Abstract
Purpose
To investigate the diagnostic values of intereye or intraeye asymmetry of retinal perfused vessel density and neural structure parameters for detection of glaucoma.
Methods
In total, 152 healthy subjects and 72 bilateral primary open-angle glaucoma (POAG) patients were enrolled. Total POAG group contains all glaucoma patients. Early to moderate POAG group contains patients whose binocular mean defect values were larger than −12 dB. The retinal perfused vessel densities were acquired using optic coherence tomography angiography. The neural structure parameters include RNFL, GCC thickness and its derivative indices like focal loss volume percentage (FLV%) and global loss volume percentage (GLV%). Intereye asymmetry equaled to the absolute difference of parameters between paired eyes. Intraeye asymmetry was defined as absolute difference between the inferior and superior hemisphere values from one random selected eye. The areas under the receiver operating characteristic curves (AUROCs) were calculated to evaluate diagnostic ability.
Results
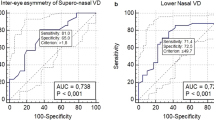
From pairwise comparison analysis of ROC curves, the intereye asymmetric parameters with the largest diagnostic accuracy were FLV and GLV% (AUROC = 0.944), which were significantly superior to the intereye asymmetry of perfused vessel density in peripapillary area and parafoveal area (P < 0.05). Particularly, the intereye asymmetry of FLV% (AUROC = 0.926) and GLV% (AUROC = 0.950) showed excellent diagnostic precision for detecting early to moderate glaucoma patients. However, the intraeye asymmetry of microvascular parameters and neural structure parameters showed fair diagnostic ability for identifying POAG patients.
Conclusions
The intereye asymmetry of neural structure parameters, particularly the FLV% and GLV%, outperformed the microvascular parameters for identifying POAG patients.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Weinreb RN, Khaw PT. Primary open-angle glaucoma. Lancet Lond Engl. 2004;363:1711–20. https://doi.org/10.1016/S0140-6736(04)16257-0
Poinoosawmy D. Frequency of asymmetric visual field defects in normal-tension and high-tension glaucoma. Ophthalmology. 1998;105:988–91. https://doi.org/10.1016/S0161-6420(98)96049-3
Williams AL, Gatla S, Leiby BE, Fahmy I, Biswas A, De Barros DM, et al. The value of intraocular pressure asymmetry in diagnosing glaucoma. J Glaucoma. 2013;22:215–8. https://doi.org/10.1097/IJG.0b013e318237bfb8
Miglior S, Torri V, Zeyen T, Pfeiffer N, Vaz JC, Adamsons I, et al. Intercurrent factors associated with the development of open-angle glaucoma in the European glaucoma prevention study. Am J Ophthalmol. 2007;144:266–75. https://doi.org/10.1016/j.ajo.2007.04.040
Li H, Healey PR, Tariq YM, Teber E, Mitchell P. Symmetry of optic nerve head parameters measured by the heidelberg retina tomograph 3 in healthy eyes: the Blue Mountains Eye study. Am J Ophthalmol. 2013;155:518–23.e1. https://doi.org/10.1016/j.ajo.2012.09.019
Fansi AaK, Boisjoly H, Chagnon M, Harasymowycz P.J. Comparison of different methods of inter-eye asymmetry of rim area and disc area analysis. Eye Lond Engl. 2011;25:1590–7. https://doi.org/10.1038/eye.2011.217
Iester M, Telani S, Frezzotti P, Vittone P, Traverso CE, Calabria G, et al. Differences in central corneal thickness between the paired eyes and the severity of the glaucomatous damage. Eye Lond Engl. 2012;26:1424–30. https://doi.org/10.1038/eye.2012.179
Sullivan-Mee M, Gentry JM, Qualls C. Relationship between asymmetric central corneal thickness and glaucomatous visual field loss within the same patient. Optom Vis Sci Publ Am Acad Optom. 2006;83:516–9. https://doi.org/10.1097/01.opx.0000218433.49803.e7
