Abstract
Purpose
The purpose of this study is to evaluate tumour vasculature with optical coherence tomography angiography (OCTA) in malignant choroidal melanoma (CM).
Materials and methods
Patients with unilateral CM were included in this cross-sectional observational clinical study. Applying OCTA systems operating at 840-nm wavelengths, eyes with CM were imaged. The primary main outcome measures were OCTA images, qualitative evaluation of macular and tumour vasculature, quantitative vascular density (VD), perfusion density (PD) and the foveal avascular zone disruption.
Results
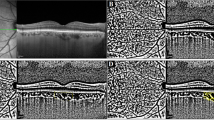
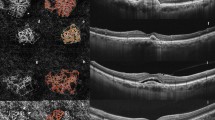
The study included 11 patients with unilateral CM and contralateral unaffected eyes as the control group. Eyes of 11 patients with CM and contralateral unaffected eyes were imaged before brachytherapy and 5 patients were imaged post brachytherapy. CM is demonstrated dense, tortuous blood vessels, uneven thickness and relatively disorganised intratumoural vasculature. In 11 eyes with CM, the VD and PD in the macular area were significantly lower within affected eyes (131.333 ± 27.807%, 3.152 ± 0.714%, p < 0.0001) than in contralateral eyes (154.208 ± 5.599%, 3.662 ± 0.127%, p < 0.0001). The VD and PD in the tumour area (67.990 ± 34.899%, 1.617 ± 0.847%, p < 0.0001) were significantly lower when compared to the macular area of affected eyes (131.333 ± 27.807%, 3.152 ± 0.714%, p < 0.0001) and the macular area of contralateral eyes (154.208 ± 5.599%, 3.662 ± 0.127%, p < 0.0001). After radiation treatment, the VD and PD in five CM eyes (116.526 ± 7.598%, 2.438 ± 0.358%, p < 0.05) were significantly lower than before treatment (141.544 ± 14.645%, 3.327 ± 0.354%, p < 0.05). Tumour regression after radiation therapy for melanomas was associated with decreased vessel density.
Conclusions
OCTA can provide a dye-free, non-invasive, reliable method to monitor a variety of tumours, including CM for growth and vascularity. Upon OCTA, this could be helpful in evaluating the variety of tumour blood vessels before and after brachytherapy to judge the curative effect and whether the tumour recurred. Detection of the characteristic vascular features of CM by OCTA could make OCTA an assuring diagnostic modality to differentiate malignant lesions.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The data of this case report are available from the corresponding author on reasonable request.
References
Jia Y, Bailey ST, Wilson DJ, Tan OU, Klein ML, Flaxel CJ, et al. Quantitative optical coherence tomography angiography of choroidal neovascularization in age-related macular degeneration. Ophthalmology. 2014;121:1435–44.
Jia Y, Bailey ST, Hwang TS, McClintic SM, Gao SS, Pennesi ME, et al. Quantitative optical coherence tomography angiography of vascular abnormalities in the living human eye. Proc Natl Acad Sci USA. 2015;112:E2395–402.
Cennamo, G. OCT angiography examination of choroidal nevi and melanomas. In: Lumbroso B., Huang D., Jia, Y., et al., editors. Clinical OCT angiography atlas. New Delhi: Jaypee Brothers Medical Publishers, Ltd; 2015. p. 150–5.
Ang M, Cai Y, Shahipasand S, Sim DA, Keane PA, Sng CCA, et al. En face optical coherence tomography angiography for corneal neovascularisation. Br J Ophthalmol. 2016;100:616–21.
Wilkes SR, Robertson DM, Kurland LT, Campbell RJ. Incidence of uveal malignant melanoma in the resident population of Rochester and Olmsted County, Minnesota. Am J Ophthalmol. 1979;87:639–41.
Singh AD, Turell ME, Topham AK. Uveal melanoma: trends in incidence, treatment, and survival. Ophthalmology. 2011;118:1881–5.
Mahendraraj K, Lau C, Lee I, Chamberlain R. Trends in incidence, survival, and management of uveal melanoma: a population-based study of 7,516 patients from the Surveillance, Epidemiology, and End Results database (1973–2012). Clin Ophthalmol. 2016;10:2113–9.
Desjardins L, Lumbroso L, Levy C, Mazal A, Delacroix S, Rosenwald JC, et al. Treatment of uveal melanoma with iodine 125 plaques or proton beam radiotherapy: indications and comparison of local recurrence rates. J Fr Ophtalmol. 2003;26:269–76.
Damato B, Kacperek A, Chopra M, Campbell IR, Errington RD. Proton beam radiotherapy of choroidal mela- noma: the Liverpool-Clatterbridge experience. Int J Radiat Oncol Biol Phys. 2005;62:1405–11.
Dendale R, Rouic LL-L, Noel G, Feuvret L, Levy C, Delacroix S, et al. Proton beam radiotherapy for uveal melanoma: results of Curie Institut-Orsay proton therapy center (ICPO). Int J Radiat Oncol Biol Phys. 2006;65:780–7.
Marconi DG, Castro DG, Rebouças LM, Gil GOB, Fogaroli RC, Maia MAC, et al. Tumor control, eye preservation, and visual outcomes of ruthenium plaque brachytherapy for choroidal melanoma. Brachytherapy. 2013;12:235–9.
Spaide RF, Fujimoto JG, Waheed NK. Image aritfats in optical tomography angiography. Retina. 2015;35:2163–80. https://doi.org/10.1097/IAE.0000000000000765.
Lupidi M, Coscas F, Cagini C, Fiore T, Spaccini E, Fruttini D, et al. Automated quantitative analysis of retinal microvasculature in normal eyes on optical coherence tomography angiography. Am J Ophthalmol. 2016;169:9–23.
