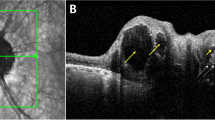
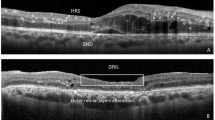
Abstract
Optical coherence tomography (OCT) is a non-invasive medical imaging technology that is playing an increasing role in the routine assessment and management of patients with neuro-ophthalmic conditions. Its ability to characterise the optic nerve head, peripapillary retinal nerve fibre layer and cellular layers of the macula including the ganglion cell layer enables qualitative and quantitative assessment of optic nerve disease. In this review, we discuss technical features of OCT and OCT-based imaging techniques in the neuro-ophthalmic context, potential pitfalls to be aware of, and specific applications in more common neuro-ophthalmic conditions including demyelinating, inflammatory, ischaemic and compressive optic neuropathies, optic disc drusen and raised intracranial pressure. We also review emerging applications of OCT angiography within neuro-ophthalmology.
摘要
相干光断层扫描 (OCT) 是一种非侵入性的医学成像技术, 对于神经眼科疾病患者的常规评估和管理中发挥越来越重的要作用。它可以显示视神经乳头、视乳头周围视网膜的神经纤维层和黄斑细胞层 (包括神经节细胞层) 等, 并可对视神经疾病进行定性和定量评估。在这篇综述中, 我们讨论了OCT和基于OCT的成像技术在神经眼科领域的技术特点、需要注意的潜在缺陷、以及在常见神经眼科疾病中的具体应用, 包括脱髓鞘性、炎症性、缺血性和压迫性视神经病变、视盘玻璃膜疣和颅内压升高等疾病。同时, 我们也回顾了OCT血管造影术在神经眼科方面的新兴应用
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Prasad S, Galetta S. Anatomy and physiology of the afferent visual system. In: Kennard C, Leigh RJ (eds). Handbook of Clinical Neurology, Vol 102. Elsevier: Amsterdam, The Netherlands; 2011, pp 3-19.
Kanamori A, Escano MF, Eno A, et al. Evaluation of the effect of aging on retinal nerve fiber layer thickness measured by optical coherence tomography. Ophthalmologica. 2003;217:273–8.
Putnam NM, Hofer HJ, Doble N, Chen L, Carroll J, Williams DR. The locus of fixation and the foveal cone mosaic. J Vis. 2005;5:632–9.
El-Ashry M, Hegde V, James P, Pagliarini S. Analysis of macular thickness in British population using optical coherence tomography (OCT): An emphasis on interocular symmetry. Curr Eye Res. 2008;33:693–9.
Zeffren BS, Applegate RA, Bradley A, Vanheuven WAJ. Retinal fixation point location in the foveal avascular zone. Invest Ophth Vis Sci. 1990;31:2099–105.
Ctori I, Huntjens B. Repeatability of foveal measurements using spectralis optical coherence tomography segmentation software. Plos ONE. 2015;10:e0129005.
Langenegger SJ, Funk J, Toteberg-Harms M. Reproducibility of retinal nerve fiber layer thickness measurements using the eye tracker and the retest function of spectralis SD-OCT in glaucomatous and healthy control eyes. Invest Ophth Vis Sci. 2011;52:3338–44.
Eriksson U, Alm A. Repeatability in and interchangeability between the macular and the fast macular thickness map protocols: a study on normal eyes with Stratus optical coherence tomography. Acta Ophthalmologica. 2009;87:725–30.
Comyn O, Heng LZ, Ikeji F, Bibi K, Hykin PG, Bainbridge JW, et al. Repeatability of spectralis OCT measurements of macular thickness and volume in diabetic macular edema. Invest Ophth Vis Sci. 2012;53:7754–9.
Gilmore ED, Hudson C. Eccentricity and measurement variability and repeatability with the retinal thickness analyser. Brit J Ophthalmol. 2004;88:62–5.
Parisi V, Manni G, Spadaro M, Colacino G, Restuccia R, Marchi S, et al. Correlation between morphological and functional retinal impairment in multiple sclerosis patients. Invest Ophthalmol Vis Sci. 1999;40:2520–7.
Kupersmith MJ, Mandel G, Anderson S, Meltzer D, Kardon R. Baseline, one and three month changes in the peripapillary retinal nerve fiber layer in acute optic neuritis: relation to baseline vision and MRI. J Neurol Sci. 2011;308:117–23.
