Abstract
Background and objective
To evaluate the proportion of patients achieving a 12-week (q12) aflibercept dosing interval in patients with neovascular age-related macular degeneration (nAMD).
Patients and methods
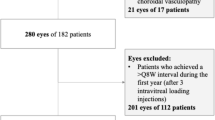
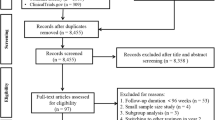
Retrospective, comparative, non-randomised electronic medical record (EMR) database study of the Moorfields database of treatment-naïve nAMD eyes. Extraction criteria included at least 7 aflibercept injections in first year of treatment, AMD in the diagnosis field of EMR, and minimum of 1 year follow-up data.
Results
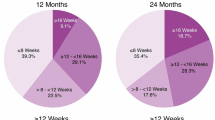
There were 2416 eyes of 2163 patients started on anti-vascular endothelial growth factor (anti-VEGF) between 01-11-2013 & 14-02-2020 who had received at least 7 aflibercept intravitreal injections (electronic database accessed March 2021). Of these, 1674 (68%) eyes of 1537 patients had at least one q12 dosing interval (>=84 and < =98 days between injections) during the first 2 years of treatment. This included 926 (61.8%) female patients and 856 (right eyes age at 1st injection), 936 (62.4%) Caucasian, and 32 (2.1%) Afro-Caribbean patients. The median time to the first q12 injection (95% confidence interval) was 1.76 years (1.70–1.86) with mean (±SD) of 11.8 (±6.0) injections. Visual acuity (ETDRS letters) of the eyes without q12 injection and eyes with a q12 injection was 57.9 ± 14.7 and 56.7 ± 14.8 respectively at baseline, 61.4 ± 18.1 and 63.0 ± 15.9 respectively at 12 months and 61.2 ± 20.1 and 61.1 ± 17.8 respectively at 24 months.
Conclusion
68% of eyes were able to achieve a q12 injection dose within the first 2 years of treatment. Eyes achieving a q12 injection in the first 2 years achieved a similar visual acuity outcome at both 1 and 2-year follow-up to those unable to do so, with a fewer number of total injections.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
McKibbin M, Devonport H, Gale R, Gavin M, Lotery A, Mahmood S, et al. Aflibercept in wet AMD beyond the first year of treatment: recommendations by an expert roundtable panel. Eye. 2015;29 Suppl 1:S1–11. https://doi.org/10.1038/eye.2015.77.
Hsu J, Regillo CD. Poorer outcomes in real-world studies of anti-vascular endothelial growth factor therapy for neovascular age-related macular degeneration. Ophthalmology. 2020;127:1189–90. https://doi.org/10.1016/j.ophtha.2020.03.034.
Almuhtaseb H, Johnston RL, Talks JS, Lotery AJ. Second-year visual acuity outcomes of nAMD patients treated with aflibercept: data analysis from the UK Aflibercept Users Group. Eye. 2017;31:1582–8. https://doi.org/10.1038/eye.2017.108.
Little K, Ma JH, Yang N, Chen M, Xu H. Myofibroblasts in macular fibrosis secondary to neovascular age-related macular degeneration - the potential sources and molecular cues for their recruitment and activation. EBioMedicine. 2018;38:283–91. https://doi.org/10.1016/j.ebiom.2018.11.029.
Okeagu CU, Agrón E, Vitale S, Domalpally A, Chew EY, Keenan TDL. Principal cause of poor visual acuity after neovascular age-related macular degeneration: age-related eye disease study 2 report number 23. Ophthalmol Retin. 2021;5:23–31. https://doi.org/10.1016/j.oret.2020.09.025.
Eleftheriadou M, Gemenetzi M, Lukic M, Sivaprasad S, Hykin PG, Hamilton RD, et al. Three-year outcomes of aflibercept treatment for neovascular age-related macular degeneration: evidence from a clinical setting. Ophthalmol Ther. 2018;7:361–8. https://doi.org/10.1007/s40123-018-0139-5.
Richard G, Monés J, Wolf S, Korobelnik JF, Guymer R, Goldstein M, et al. Scheduled versus Pro Re Nata dosing in the VIEW trials. Ophthalmology. 2015;122:2497–503. https://doi.org/10.1016/j.ophtha.2015.08.014.
Traine PG, Pfister IB, Zandi S, Spindler J, Garweg JG. Long-term outcome of intravitreal aflibercept treatment for neovascular age-related macular degeneration using a “Treat-and-Extend” regimen. Ophthalmol Retin. 2019;3:393–9. https://doi.org/10.1016/j.oret.2019.01.018.
Barthelmes D, Nguyen V, Daien V, Campain A, Walton R, Guymer R, et al. Two year outcomes of “treat and extend” intravitreal therapy using aflibercept preferentially for neovascular age-related macular degeneration. Retina. 2018;38:20–28. https://doi.org/10.1097/iae.0000000000001496.
Heier JS, Brown DM, Chong V, Korobelnik JF, Kaiser PK, Nguyen QD, et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology. 2012;119:2537–48. https://doi.org/10.1016/j.ophtha.2012.09.006.
