, Leger D, et al. Shift work: a risk factor for central serous chorioretinopathy. Am J Ophthalmol. 2016;165:23–8.
Matet A, Daruich A, Zola M, Behar-Cohen F. Risk factors for recurrences of central serous chorioretinopathy. Retina. 2017;38:1403–14.
Brodie FL, Charlson ES, Aleman TS, Salvo RT, Gewaily DY, Lau MK, et al. Obstructive sleep apnea and central serous chorioretinopathy. Retina. 2015;35:238–43.
Cheung CMG, Lee WK, Koizumi H, Dansingani K, Lai TYY, Freund KB. Pachychoroid disease. Eye (Lond). 2019;33:18–33.
Warrow DJ, Hoang QV, Freund KB. Pachychoroid pigment epitheliopathy. Retina. 2013;33:1659–72.
Pang CE, Freund KB. Pachychoroid pigment epitheliopathy may masquerade as acute retinal pigment epitheliitis. Invest Ophthalmol Vis Sci. 2014;55:5252.
Margolis R, Mukkamala SK, Jampol LM, Spaide RF, Ober MD, Sorenson JA, et al. The expanded spectrum of focal choroidal excavation. Arch Ophthalmol. 2011;129:1320–5.
Balaratnasingam C, Lee WK, Koizumi H, Dansingani K, Inoue M, Freund KB. Polypoidal choroidal vasculopathy: a distinct disease or manifestation of many? Retina. 2016;36:1–8.
Spitznas M, Huke J. Number, shape, and topography of leakage points in acute type I central serous retinopathy. Graefes Arch Clin Exp Ophthalmol. 1987;225:437–40.
Yang L, Jonas JB, Wei W. Optical coherence tomography-assisted enhanced depth imaging of central serous chorioretinopathy. Invest Ophthalmol Vis Sci. 2013;54:4659–65.
Piccolino FC, De La Longrais RR, Manea M, Cicinelli S. Posterior cystoid retinal degeneration in central serous chorioretinopathy. Retina. 2008;28:1008–12.
Lim Z, Wong D. Retinal pigment epithelial rip associated with idiopathic central serous chorioretinopathy. Eye (Lond). 2008;22:471–3.
Ojima Y, Hangai M, Sasahara M, Gotoh N, Inoue R, Yasuno Y, et al. Three-dimensional imaging of the foveal photoreceptor layer in central serous chorioretinopathy using high-speed optical coherence tomography. Ophthalmology. 2007;114:2197–207.
Quaranta-El Maftouhi M, El Maftouhi A, Eandi CM. Chronic central serous chorioretinopathy imaged by optical coherence tomographic angiography. Am J Ophthalmol. 2015;160:581–7. e1
Framme C, Walter A, Gabler B, Roider J, Sachs HG, Gabel VP. Fundus autofluorescence in acute and chronic-recurrent central serous chorioretinopathy. Acta Ophthalmol Scand. 2005;83:161–7.
Zola M, Chatziralli I, Menon D, Schwartz R, Hykin P, Sivaprasad S. Evolution of fundus autofluorescence patterns over time in patients with chronic central serous chorioretinopathy. Acta Ophthalmol. 2018;96:e835–39.
Eandi CM, Piccolino FC, Alovisi C, Tridico F, Giacomello D, Grignolo FM. Correlation between fundus autofluorescence and central visual function in chronic central serous chorioretinopathy. Am J Ophthalmol. 2015;159:652–8.
Yang L, Jonas JB, Wei W. Choroidal vessel diameter in central serous chorioretinopathy. Acta Ophthalmol. 2013;91:e358–62.
Chan WM, Lai TY, Lai RY, Tang EW, Liu DT, Lam DS. Safety enhanced photodynamic therapy for chronic central serous chorioretinopathy: one-year results of a prospective study. Retina. 2008;28:85–93.
Valmaggia C, Haueter I, Niederberger H. Photodynamic therapy in the treatment of persistent central serous chorioretinopathy: a two-year follow-up. Klin Monbl Augenheilkd. 2012;229:323–6.