Pro MJ, Pons ME, Liebmann JM, Ritch R, Zafar S, Lefton D, et al. Imaging of the optic disc and retinal nerve fiber layer in acute optic neuritis. J Neurol Sco. 2006;250:114–9.
Costello FE, Klistorner A, Kardon R. Optical coherence tomography in the diagnosis and management of optic neuritis and multiple sclerosis. Ophthalmic Surg, Lasers Imaging. 2011;42:S28–40.
Ye C, Lam DS, Leung CK. Investigation of floor effect for OCT RNFL measurement. Invest Ophthalmol Vis Sci. 2011;52:176.
Kupersmith MJ, Garvin MK, Wang JK, Durbin M, Kardon R. Retinal ganglion cell layer thinning within one month of presentation for optic neuritis. Mult Scler. 2016;22:641–8.
Trip SA, Schlottmann PG, Jones SJ, Altmann DR, Garway-Heath DF, Thompson AJ, et al. Retinal nerve fiber layer axonal loss and visual dysfunction in optic neuritis. Ann Neurol. 2005;58:383–91.
Fisher JB, Jacobs DA, Markowitz CE, Galetta SL, Volpe NJ, Nano-Schiavi ML, et al. Relation of visual function to retinal nerve fiber layer thickness in multiple sclerosis. Ophthalmology. 2006;113:324–32.
Walter SD, Ishikawa H, Galetta KM, Sakai RE, Feller DJ, Henderson SB, et al. Ganglion cell loss in relation to visual disability in multiple sclerosis. Ophthalmology. 2012;119:1250–7.
Saidha S, Syc SB, Durbin MK, Eckstein C, Oakley JD, Meyer SA, et al. Visual dysfunction in multiple sclerosis correlates better with optical coherence tomography derived estimates of macular ganglion cell layer thickness than peripapillary retinal nerve fiber layer thickness. Mult Scler. 2011;17:1449–63.
Narayanan D, Cheng H, Bonem KN, Saenz R, Tang RA, Frishman LJ. Tracking changes over time in retinal nerve fiber layer and ganglion cell-inner plexiform layer thickness in multiple sclerosis. Mult Scler. 2014;20(10):1331–41.
Garcia-Martin E, Pueyo V, Martin J, Almarcegui C, Ara JR, Dolz I, et al. Progressive changes in the retinal nerve fiber layer in patients with multiple sclerosis. Eur J Ophthalmol. 2010;20:167–73.
Petzold A, Balcer LJ, Calabresi PA, Costello F, Frohman TC, Frohman EM, et al. Retinal layer segmentation in multiple sclerosis: a systematic review and meta-analysis. Lancet Neurol. 2017;16:797–812.
Gabilondo I, Sepúlveda M, Ortiz-Perez S, Graga-Pumar E, Martínez-Lapiscina EH, Llufriu S, et al. Retrograde retinal damage after acute optic tract lesion in MS [letter]. J Neurol Neurosurg Psychiatry. 2013;84:824–6.
Saidha S, Al‐Louzi O, Ratchford JN, Bhargava P, Oh J, Newsome SD, et al. Optical coherence tomography reflects brain atrophy in multiple sclerosis: a four‐year study. Ann Neurol. 2015;78:801–13.
Abalo‐Lojo JM, Limeres CC, Gómez MA, Baleato-González S, Cadarso-Suárez, Capeáns-Tomé C, et al. Retinal nerve fiber layer thickness, brain atrophy, and disability in multiple sclerosis patients. J Neuroophthalmol. 2014;34:23–28.
Oh J, Sotirchos ES, Saidha S, Whetstone A, Chen M, Newsome SD, et al. Relationships between quantitative spinal cord MRI and retinal layers in multiple sclerosis. Neurology. 2015;84:720–8.
Garcia‐Martin E, Ara JR, Martin J, Almarcegui C, Dolz I, Vilades E, et al. Retinal and optic nerve degeneration in patients with multiple sclerosis followed up for 5 years. Ophthalmology. 2017;124:688–96.
Ratchford JN, Saidha S, Sotirchos ES, Oh JA, Seigo MA, Eckstein C, et al. Active MS is associated with accelerated retinal ganglion cell/inner plexiform layer thinning. Neurology. 2013;80:47–54.
Rothman A, Murphy OC, Fitzgerald KC, Button J, Gordon-Lipkin E, Ratchford JN, et al. Retinal measurements predict 10-year disability in multiple sclerosis. Ann Clin Transl Neurol. 2019;6:222–32.