Sullivan-Mee M, Ruegg CC, Pensyl D, Halverson K, Qualls C, et al. Diagnostic precision of retinal nerve fiber layer and macular thickness asymmetry parameters for identifying early primary open-angle glaucoma. Am J Ophthalmol. 2013;156:567–77.e1. https://doi.org/10.1016/j.ajo.2013.04.037
Mwanza J-C, Durbin MK, Budenz DL. Interocular symmetry in peripapillary retinal nerve fiber layer thickness measured with the cirrus HD-OCT in healthy eyes. Am J Ophthalmol. 2011;151:514–21.e1. https://doi.org/10.1016/j.ajo.2010.09.015
Lee S-Y, Lee EK, Park KH, Kim DM, Jeoung JW, et al. Asymmetry analysis of macular inner retinal layers for glaucoma diagnosis: Swept-source optical coherence tomography study. PLoS ONE. 2016;11:e0164866. https://doi.org/10.1371/journal.pone.0164866
Levine RA, Demirel S, Fan J, Keltner JL, Johnson CA, Kass MA. Asymmetries and visual field summaries as predictors of glaucoma in the ocular hypertension treatment study. Invest Ophthalmol Vis Sci. 2006;47:3896–903. https://doi.org/10.1167/iovs.05-0469
Um TW, Sung KR, Wollstein G, Yun SC, Na JH, Schuman JS. Asymmetry in hemifield macular thickness as an early indicator of glaucomatous change. Invest Ophthalmol Vis Sci. 2012;53:1139–44. https://doi.org/10.1167/iovs.11-8373
Yamada H, Hangai M, Nakano N, Takayama K, Miyake M, et al. Asymmetry analysis of macular inner retinal layers for glaucoma diagnosis. Am J Ophthalmol. 2014;158:1318–29.e3. https://doi.org/10.1016/j.ajo.2014.08.040
Asman P, Heijl A. Evaluation of methods for automated Hemifield analysis in perimetry. Arch Ophthalmol Chic Ill 1960. 1992;110:820–6.
Asman P, Heijl A. Glaucoma Hemifield test. Automated visual field evaluation. Arch Ophthalmol Chic Ill 1960. 1992;110:812–9.
Jia Y, Morrison JC, Tokayer J, Tan O, Lombardi L, Baumann B, et al. Quantitative OCT angiography of optic nerve head blood flow. Biomed Opt Express. 2012;3:3127–37. https://doi.org/10.1364/BOE.3.003127
Huang D, Jia Y, Gao SS, Lumbroso B, Rispoli M, et al. Optical coherence tomography angiography using the optovue device. Dev Ophthalmol. 2016;56:6–12. https://doi.org/10.1159/000442770
Liu L, Jia Y, Takusagawa HL, Pechauer AD, Edmunds B, Lombardi L, et al. Optical coherence tomography angiography of the peripapillary retina in glaucoma. JAMA Ophthalmol. 2015;133:1045–52. https://doi.org/10.1001/jamaophthalmol.2015.2225
Wang X, Jiang C, Ko T, Kong XM, Yu XB, Wang M, et al. Correlation between optic disc perfusion and glaucomatous severity in patients with open-angle glaucoma: an optical coherence tomography angiography study. Graefes Arch Clin Exp Ophthalmol Albrecht Von Graefes Arch Für Klin Exp Ophthalmol. 2015;253:1557–64. https://doi.org/10.1007/s00417-015-3095-y
Xu H, Yu J, Kong X, Sun XH, Jiang CH. Macular microvasculature alterations in patients with primary open-angle glaucoma: A cross-sectional study. Med (Baltim). 2016;95:e4341. https://doi.org/10.1097/MD.0000000000004341
Rao HL, Pradhan ZS, Weinreb RN, Riyazuddin M, Dasari S, Puttaiah NP, et al. A comparison of the diagnostic ability of vessel density and structural measurements of optical coherence tomography in primary open angle glaucoma. PLoS ONE. 2017;12:e0173930. https://doi.org/10.1371/journal.pone.0173930
Rao HL, Kadambi SV, Weinreb RN, Puttaiah NK, Pradhan ZS, Rao DAS, et al. Diagnostic ability of peripapillary vessel density measurements of optical coherence tomography angiography in primary open-angle and angle-closure glaucoma. Br J Ophthalmol. 2017;101:1066–70. https://doi.org/10.1136/bjophthalmol-2016-309377
Tan O, Chopra V, Lu AT-H, Schuman JS, Ishikawa H, Wollstein G, et al. Detection of macular ganglion cell loss in glaucoma by Fourier-domain optical coherence tomography. Ophthalmology. 2009;116:2305–14. https://doi.org/10.1016/j.ophtha.2009.05.025.