Lim JI, Glassman AR, Aiello LP, Chakravarthy U, Flaxel CJ, Spaide RF, et al. Collaborative retrospective macula society study of photodynamic therapy for chronic central serous chorioretinopathy. Ophthalmology. 2014;121:1073–8.
Ruiz-Del-Tiempo MP, Calvo P, Ferreras A, Lecinena J, Pablo L, Ruiz-Moreno O. Anatomical retinal changes after photodynamic therapy in chronic central serous chorioretinopathy. J Ophthalmol. 2018;2018:4081874.
Iacono P, Tedeschi M, Boccassini B, Chiaravalloti A, Varano M, Parravano M. Chronic central serous chorioretinopathy: early and late morphological and functional changes after verteporfin photodynamic therapy. Retina. 2018. https://doi.org/10.1097/IAE.0000000000002040.
Inoue R, Sawa M, Tsujikawa M, Gomi F. Association between the efficacy of photodynamic therapy and indocyanine green angiography findings for central serous chorioretinopathy. Am J Ophthalmol. 2010;149:441–6. e1-2
Rouvas A, Stavrakas P, Theodossiadis PG, Stamatiou P, Milia M, Giannakaki E, et al. Long-term results of half-fluence photodynamic therapy for chronic central serous chorioretinopathy. Eur J Ophthalmol. 2012;22:417–22.
Quin G, Liew G, Ho IV, Gillies M, Fraser-Bell S. Diagnosis and interventions for central serous chorioretinopathy: review and update. Clin Exp Ophthalmol. 2013;41:187–200.
Ma J, Meng N, Xu X, Zhou F, Qu Y. System review and meta-analysis on photodynamic therapy in central serous chorioretinopathy. Acta Ophthalmol. 2014;92:e594–601.
Hwang S, Kang SW, Kim SJ, Jang JW, Kim KT. Photodynamic therapy for symptomatic subfoveal retinal pigment epithelial detachment in central serous chorioretinopathy: outcomes and prognostic factors. Retina. 2018. https://doi.org/10.1097/IAE.0000000000002108.
Maruko I, Iida T, Sugano Y, Ojima A, Ogasawara M, Spaide RF. Subfoveal choroidal thickness after treatment of central serous chorioretinopathy. Ophthalmology. 2010;117:1792–9.
Izumi T, Koizumi H, Maruko I, Takahashi Y, Sonoda S, Sakamoto T, et al. Structural analyses of choroid after half-dose verteporfin photodynamic therapy for central serous chorioretinopathy. Br J Ophthalmol. 2017;101:433–7.
Erikitola OC, Crosby-Nwaobi R, Lotery AJ, Sivaprasad S. Photodynamic therapy for central serous chorioretinopathy. Eye (Lond). 2014;28:944–57.
Cheng CK, Chang CK, Peng CH. Comparison of photodynamic therapy using half-dose of verteporfin or half-fluence of laser light for the treatment of chronic central serous chorioretinopathy. Retina. 2017;37:325–33.
Nicolo M, Eandi CM, Alovisi C, Grignolo FM, Traverso CE, Musetti D, et al. Half-fluence versus half-dose photodynamic therapy in chronic central serous chorioretinopathy. Am J Ophthalmol. 2014;157:1033–7.
Lanzetta P, Furlan F, Morgante L, Veritti D, Bandello F. Nonvisible subthreshold micropulse diode laser (810 nm) treatment of central serous chorioretinopathy. A pilot study. Eur J Ophthalmol. 2008;18:934–40.
Khosla PK, Rana SS, Tewari HK, Azad RU, Talwar D. Evaluation of visual function following argon laser photocoagulation in central serous retinopathy. Ophthalmic Surg Lasers. 1997;28:693–7.
Loo RH, Scott IU, Flynn HW Jr, Gass JD, Murray TG, Lewis ML, et al. Factors associated with reduced visual acuity during long-term follow-up of patients with idiopathic central serous chorioretinopathy. Retina. 2002;22:19–24.
Lim JW, Kang SW, Kim YT, Chung SE, Lee SW. Comparative study of patients with central serous chorioretinopathy undergoing focal laser photocoagulation or photodynamic therapy. Br J Ophthalmol. 2011;95:514–7.