Gelfand JM, Nolan R, Schwarz DM, Graves J, Green AJ. Microcystic macular oedema in multiple sclerosis is associated with disease severity. Brain. 2012;135:1786–93.
Saidha S, Sotirchos ES, Ibrahim MA, Crainiceanu CM, Gelfand JM, Sepah YJ, et al. Microcystic macular oedema, thickness of the inner nuclear layer of the retina, and disease characteristics in multiple sclerosis: a retrospective study. Lancet Neurol. 2012;11:963–72.
Wolff B, Azar G, Vasseur V, Sahel JA, Vignal C, Mauget-Faÿsse M. Microcystic changes in the retinal internal nuclear layer associated with optic atrophy: a prospective study. J Ophthalmol. 2014;2014:395189.
Barboni P, Carelli V, Savini G, Carbonelli M, La Morgia C, Sadun AA. Microcystic macular degeneration from optic neuropathy: not inflammatory, not trans-synaptic degeneration. Brain. 2013;136:e239.
Wolff B, Basdekidou C, Vasseur V, Mauget-Faÿsse M, Sahel JA, Vignal C. Retinal inner nuclear layer microcystic changes in optic nerve atrophy: a novel spectral-domain OCT finding. Retina. 2013;33:2133–8.
Pott JWR, de Vries-Knoppert WAEJ, Petzold A. The prevalence of microcystic macular changes on optical coherence tomography of the macular region in optic nerve atrophy of non-neuritis origin: a prospective study. Br J Ophthalmol. 2016;100:216–21.
Bennett JL, de Seze J, Lana-Peixoto M, Waldman A, Schippling S, Tenembaum S, et al. Neuromyelitis optica and multiple sclerosis: Seeing differences through optical coherence tomography. Mult Scler. 2015;21:678–88.
Ratchford JN, Quigg ME, Conger A, Frohman E, Balcer LJ, Calabresi PA, et al. Optical coherence tomography helps differentiate neuromyelitis optica and MS optic neuropathies. Neurology 2009;73:302–8.
Manogaran P, Traboulsee AL, Lange A. Longitudinal study of retinal nerve fiber layer thickness and macular volume in patients with neuromyelitis optica spectrum disorder. J Neuroophthalmol. 2016;36:363–8.
Pisa M, Ratti F, Vabanesi M, Radaelli M, Guerrieri S, Moiola L, et al. Subclinical neurodegeneration in multiple sclerosis and neuromyelitis optica spectrum disorder revealed by optical coherence tomography. Mult Scler. 2020;26:1196–206.
Oertel FC, Outteryck O, Knier B, Zimmermann H, Borisow N, Bellmann-Strobl J, et al. Optical coherence tomography in myelin-oligodendrocyte-glycoprotein antibody-seropositive patients: a longitudinal study. J Neuroinflammation. 2019;16:154.
Balk LJ, Killestein J, Polman C, Uitdehaag BMJ, Petzold A. Microcystic macular oedema confirmed, but not specific for multiple sclerosis [letter]. Brain. 2012;135:e226.
Schneider E, Zimmermann H, Oberwahrenbrock T, Kaufhold F, Kadas EM, Petzold A, et al. Optical coherence tomography reveals distinct patterns of retinal damage in neuromyelitis optica and multiple sclerosis. PLoS ONE. 2013;8:e66151.
Fernandes DB, Raza AS, Nogueira RG, Wang D, Callegaro D, Hood DC, et al. Evaluation of inner retinal layers in patients with multiple sclerosis or neuromyelitis optica using optical coherence tomography. Ophthalmology. 2013;120:387–94.
Hamann S, Malmqvist L, Costello F. Optic disc drusen: understanding an old problem from a new perspective. Acta Ophthalmol. 2018;96:673–84.
Chen JJ, Costello F. The role of optical coherence tomography in neuro-ophthalmology. Ann Eye Sci. 2018;3:35.
Lee KM, Woo SJ, Hwang JM. Differentiation of optic nerve head drusen and optic disc edema with spectral domain optical coherence tomography. Ophthalmology. 2011;228:971–7.
Bassi ST, Mohana KP. Optical coherence tomography in papilledema and pseudopapilledema with and without optic nerve head drusen. Indian J Ophthalmol. 2014;62:1146–51.
Kulkarni KM, Pasol J, Rosa PR, Lam BL. Differentiating mild papilledema and buried optic nerve head drusen using spectral domain optical coherence tomography. Ophthalmology. 2014;121:959–63.