Xu H, Deng G, Jiang C, Kong XM, Yu J, Sun XH, et al. Microcirculatory responses to hyperoxia in macular and peripapillary regions. Invest Ophthalmol Vis Sci. 2016;57:4464–8. https://doi.org/10.1167/iovs.16-19603
Rao HL, Pradhan ZS, Weinreb RN, Reddy HB, Riyazuddin M, Dasari S, et al. Regional comparisons of optical coherence tomography angiography vessel density in primary open-angle glaucoma. Am J Ophthalmol. 2016;171:75–83. https://doi.org/10.1016/j.ajo.2016.08.030
Nakano N, Hangai M, Nakanishi H, Mori S, Nukada M, Kotera Y, et al. Macular ganglion cell layer imaging in preperimetric glaucoma with speckle noise–reduced spectral domain optical coherence tomography. Ophthalmology. 2011;118:2414–26. https://doi.org/10.1016/j.ophtha.2011.06.015
Susanna R, Nicolela MT, Soriano DS, Carvalho C, et al. Automated perimetry: a study of the glaucoma hemifield test for the detection of early glaucomatous visual field loss. J Glaucoma. 1994;3:12–6.
Duggan C, Sommer A, Auer C, Burkhard K, et al. Automated differential threshold perimetry for detecting glaucomatous visual field loss. Am J Ophthalmol. 1985;100:420–3.
Sommer A, Enger C, Witt K. Screening for glaucomatous visual field loss with automated threshold perimetry. Am J Ophthalmol. 1987;103:681–4.
Zong Y, Xu H, Yu J, Jiang CH, Kong XM, He Y, et al. Retinal vascular autoregulation during phase IV of the valsalva maneuver: An optical coherence tomography angiography study in healthy chinese adults. Front Physiol. 2017;8. https://doi.org/10.3389/fphys.2017.00553
Acknowledgements
All of authors would like to appreciate Jianfeng Luo, Fudan University, Shanghai, China, for assistance with the statistical analyses in this study.
Funding
This research was supported by the Surface Project of National Natural Science Foundation of China (Grant No. 81770922, China), the project of Shanghai Municipal Commission of Health and Family Planning (Grant No. 201740204, China) the clinical science and technology innovation project of Shanghai Shenkang Hospital Development Center (SHDC12017X18), the western medicine guidance project of Shanghai Committee of Science and Technology (19411961600), the National Key Research & Development Plan (2017YFC0108200) and the Shanghai Committee of Science and Technology (Grant No.13430710500, China).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Xu, H., Zong, Y., Zhai, R. et al. Intereye and intraeye asymmetry analysis of retinal microvascular and neural structure parameters for diagnosis of primary open-angle glaucoma. Eye 33, 1596–1605 (2019). https://doi.org/10.1038/s41433-019-0451-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-019-0451-x
This article is cited by
-
Refractive associations with corneal biomechanical properties among young adults: a population-based Corvis ST study
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)
-
Effect of biomechanical properties on myopia: a study of new corneal biomechanical parameters
BMC Ophthalmology (2020)