Lanzetta P, Dorin G, Pirracchio A, Bandello F. Theoretical bases of non-ophthalmoscopically visible endpoint photocoagulation. Semin Ophthalmol. 2001;16:8–11.
Maruko I, Koizumi H, Hasegawa T, Arakawa H, Iida T. Subthreshold 577 nm micropulse laser treatment for central serous chorioretinopathy. PLoS ONE. 2017;12:e0184112.
Scholz P, Altay L, Fauser S. A review of subthreshold micropulse laser for treatment of macular disorders. Adv Ther. 2017;34:1528–55.
Wood EH, Karth PA, Sanislo SR, Moshfeghi DM, Palanker DV. Nondamaging retinal laser therapy for treatment of central serous chorioretinopathy: what is the evidence? Retina. 2017;37:1021–33.
Mainster MA. Wavelength selection in macular photocoagulation. Tissue optics, thermal effects, and laser systems. Ophthalmology. 1986;93:952–8.
Arsan A, Kanar HS, Sonmez A. Visual outcomes and anatomic changes after sub-threshold micropulse yellow laser (577-nm) treatment for chronic central serous chorioretinopathy: long-term follow-up. Eye (Lond). 2018;32:726–33.
Ntomoka CG, Rajesh B, Muriithi GM, Goud A, Chhablani J. Comparison of photodynamic therapy and navigated microsecond laser for chronic central serous chorioretinopathy. Eye (Lond). 2018;32:1079–86.
van Dijk EHC, Fauser S, Breukink MB, Blanco-Garavito R, Groenewoud JMM, Keunen JEE, et al. Half-dose photodynamic therapy versus high-density subthreshold micropulse laser treatment in patients with chronic central serous chorioretinopathy: the PLACE trial. Ophthalmology. 2018;125:1547–55.
Roca JA, Wu L, Fromow-Guerra J, Rodriguez FJ, Berrocal MH, Rojas S, et al. Yellow (577 nm) micropulse laser versus half-dose verteporfin photodynamic therapy in eyes with chronic central serous chorioretinopathy: results of the Pan-American Collaborative Retina Study (PACORES) Group. Br J Ophthalmol. 2018;102:1696–1700.
Golestaneh N, Picaud S, Mirshahi M. The mineralocorticoid receptor in rodent retina: ontogeny and molecular identity. Mol Vis. 2002;8:221–5.
Wilkinson-Berka JL, Tan G, Jaworski K, Miller AG. Identification of a retinal aldosterone system and the protective effects of mineralocorticoid receptor antagonism on retinal vascular pathology. Circ Res. 2009;104:124–33.
Zhao M, Bousquet E, Valamanesh F, Farman N, Jeanny JC, Jaisser F, et al. Differential regulations of AQP4 and Kir4.1 by triamcinolone acetonide and dexamethasone in the healthy and inflamed retina. Invest Ophthalmol Vis Sci. 2011;52:6340–7.
Zhao M, Celerier I, Bousquet E, Jeanny JC, Jonet L, Savoldelli M, et al. Mineralocorticoid receptor is involved in rat and human ocular chorioretinopathy. J Clin Investig. 2012;122:2672–9.
van Dijk EH, Nijhoff MF, de Jong EK, Meijer OC, de Vries AP, Boon CJ. Central serous chorioretinopathy in primary hyperaldosteronism. Graefes Arch Clin Exp Ophthalmol. 2016;254:2033–42.
Ghadiali Q, Jung JJ, Yu S, Patel SN, Yannuzzi LA. Central serous chorioretinopathy treated with mineralocorticoid antagonists: a one-year pilot study. Retina. 2016;36:611–8.
Cakir B, Fischer F, Ehlken C, Buhler A, Stahl A, Schlunck G, et al. Clinical experience with eplerenone to treat chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2016;254:2151–7.
Daruich A, Matet A, Dirani A, Gallice M, Nicholson L, Sivaprasad S, et al. Oral mineralocorticoid-receptor antagonists: real-life experience in clinical subtypes of nonresolving central serous chorioretinopathy with chronic epitheliopathy. Transl Vis Sci Technol. 2016;5:2.