Johnson LN, Diehl ML, Hamm CW, Sommerville DN, Petroski GF. Differentiating optic disc edema from optic nerve head drusen on optical coherence tomography. Arch Ophthalmol. 2009;127:45–9.
Sarac O, Tasci YY, Gurdal C, Can I. Differentiation of optic disc edema from optic nerve head drusen with spectral-domain optical coherence tomography. J Neuroophthalmol. 2012;32:207–11.
Malmqvist L, Bursztyn L, Costello F, Digre K, Fraser JA, Fraser C, et al. The optic disc drusen studies consortium recommendations for diagnosis of optic disc drusen using optical coherence tomography. J Neuroophthalmol. 2018;38:299–307.
Merchant KY, Su D, Park SC, Qayum S, Banik R, Liebmann JM, et al. Enhanced depth imaging optical coherence tomography of optic nerve head drusen. Ophthalmology. 2013;120:1409–14.
Traber GL, Weber KP, Sabah M, Keane PA, Plant GT. Enhanced depth imaging optical coherence tomography of optic nerve head drusen: a comparison of cases with and without visual field loss. Ophthalmology. 2017;124:66–73.
Skaat A, Muylaert S, Mogil RS, Furlanetto RL, Netto CF, Banik R, et al. Relationship between optic nerve head drusen volume and structural and functional optic nerve head damage. J Glaucoma. 2017;26:1095–100.
Corbett JJ, Savino PJ, Thompson HS, Kansu T, Schatz NJ, Orr LS, et al. Visual loss in pseudotumour cerebri. Follow-up of 57 patients from five to 41 years and a profile of 14 patients with permanent severe visual loss. Arch Neurol. 1982;39:461–74.
Scott CJ, Kardon RH, Lee AG, Frisén L, Wall M. Diagnosis and grading of papilledema in patients with raised intracranial pressure using optical coherence tomography vs clinical expert assessment using a clinical staging scale. Arch Ophthalmol. 2010;128:705–11.
Sinclair AJ, Burdon MA, Nightingale PG, Matthews TD, Jacks A, Lawden M, et al. Rating papilloedema: an evaluation of the Frisén classification in idiopathic intracranial hypertension. J Neurol. 2012;259:1406–12.
Kupersmith MJ, Sibony P, Mandel G, Durbin M, Kardon RH. Optical coherence tomography of the swollen optic nerve head: deformation of the peripapillary retinal pigment epithelium layer in papilledema. Invest Ophthalmol Vis Sci. 2011;52:6558–64.
Sibony P, Kupersmith MJ, Rohlf FJ. Shape analysis of the peripapillary RPE layer in papilledema and ischemic optic neuropathy. Invest Ophthalmol Vis Sci. 2011;52:7987–95.
Sibony P, Kupersmith MJ, Honkanen R, Rohlf FJ, Torab-Parhiz A. Effects of lowering cerebrospinal fluid pressure on the shape of the peripapillary retina in intracranial hypertension. Invest Ophthalmol Vis Sci. 2014;55:8223–31.
Gampa A, Vangipuram G, Shirazi Z, Moss HE. Quantitative association between peripapillary Bruch’s membrane shape and intracranial pressure. Invest Ophthalmol Vis Sci. 2017;58:2739–45.
Anand A, Pass A, Urfy MZ, Tang R, Cajavilca C, Calvillo E, et al. Optical coherence tomography of the optic nerve head detects acute changes in intracranial pressure. J Clin Neurosci. 2016;29:73–6.
Hoye VJ 3rd, Berrocal AM, Hedges TR 3rd, Amaro-Quireza ML. Optical coherence tomography demonstrates subretinal macular edema from papilledema. J Clin Neurosci. 2016;29:73–6.
Dotan G, Goldstein M, Kesler A, Skarf B. Long-term retinal nerve fiber layer changes following nonarteritic anterior ischaemic optic neuropathy. Clin Ophthalmol. 2013;7:735–40.
Contreras I, Rebolleda G, Noval S, Muñoz-Negrete FJ. Optic disc evaluation by optical coherence tomography in nonarteritic anterior ischaemic optic neuropathy. Invest Ophthalmol Vis Sci. 2007;48:4087–92.
Contreras I, Rebolleda G, Noval S, Muñoz-Negrete FJ. Ischemic optic neuropathy. Ophthalmology. 2009;116:814.
