Abstract
Objectives
To evaluate respiratory outcomes in preterm infants with retinopathy of prematurity (ROP) following intravitreal bevacizumab injection (IVB).
Methods
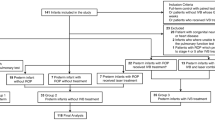
This single-centre study enroled preterm infants with a gestational age (GA) < 34 weeks or a birth weight (BW) < 1500 g with bilateral type 1 ROP who received a single IVB, and a treatment-free control group matched by GA, postmenstrual age, and respiratory status at the time of the IVB. The primary outcome was serial respiratory changes in mean airway pressure (MAP), fraction of inspired oxygen (FiO2), and respiratory severity score (RSS, MAP x FiO2) during the 28-day post-IVB/matching period and overall respiratory improvement at day 28 and at discharge. The duration of supplemental oxygen therapy following IVB/matching was documented.
Results
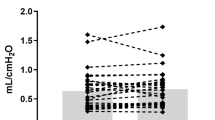
A total of 5578 infants were included. Seventy-eight infants were enroled in the IVB group, and another 78 infants were matched as the control group. Both groups had downward trends in the MAP, FiO2, and RSS over the study period (all P < 0.001), but there were no between-group differences in these measures. The percentage of overall respiratory improvement was similar between the IVB and control groups, so was the duration of invasive and in-hospital oxygen ventilation. A lower percentage of oxygen dependence at discharge in the IVB group (P = 0.03) remained significant after adjusting for GA and BW.
Conclusions
This is a matched case study to evaluate respiratory outcomes in preterm infants following IVB for ROP. We found that the IVBs did not compromise respiratory outcomes in preterm infants during the 28-day post-IVB period and at discharge.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
Due to sensitivity concerns, the data supporting the study’s conclusions are not publicly accessible, but can be obtained from the corresponding author upon request. The data are held in a secure, controlled-access storage facility at Chang Gung Memorial Hospital.
References
Stahl A, Krohne TU, Eter N, Oberacher-Velten I, Guthoff R, Meltendorf S, et al. Comparing alternative ranibizumab dosages for safety and efficacy in retinopathy of prematurity: a randomized clinical trial. JAMA Pediatr. 2018;172:278–86.
Mintz-Hittner HA, Kennedy KA, Chuang AZ. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Engl J Med. 2011;364:603–15.
Jo DH, Park SW, Cho CS, Powner MB, Kim JH, Fruttiger M, et al. Intravitreally injected Anti-VEGF antibody reduces brown fat in neonatal mice. PLoS One. 2015;10:e0134308.
Khalili S, Shifrin Y, Pan J, Belik J, Mireskandari K. The effect of a single anti-vascular endothelial growth factor injection on neonatal growth and organ development: In-vivo study. Exp Eye Res. 2018;169:54–9.
Monacci WT, Merrill MJ, Oldfield EH. Expression of vascular permeability factor/vascular endothelial growth factor in normal rat tissues. Am J Physiol-Cell Physiol. 1993;264:C995–C1002.
Sato T, Wada K, Arahori H, Kuno N, Imoto K, Iwahashi-Shima C, et al. Serum concentrations of bevacizumab (avastin) and vascular endothelial growth factor in infants with retinopathy of prematurity. Am J Ophthalmol. 2012;153:327–33.e1.
Kong L, Bhatt AR, Demny AB, Coats DK, Li A, Rahman EZ, et al. Pharmacokinetics of bevacizumab and its effects on serum VEGF and IGF-1 in infants with retinopathy of prematurity. Invest Ophthalmol Vis Sci. 2015;56:956–61.
Wu WC, Shih CP, Lien R, Wang NK, Chen YP, Chao AN, et al. Serum vascular endothelial growth factor after bevacizumab or ranibizumab treatment for retinopathy of prematurity. Retina. 2017;37:694–701.