Schwartz R, Habot-Wilner Z, Martinez MR, Nutman A, Goldenberg D, Cohen S, et al. Eplerenone for chronic central serous chorioretinopathy-a randomized controlled prospective study. Acta Ophthalmol. 2017;95:e610–e8.
Rahimy E, Pitcher JD 3rd, Hsu J, Adam MK, Shahlaee A, Samara WA, et al. A randomized double-blind placebo-control pilot study of eplerenone for the treatment of central serous chorioretinopathy (Ecselsior). Retina. 2018;38:962–9.
van Dijk EHC, Schellevis RL, van Bergen M, Breukink MB, Altay L, Scholz P, et al. Association of a haplotype in the NR3C2 gene, encoding the mineralocorticoid receptor, with chronic central serous chorioretinopathy. JAMA Ophthalmol. 2017;135:446–51.
Shin MC, Lim JW. Concentration of cytokines in the aqueous humor of patients with central serous chorioretinopathy. Retina. 2011;31:1937–43.
Lim JW, Kim MU, Shin MC. Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina. 2010;30:1465–71.
Ji S, Wei Y, Chen J, Tang S. Clinical efficacy of anti-VEGF medications for central serous chorioretinopathy: a meta-analysis. Int J Clin Pharm. 2017;39:514–21.
Chan WM, Lai TY, Liu DT, Lam DS. Intravitreal bevacizumab (avastin) for choroidal neovascularization secondary to central serous chorioretinopathy, secondary to punctate inner choroidopathy, or of idiopathic origin. Am J Ophthalmol. 2007;143:977–83.
Bandello F, Virgili G, Lanzetta P, Pirracchio A, Menchini U. [ICG angiography and retinal pigment epithelial decompensation (CRSC and epitheliopathy)]. J Fr Ophtalmol. 2001;24:448–51.
Fung AT, Yannuzzi LA, Freund KB. Type 1 (sub-retinal pigment epithelial) neovascularization in central serous chorioretinopathy masquerading as neovascular age-related macular degeneration. Retina. 2012;32:1829–37.
Peiretti E, Caminiti G, Serra R, Querques L, Pertile R, Querques G. Anti-Vegf therapy versus photodynamic therapy in the treatment of choroidal neovascularization secondary to central serous chorioretinopathy. Retina. 2017;3:1526–32.
Shukla D, Kolluru C, Vignesh TP, Karthikprakash S, Kim R. Transpupillary thermotherapy for subfoveal leaks in central serous chorioretinopathy. Eye (Lond). 2008;22:100–6.
Hussain N, Khanna R, Hussain A, Das T. Transpupillary thermotherapy for chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2006;244:1045–51.
Mathur V, Parihar J, Maggon R, Mishra SK. Role of transpupillary thermotherapy in central serous chorio-retinopathy. Med J Armed Forces India. 2009;65:323–7.
Manayath GJ, Narendran V, Arora S, Morris RJ, Saravanan VR, Shah PK. Graded subthreshold transpupillary thermotherapy for chronic central serous chorioretinopathy. Ophthalmic Surg Lasers Imaging. 2012;43:284–90.
Manayath GJ, Karandikar SS, Narendran S, Kumarswamy KA, Saravanan VR, Morris RJ, et al. Low fluence photodynamic therapy versus graded subthreshold transpupillary thermotherapy for chronic central serous chorioretinopathy: results from a prospective study. Ophthalmic Surg Lasers Imaging Retina. 2017;48:334–8.
Nudleman E, Witmer MT, Kiss S, Williams GA, Wolfe JD. Central serous chorioretinopathy in patients receiving exogenous testosterone therapy. Retina. 2014;34:2128–32.
Grieshaber MC, Staub JJ, Flammer J. The potential role of testosterone in central serous chorioretinopathy. Br J Ophthalmol. 2007;91:118–9.
Ahad MA, Chua CN, Evans NM. Central serous chorioretinopathy associated with testosterone therapy. Eye (Lond). 2006;20:503–5.