Khurana RN, Rahimy E, Joseph WA, Saroj N, Gibson A, Vitti R, et al. Extended (Every 12 Weeks or Longer) dosing interval with intravitreal aflibercept and ranibizumab in neovascular age-related macular degeneration: post hoc analysis of VIEW trials. Am J Ophthalmol. 2019;200:161–8. https://doi.org/10.1016/j.ajo.2019.01.005.
Egan C, Zhu H, Lee A, Sim D, Mitry D, Bailey C, et al. The United Kingdom Diabetic Retinopathy Electronic Medical Record Users Group, Report 1: baseline characteristics and visual acuity outcomes in eyes treated with intravitreal injections of ranibizumab for diabetic macular oedema. Br J Ophthalmol. 2017;101:75–80. https://doi.org/10.1136/bjophthalmol-2016-309313.
The neovascular age-related macular degeneration database: multicenter study of 92976 ranibizumab injections: report 1: visual acuity. Ophthalmology. May 2014;121:1092–101. https://doi.org/10.1016/j.ophtha.2013.11.031.
Fasler K, Moraes G, Wagner S, Kortuem KU, Chopra R, Faes L, et al. One- and two-year visual outcomes from the Moorfields age-related macular degeneration database: a retrospective cohort study and an open science resource. BMJ Open. 2019;9:e027441 https://doi.org/10.1136/bmjopen-2018-027441.
Garweg JG, Gerhardt C Disease stability and extended dosing under anti-VEGF treatment of exudative age-related macular degeneration (AMD) - a meta-analysis. Graefe’s archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. Feb 2021;https://doi.org/10.1007/s00417-020-05048-1.
Garweg JG. Twelve-week dosing with Aflibercept in the treatment of neovascular age-related macular degeneration. Clin Ophthalmol. 2019;13:1289–95. https://doi.org/10.2147/opth.S185756.
Fu DJ, Keenan TD, Faes L, Lim E, Wagner SK, Moraes G, et al. Insights from survival analyses during 12 years of anti-vascular endothelial growth factor therapy for neovascular age-related macular degeneration. JAMA Ophthalmol. 2021;139:57–67. https://doi.org/10.1001/jamaophthalmol.2020.5044.
Bunce C, Fu DJ, Stratton I. Survival analysis vs longitudinal modeling with multiple imputation-a false dichotomy-reply. JAMA Ophthalmol. 2021;139:588–9. https://doi.org/10.1001/jamaophthalmol.2021.0518.
Fasler K, Fu DJ, Moraes G, Wagner S, Gokhale E, Kortuem K, et al. Moorfields AMD database report 2: fellow eye involvement with neovascular age-related macular degeneration. Br J Ophthalmol. 2020;104:684–90. https://doi.org/10.1136/bjophthalmol-2019-314446.
Farmer R, Mathur R, Bhaskaran K, Eastwood SV, Chaturvedi N, Smeeth L. Promises and pitfalls of electronic health record analysis. Diabetologia. 2018;61:1241–8. https://doi.org/10.1007/s00125-017-4518-6.
Chakravarthy U, Havilio M, Syntosi A, Pillai N, Wilkes E, Benyamini G, et al. Impact of macular fluid volume fluctuations on visual acuity during anti-VEGF therapy in eyes with nAMD. Eye. 2021;35:2983–90. https://doi.org/10.1038/s41433-020-01354-4.
Author information
Authors and Affiliations
Contributions
DJF designed the study, collected the study data and completed the analysis. DH designed the study and wrote the manuscript; SW and TK provided critical appraisal of the manuscript. PP, PK and KB designed the study and provided critical appraisal of the manuscript.
Corresponding author
Ethics declarations
Competing interests
Dr. Patel, Dr. Keane, and Dr. Balaskas have received a proportion of their funding from the Department of Health’s NIHR Biomedical Research Centre at Moorfields Eye Hospital and UCL Institute of Ophthalmology. Dr. Keane has acted as a consultant for DeepMind, Roche, Novartis, and Apellis and is an equity owner in Big Picture Medical. He has received speaker fees from Heidelberg Engineering, Topcon, Allergan, and Bayer. Dr. Keane is supported by a Moorfields Eye Charity Career Development Award (R190028A) and a UK Research & Innovation Future Leaders Fellowship (MR/T019050/1). Dr. Balakas has received speaker fees from Novartis, Bayer, Alimera, Allergan, and Heidelberg, consulting fees from Novartis and Roche, and research support from Apellis, Roche, Novartis, and Bayer. Dr. Patel has received speaker fees from Novartis, Bayer, Topcon, and Heidelberg, consulting fees from Novartis, Bayer, Oxford Bioelectronics, and Roche, and research support from Bayer. Dr. Fu has acted as consultant for Abbvie, Allergan, DeepMind.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fu, D.J., Hanumunthadu, D., Keenan, T.D.L. et al. Characterising treatment outcomes of patients achieving quarterly aflibercept dosing for neovascular age-related macular degeneration: real-world clinical outcomes from a large tertiary care centre. Eye 37, 779–784 (2023). https://doi.org/10.1038/s41433-022-02220-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-022-02220-1