De Dompablo E, Garcia-Montesinos J, Munoz-Negrete FJ, Rebolleda G. Ganglion cell analysis at acute episode of nonarteritic anterior ischemic optic neuropathy to predict irreversible damage. A prospective study. Graefes Arch Clin Exp Ophthalmol. 2016;254:1793–800.
Kernstock C, Beisse F, Wiethoff S, Mast A, Krapp E, Grund R, et al. Assessment of functional and morphometric endpoints in patients with non-arteritis anterior ischemic optic neuropathy (NAION). Graefes Arch Clin Exp Ophthalmol. 2014;252:515–21.
Papchenko T, Grainger BT, Savino PJ, Gamble GD, Danesh-Meyer HV. Macular thickness predictive of visual field sensitivity in ischaemic optic neuropathy. Acta Ophthalmol. 2012;90:463–9.
Gonul S, Koktekir BE, Bakbak B, Gedik S. Comparison of the ganglion cell complex and retinal nerve fiber layer measurements using Fourier domain optical coherence tomography to detect ganglion cell loss in non-arteritis anterior ischaemic optic neuropathy. Br J Ophthalmol. 2013;97:1045–50.
Zhang Y, Ye Z, Wang M, Qiao N. Ganglion cell complex loss precedes retinal nerve fiber layer thinning in patients with pituitary adenoma. J Clin Neurosci. 2017;43:274–7.
Monteiro MLR. Macular ganglion cell complex reduction preceding visual field loss in a patient with chiasmal compression with a 21-month follow-up. J Neuroophthalmol. 2018;38:124–7.
Tieger MG, Hedges TR3rd, Ho J, Erlich-Malona NK, Vuong LN, Athappilly GK. Ganglion cell complex loss in chiasmal compression by brain tumours. J Neuroophthalmol. 2017;37:7–12.
Unsold R, Hoyt WF. Band atrophy of the optic nerve. The histology of temporal hemianopsia. Arch Ophthalmol. 1980;98:1637–8.
Monteiro ML, Leal BC, Rosa AA, Bronstein MD. Optical coherence tomography analysis of axonal loss in band atrophy of the optic nerve. Br J Ophthalmol. 2004;88:896–9.
Kanamori A, Nakamura M, Matsui N, Nagai A, Nakanishi Y, Kusuhara S, et al. Optical coherence tomography detects characteristic retinal nerve fiber layer thickness corresponding to band atrophy of the optic discs. Ophthalmology. 2004;111:2278–83.
Danesh-Meyer HV, Carroll SC, Foroozan R, Savino PJ, Fan J, Jiang Y, et al. Relationship between retinal nerve fiber layer and visual field sensitivity as measured by optical coherence tomography in chiasmal compression. Invest Ophthalmol Vis Sci. 2006;47:4827–35.
Costa-Cunha LV, Cunha LP, Malta RF, Monteiro MLR. Comparison of Fourier-domain and time-domain optical coherence tomography in the detection of band atrophy of the optic nerve. Am J Ophthalmol. 2009;147:56–63.e2.
Monteiro ML, Cunha LP, Vessani RM. Comparison of retinal nerve fiber layer measurements using Stratus OCT fast and regular scan protocols in eyes with band atrophy of the optic nerve and normal controls. Arq Bras Oftalmol. 2008;71:534–9.
Sun M, Zhang A, Ma C, Chin S, Chen X. Quantitative analysis of retinal layers on three-dimensional spectral-domain optical coherence tomography for pituitary adenoma. PLoS ONE. 2017;12:e0179532.
Yum HR, Park SH, Park HY, Shin SY. Macular ganglion cell analysis determined by cirrus HD optical coherence tomography for early detecting chiasmal compression. PLoS ONE. 2016;11:e0153064.
Zehnder S, Wildberger H, Hanson JVM, Lukas S, Pelz S, Landau K, et al. Retinal ganglion cell topography in patients with visual pathway pathology. J Neuroophthalmol. 2018;38:172–8.
Dinkin M. Trans-synaptic retrograde degeneration in the human visual system: slow, silent, and real. Curr Neurol Neurosci Rep. 2017;17:16.
Fisher MJ, Loguidice M, Gutmann DH, Listernick R, Ferner RE, Ullrich NJ, et al. Visual outcomes in children with neurofibromatosis type I-associated optic pathway glioma following chemotherapy: a multicenter retrospective analysis. Neuro Oncol. 2012;14:790–7.