Avery RL, Castellarin AA, Steinle NC, Dhoot DS, Pieramici DJ, See R, et al. Systemic pharmacokinetics and pharmacodynamics of intravitreal aflibercept, bevacizumab, and ranibizumab. Retin (Phila, Pa). 2017;37:1847–58.
Avery RL. Bevacizumab (Avastin) for retinopathy of prematurity: wrong dose, wrong drug, or both? J AAPOS. 2012;16:2–4.
Fan Y-Y, Huang Y-S, Huang C-Y, Hsu J-F, Shih C-P, Hwang Y-S, et al. Neurodevelopmental outcomes after intravitreal bevacizumab therapy for retinopathy of prematurity: a prospective case-control study. Ophthalmology. 2019;126:1567–77.
Group. ETfRoPC. Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol (Chic, Ill: 1960). 2003;121:1684–94.
Good WV, Hardy RJ, Dobson V, Palmer EA, Phelps DL, Tung B, et al. Final visual acuity results in the early treatment for retinopathy of prematurity study. Arch Ophthalmol (Chic, Ill: 1960). 2010;128:663–71.
Backes CH, Cooper JN, Notestine JL, Alfred CM, Ball MK, Rivera BK, et al. A trial comparing continuous positive airway pressure (CPAP) devices in preterm infants. J Perinatol. 2020;40:1193–201.
Jung YH, Jang J, Kim H-S, Shin SH, Choi CW, Kim E-K, et al. Respiratory severity score as a predictive factor for severe bronchopulmonary dysplasia or death in extremely preterm infants. BMC Pediatr. 2019;19:121.
Şahin A, Şahin M, Türkcü FM, Cingü AK, Yüksel H, Çınar Y, et al. Incidence of retinopathy of prematurity in extremely premature infants. ISRN Pediatr. 2014;2014:134347.
Woo SJ, Park KH, Ahn J, Oh KJ, Lee SY, Jeong EH, et al. A co-twin study of the relative effect of birth weight and gestational age on retinopathy of prematurity. Eye. 2011;25:1478–83.
Good WV. Final results of the early treatment for retinopathy of prematurity (ETROP) randomized trial. Trans Am Ophthalmol Soc. 2004;102:233–48.
Huang C-Y, Lien R, Wang N-K, Chao A-N, Chen K-J, Chen T-L, et al. Changes in systemic vascular endothelial growth factor levels after intravitreal injection of aflibercept in infants with retinopathy of prematurity. Graefe’s Arch Clin Exp Ophthalmol. 2017;256:479–87.
Owen LS, Manley BJ, Davis PG, Doyle LW. The evolution of modern respiratory care for preterm infants. Lancet (Lond, Engl). 2017;389:1649–59.
Kirpalani H, Ratcliffe SJ, Keszler M, Davis PG, Foglia EE, te Pas A, et al. Effect of sustained inflations vs intermittent positive pressure ventilation on bronchopulmonary dysplasia or death among extremely preterm infants: The SAIL randomized clinical trial. JAMA. 2019;321:1165–75.
Roberts CT, Owen LS, Manley BJ, Frøisland DH, Donath SM, Dalziel KM, et al. Nasal high-flow therapy for primary respiratory support in preterm infants. N Engl J Med. 2016;375:1142–51.
Backes CH, Notestine JL, Lamp JM, Balough JC, Notestine AM, Alfred CM, et al. Evaluating the efficacy of Seattle-PAP for the respiratory support of premature neonates: study protocol for a randomized controlled trial. Trials. 2019;20:63.
Nankervis CA, Martin EM, Crane ML, Samson KS, Welty SE, Nelin LD. Implementation of a multidisciplinary guideline-driven approach to the care of the extremely premature infant improved hospital outcomes. Acta Paediatr (Oslo, Nor: 1992). 2010;99:188–93.
Supplemental Therapeutic Oxygen for Prethreshold Retinopathy Of Prematurity (STOP-ROP), a randomized, controlled trial. I: primary outcomes. Pediatrics. 2000;105:295–310.