Lei J, Durbin MK, Shi Y, Uji A, Balasubramanian S, Baghdasaryan E. et al. Repeatability and reproducibility of superficial macular retinal vessel density measurementsusing optical coherence tomography angiography en face images. JAMA Ophthalmol. 2017;135:1092–8. https://doi.org/10.1001/jamaophthalmol.2017.3431.
Vakoc BJ, Fukumura D, Jain RK, Bouma BE. Cancer imaging by optical coherence tomography: preclinical progress and clinical potential. Nat Rev Cancer. 2012;12:363–8.
Chien JL, Sioufi K, Shields CL. Optical coherence tomography angiography of conjunctival racemose hemangioma. Ophthalmology. 2017;124:449.
Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285:1182–6.
Ribatti D. Judah Folkman, a pioneer in the study of angiogenesis. Angiogenesis. 2008;11:3–10.
Nishida N, Yano H, Nishida T, Kamura T, Kojiro M. Angiogenesis in cancer. Vasc Health Risk Manag. 2006;2:213–9.
Weidner N. Intratumor microvessel density as a prognostic factor in cancer. Am J Pathol. 1995;147:9–19.
Shields CL, Kaliki S, Rojanaporn D, Ferenczy SR, Shields JA. Enhanced depth imaging optical coherence tomography of small choroidal melanoma: comparison with choroidal nevus. Arch Ophthalmol. 2012;130:850–6.
Cennamo G, Romano MR, Breve MA, Velotti N, Reibaldi M, de Crecchio G. et al. Evaluation of choroidal tumors with optical coherence tomography: enhanced depth imaging and OCT-angiography features. Eye. 2017;31:906–15. https://doi.org/10.1038/eye.2017.14.
Shields CL, Manalac J, Das C, Ferguson K, Shields JA. Choroidal melanoma: clinical features, classification, and top 10 pseudomelanomas. Curr Opin Ophthalmol. 2014;25:177–85.
Ghassemi F, Mirshahi R, Fadakar K, Sabour S. Optical coherence tomography angiography in choroidal melanoma and nevus. Clin Ophthalmol. 2018;12:207–14. https://doi.org/10.2147/OPTH.S148897.
Ghassemi F, Mirshahi R, Fadakar K, Sabour. S. Optical coherence tomography angiography in choroidal melanoma and nevus. Clin Ophthalmol. 2018;22:207–14. https://doi.org/10.2147/OPTH.S148897.
Toledo JJ, Asencio M, García JR, Morales LA, Tomkinson C, Cajigal C. OCT angiography: imaging of choroidal and retinal tumors. Ophthalmol Retina. 2018;2:613–22. https://doi.org/10.1016/j.oret.2017.10.006.
Valverde-Megías A, Say EAT, Ferenczy SR, Shields. CL. Differential macular features on optical coherence tomography angiography in eyes with choroidal nevus and melanoma. Retina. 2017;37:731–40. https://doi.org/10.1097/IAE.0000000000001233.
Eldaly H, Eldaly Z. Melanocytoma of the optic nerve head, thirty-month follow-up. Semin Ophthalmol. 2015;30:464–9. https://doi.org/10.3109/08820538.2013.874485.
Veverka KK, AbouChehade JE, Iezzi R Jr, Pulido JS. Noninvasive grading of radiation retinopathy: the use of optical coherence tomography angiography. Retina. 2015;35:2400–10.
Matet A, Daruich A, Zografos. L. Radiation maculopathy after proton beam therapy for uveal melanoma: optical coherence tomography angiography alterations influencing visual acuity. Invest Ophthalmol Vis Sci. 2017;58:3851–61.
Skalet AH, Liu L, Binder C, Miller AK, Wang J, Wilson DJ. et al. Quantitative OCT angiography evaluation of peripapillary retinal circulation after plaque brachytherapy. Ophthalmol Retina.2018;2:244–50. https://doi.org/10.1016/j.oret.2017.06.005.
Skalet AH, Liu L, Binder C, Miller AK, Crilly R, Hung AY. et al. Longitudinal detection of radiation-induced peripapillary and macular retinal capillary ischemia using OCT angiography. Ophthalmol Retina . 2020;4:320–6. https://doi.org/10.1016/j.oret.2019.10.001.
Shields CL, Say EA, Samara WA, Khoo CT, Mashayekhi A, Shields JA. Optical coherence tomography angiography of the macula after plaque radiotherapy of choroidal melanoma: comparison of irradiated versus nonirradiated eyes in 65 patients. Retina. 2016;36:1493–505.
Skalet AH, Li Y, Lu CD, Jia Y, Lee BK, Husvogt L. et al. Optical coherence tomography angiography characteristics of iris melanocytic tumors. Ophthalmology. 2017;124:197–204. https://doi.org/10.1016/j.ophtha.2016.10.003.
Acknowledgements
Funding
The National Natural Science Foundation of China (Nr. 81272981), and the Beijing Natural Science Foundation (Nr. 7151003) provided financial support. Funding support was used to reimburse the travel-related expenses of conference attendees (recipient: WW).
Author information
Authors and Affiliations
Contributions
WW: examination of the patient, interpretation of the results and writing the paper; ZN: interpretation of the results and writing/reviewing of the paper; XX: interpretation of the results and writing/reviewing of the paper. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All authors consent to publish the paper.
Ethical approval
The study followed the tenets of the Declaration of Helsinki and its ethical standards of 1964. The study was approved by the Medical Ethics Committee of the Beijing Tongren Hospital.
Informed consent
Written informed consent was obtained from all participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, N., Xu, X. & Wei, W. Optical coherence tomography angiography characteristics of choroidal melanoma. Eye 35, 2514–2523 (2021). https://doi.org/10.1038/s41433-020-01250-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-020-01250-x