Avery RA, Cnaan A, Schuman JS, Trimboli-Heidler C, Chen CL, Packer RJ, et al. Longitudinal change of circumpapillary retinal fiber layer thickness in children with optic pathway gliomas. Am J Ophthalmol. 2015;160:944–52.
Avery RA, Mansoor A, Idrees R, Trimboli-Heidler C, Ishikawa H, Packer RJ, et al. Optic pathway glioma volume predicts retinal axon degeneration in neurofibromatosis type 1. Neurology. 2016;87:2403–7.
Yoneoka Y, Hatase T, Watanabe N, Jinguji S, Okada M, Takagi M, et al. Early morphological recovery of the optic chiasm is associated with excellent visual outcome in patients with compressive chiasmal syndrome caused by pituitary tumors. Neurol Res. 2015;37:1–8.
Ohkubo S, Higashide T, Takeda H, Murotani E, Hayashi Y, Sugiyama K. Relationship between macular ganglion cell complex parameters and visual field parameters after tumor resection in chiasmal compression. Jpn J Ophthalmol. 2012;56:68–75.
Moon CH, Hwang SC, Kim BT, Ohn YH, Park TK. Visual prognostic value of optical coherence tomography and photopic negative response in chiasmal compression. Invest Ophthalmol Vis Sci. 2011;52:8527–33.
Matsunaga D, Yi J, Puliafito CA, Kashani AH. OCT angiography in healthy human subjects. Osli Retin 2014;45:510–5.
Mase T, Ishibazawa A, Nagaoka T, Yokota H, Yoshida A. Radial peripapillary capillary network visualized using wide-field montage optical coherence tomography angiography. Invest Ophth Vis Sci. 2016;57:OCT504–10.
Bojikian KD, Chen CL, Wen JC, Zhang QQ, Xin C, Gupta D, et al. Optic disc perfusion in primary open angle and normal tension glaucoma eyes using optical coherence tomography-based microangiography. PLoS ONE. 2016;11:e0154691.
Wang XG, Jia YL, Spain R, Potsaid B, Liu JJ, Baumann B, et al. Optical coherence tomography angiography of optic nerve head and parafovea in multiple sclerosis. Brit J Ophthalmol. 2014;98:1368–73.
Hata M, Oishi A, Muraoka Y, Miyamoto K, Kawai K, Yokota S, et al. Structural and functional analyses in nonarteritic anterior ischemic optic neuropathy: optical coherence tomography angiography study. J Neuro-Ophthalmol. 2017;37:140–8.
Sharma S, Ang M, Najjar RP, Sng C, Cheung CY, Rukmini AV, et al. Optical coherence tomography angiography in acute non-arteritic anterior ischaemic optic neuropathy. Brit J Ophthalmol. 2017;101:1045–51.
Gaier ED, Gilbert AL, Cestari DM, Miller JB. Optical coherence tomographic angiography identifies peripapillary microvascular dilation and focal non-perfusion in giant cell arteritis. Brit J Ophthalmol. 2018;102:1141–6.
Balducci N, Cascavilla ML, Ciardella A, La Morgia C, Triolo G, Parisi V, et al. Peripapillary vessel density changes in Leber’s hereditary optic neuropathy: a new biomarker. Clin Exp Ophthalmol. 2018;46:1055–62.
De Rojas JO, Rasool N, Chen RWS, Horowitz J, Odel JG. Optical coherence tomography angiography in Leber hereditary optic neuropathy. Neurology. 2016;87:2065–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Minakaran, N., de Carvalho, E.R., Petzold, A. et al. Optical coherence tomography (OCT) in neuro-ophthalmology. Eye 35, 17–32 (2021). https://doi.org/10.1038/s41433-020-01288-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-020-01288-x
This article is cited by
-
MOG35 − 55-induced EAE model of optic nerve inflammation compared to MS, MOGAD and NMOSD related subtypes of human optic neuritis
Journal of Neuroinflammation (2025)
-
Optical coherence tomography surpasses fundus imaging and intracranial pressure measurement in monitoring idiopathic intracranial hypertension
Scientific Reports (2025)
-
Impact of light polarization on laser speckle contrast imaging with a custom phantom for microvascular flow
Scientific Reports (2024)
-
Conservative or surgical management of orbital schwannomas: a population-based case series
Acta Neurochirurgica (2024)
-
Detection sensitivity of fluorescence lifetime imaging ophthalmoscopy for laser-induced selective damage of retinal pigment epithelium
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)