Hsu KH, Wong P, Ram Kumar S, Evans J, Noori S. Predictors of respiratory improvement 1 week after ligation of patent ductus arteriosus in preterm infants. J Pediatr. 2019;205:49–54.e2.
Atalay D, Salihoğlu O, Can E, Beşkardeş A, Hatipoğlu S. Short-term outcomes of very low birth weight infants born at a tertiary care hospital, istanbul, Turkey. Iran J Pediatr. 2013;23:205–11.
Apisarnthanarak A, Holzmann-Pazgal G, Hamvas A, Olsen MA, Fraser VJ. Ventilator-associated pneumonia in extremely preterm neonates in a neonatal intensive care unit: characteristics, risk factors, and outcomes. Pediatrics. 2003;112:1283.
Grisaru-Granovsky S, Reichman B, Lerner-Geva L, Boyko V, Hammerman C, Samueloff A, et al. Mortality and morbidity in preterm small-for-gestational-age infants: a population-based study. Am J Obstet Gynecol. 2012;206:150.e1–e7.
Jiang J-B, Strauss R, Luo X-Q, Nie C, Wang Y-L, Zhang J-W, et al. Anaesthesia modalities during laser photocoagulation for retinopathy of prematurity: a retrospective, longitudinal study. BMJ Open. 2017;7:e013344.
Hartrey R. Anaesthesia for the laser treatment of neonates with retinopathy of prematurity. Eye. 2007;21:1025–7.
Binenbaum G, Ying G-S, Quinn GE, Huang J, Dreiseitl S, Antigua J, et al. The CHOP postnatal weight gain, birth weight, and gestational age retinopathy of prematurity risk model. Arch Ophthalmol. 2012;130:1560–5.
Simonsen KA, Anderson-Berry AL, Delair SF, Davies HD. Early-onset neonatal sepsis. Clin Microbiol Rev. 2014;27:21–47.
Hermansen CL, Lorah KN. Respiratory distress in the newborn. Am Fam physician. 2007;76:987–94.
Gephart SM, McGrath JM, Effken JA, Halpern MD. Necrotizing enterocolitis risk: state of the science. Adv Neonatal Care. 2012;12:77–89.
Jakuskiene R, Vollmer B, Saferis V, Daugeliene D. Neonatal outcomes of very preterm infants admitted to a tertiary center in Lithuania between the years 2003 and 2005. Eur J Pediatr. 2011;170:1293–303.
Thébaud B, Abman SH. Bronchopulmonary dysplasia. Am J Respiratory Crit Care Med. 2007;175:978–85.
Higgins RD, Jobe AH, Koso-Thomas M, Bancalari E, Viscardi RM, Hartert TV, et al. Bronchopulmonary dysplasia: executive summary of a workshop. J Pediatr. 2018;197:300–8.
Valencia AM, Cai CL, Tan J, Duggan TJ, Valencia GB, Aranda JV, et al. Intravitreal bevacizumab alters type IV collagenases and exacerbates arrested alveologenesis in the neonatal rat lungs. Exp Lung Res. 2017;43:120–33.
Gerber HP, Hillan KJ, Ryan AM, Kowalski J, Keller GA, Rangell L, et al. VEGF is required for growth and survival in neonatal mice. Development. 1999;126:1149.
Le Cras TD, Spitzmiller RE, Albertine KH, Greenberg JM, Whitsett JA, Akeson AL. VEGF causes pulmonary hemorrhage, hemosiderosis, and air space enlargement in neonatal mice. Am J Physiol-Lung Cell Mol Physiol. 2004;287:L134–L42.
Burri PH, Moschopulos M. Structural analysis of fetal rat lung development. Anat Rec. 1992;234:399–418.
Jakkula M, Le Cras TD, Gebb S, Hirth KP, Tuder RM, Voelkel NF, et al. Inhibition of angiogenesis decreases alveolarization in the developing rat lung. Am J Physiol-Lung Cell Mol Physiol. 2000;279:L600–L7.