Grino PB, Griffin JE, Wilson JD. Testosterone at high concentrations interacts with the human androgen receptor similarly to dihydrotestosterone. Endocrinology. 1990;126:1165–72.
Forooghian F, Meleth AD, Cukras C, Chew EY, Wong WT, Meyerle CB. Finasteride for chronic central serous chorioretinopathy. Retina. 2011;31:766–71.
Moisseiev E, Holmes AJ, Moshiri A, Morse LS. Finasteride is effective for the treatment of central serous chorioretinopathy. Eye (Lond). 2016;30:850–6.
Kerbel RS, Kamen BA. The anti-angiogenic basis of metronomic chemotherapy. Nat Rev Cancer. 2004;4:423–36.
Munoz R, Shaked Y, Bertolini F, Emmenegger U, Man S, Kerbel RS. Anti-angiogenic treatment of breast cancer using metronomic low-dose chemotherapy. Breast. 2005;14:466–79.
Kurup SK, Oliver A, Emanuelli A, Hau V, Callanan D. Low-dose methotrexate for the treatment of chronic central serous chorioretinopathy: a retrospective analysis. Retina. 2012;32:2096–101.
Shichiri M, Fukai N, Kono Y, Tanaka Y. Rifampicin as an oral angiogenesis inhibitor targeting hepatic cancers. Cancer Res. 2009;69:4760–8.
Guengerich FP. Cytochrome P-450 3A4: regulation and role in drug metabolism. Annu Rev Pharmacol Toxicol. 1999;39:1–17.
Shulman S, Goldenberg D, Schwartz R, Habot-Wilner Z, Barak A, Ehrlich N, et al. Oral Rifampin treatment for longstanding chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2016;254:15–22.
Pandi-Perumal SR, Trakht I, Spence DW, Srinivasan V, Dagan Y, Cardinali DP. The roles of melatonin and light in the pathophysiology and treatment of circadian rhythm sleep disorders. Nat Clin Pract Neurol. 2008;4:436–47.
Lissoni P, Rovelli F, Malugani F, Bucovec R, Conti A, Maestroni GJ. Anti-angiogenic activity of melatonin in advanced cancer patients. Neuro Endocrinol Lett. 2001;22:45–7.
Siu AW, Maldonado M, Sanchez-Hidalgo M, Tan DX, Reiter RJ. Protective effects of melatonin in experimental free radical-related ocular diseases. J Pineal Res. 2006;40:101–9.
Esposito E, Cuzzocrea S. Antiinflammatory activity of melatonin in central nervous system. Curr Neuropharmacol. 2010;8:228–42.
Shi D, Xiao X, Wang J, Liu L, Chen W, Fu L, et al. Melatonin suppresses proinflammatory mediators in lipopolysaccharide-stimulated CRL1999 cells via targeting MAPK, NF-kappaB, c/EBPbeta, and p300 signaling. J Pineal Res. 2012;53:154–65.
Gramajo AL, Marquez GE, Torres VE, Juarez CP, Rosenstein RE, Luna JD, et al. Therapeutic benefit of melatonin in refractory central serous chorioretinopathy. Eye (Lond). 2015;29:1036–45.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sartini, F., Figus, M., Nardi, M. et al. Non-resolving, recurrent and chronic central serous chorioretinopathy: available treatment options. Eye 33, 1035–1043 (2019). https://doi.org/10.1038/s41433-019-0381-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-019-0381-7
This article is cited by
-
Gender differences in central serous chorioretinopathy based on the new multimodal imaging classification
Eye (2024)
-
Importance of OCT-derived biomarkers for the recurrence of central serous chorioretinopathy using statistics and predictive modelling
Scientific Reports (2024)
-
Visual and anatomical evaluation of navigated subthreshold micropulse laser versus photodynamic therapy in managing chronic central serous chorioretinopathy
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)
-
Longitudinal follow-up and outcome analysis in central serous chorioretinopathy
Eye (2023)
-
Ten-year follow-up and sequential evaluation of multifocal retinal pigment epithelium abnormalities in central serous chorioretinopathy
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)