Le Cras TD, Markham NE, Tuder RM, Voelkel NF, Abman SH. Treatment of newborn rats with a VEGF receptor inhibitor causes pulmonary hypertension and abnormal lung structure. Am J Physiol-Lung Cell Mol Physiol. 2002;283:L555–L62.
Stahl A, Lepore D, Fielder A, Fleck B, Reynolds JD, Chiang MF, et al. Ranibizumab versus laser therapy for the treatment of very low birthweight infants with retinopathy of prematurity (RAINBOW): an open-label randomised controlled trial. Lancet. 2019;394:1551–9.
Niedermaier S, Hilgendorff A. Bronchopulmonary dysplasia—an overview about pathophysiologic concepts. Mol Cell Pediatr. 2015;2:2.
Bose CL, Dammann CEL, Laughon MM. Bronchopulmonary dysplasia and inflammatory biomarkers in the premature neonate. Arch Dis Child - Fetal Neonatal Ed. 2008;93:F455.
de Jesus Perez VA, Alastalo T-P, Wu JC, Axelrod JD, Cooke JP, Amieva M, et al. Bone morphogenetic protein 2 induces pulmonary angiogenesis via Wnt-beta-catenin and Wnt-RhoA-Rac1 pathways. J Cell Biol. 2009;184:83–99.
Benesch M, Windelberg M, Sauseng W, Witt V, Fleischhack G, Lackner H, et al. Compassionate use of bevacizumab (Avastin®) in children and young adults with refractory or recurrent solid tumors. Ann Oncol. 2008;19:807–13.
Warburton D. Overview of lung development in the newborn human. Neonatology. 2017;111:398–401.
Been JV, Debeer A, van Iwaarden JF, Kloosterboer N, Passos VL, Naulaers G, et al. Early alterations of growth factor patterns in bronchoalveolar lavage fluid from preterm infants developing bronchopulmonary dysplasia. Pediatr Res. 2010;67:83–9.
Acknowledgements
We would like to thank the Clinical Trial Centre in Chang Gung Memorial Hospital, which is supported by the Ministry of Health and Welfare of Taiwan (MOHW110-TDU-B-212-114005), for statistical assistance. Each author affirms that he or she has no conflict of interest.
Funding
This study was supported by Chang Gung Memorial Hospital Research Grants (CMRPG3I0071-3 and CMRPG3K0421), a Ministry of Science and Technology Research Grant (MOST 109-2314-B-182A-019-MY3). WCW work has been funded by Chang Gung Memorial Hospital Research Grants and Ministry of Science and Technology Research Grant. YCH, KHH, SMC, MCC, RL, KJC, YSH, CCL, HJT declared no proprietary or commercial interest in any materials discussed in this article.
Author information
Authors and Affiliations
Contributions
YCH was responsible for conceptualization/design, investigation, methodology, data curation, formal analysis, and drafting the initial manuscript. KHH was responsible for conceptualization/design, investigation, methodology, data curation, formal analysis, and editing of the manuscript. SMC was responsible for investigation, data curation, supervision/oversight, resources, and review of the manuscript. MCC and RL were responsible for investigation, methodology, data curation, supervision/oversight, and resources. KJC, YSH, and LCC were responsible for investigation, data curation, supervision/oversight, resources, and review of the manuscript. HJT was responsible for methodology, data curation, formal analysis, and editing of the manuscript. WWC was responsible for conceptualization/design, investigation, methodology, data curation, supervision/oversight, resources, funding acquisition, and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study was approved by the Institutional Review Board of Chang Gung Memorial Hospital in Linkou, Taiwan (IRB201900571B0) and was conducted in accordance with the Declaration of Helsinki. Patient consent for publication was not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, YC., Hsu, KH., Chu, SM. et al. Respiratory outcomes in preterm infants following intravitreal bevacizumab for retinopathy of prematurity–a 10-year matched case study. Eye 37, 3675–3681 (2023). https://doi.org/10.1038/s41433-023-02579-9
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-023-02579-9