Abstract
Objective
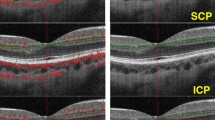
To evaluate the role of retinal and choriocapillaris blood flow in the development of diabetic foveal neovascularization (DFN) using optical coherence tomography angiography (OCTA).
Methods
Eyes with proliferative diabetic retinopathy (PDR) and DFN underwent OCTA imaging; defined as surface retinal neovascularization within the central foveal 1 mm diameter circle. 3 × 3 and 6 × 6 mm2 choriocapillaris and superficial and deep retinal capillary plexus (SCP and DCP) slabs were extracted to evaluate adjusted flow index (AFI) as a surrogate for blood flow. For choriocapillaris flow; total, subfoveal and extrafoveal AFI were assessed, while only total AFI was calculated for SCP and DCP. These findings were compared to healthy controls and eyes with PDR with no DFN.
Results
18 eyes of 18 patients were included in each of the 3 groups: healthy controls, PDR with and without DFN. Choriocapillaris AFI was significantly lower in PDR with DFN than healthy controls in all but the 6 × 6 mm2 extrafoveal AFI (p < 0.01). PDR with DFN also showed a lower AFI compared to eyes without DFN, but only in the 3 × 3 mm2 total and subfoveal AFI (p = 0.01). SCP and DCP AFI were not statistically significant.
Conclusions
Our findings suggest that choroidal hypoperfusion may be a potential driving factor for the development of DFN. The detection of these changes in the smaller scans of the total and subfoveal areas suggests a rather exaggerated and localized subfoveal distribution of ischaemia. Larger longitudinal studies are needed to explore the use of subfoveal choroidal AFI as a prognostic sign for DFN.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Available on reasonable request.
References
Mohamed Q, Gillies MC, Wong TY. Management of diabetic retinopathy: a systematic review. JAMA. 2007;298:902–16.
Bresnick GH, De Venecia G, Myers FL, Harris JA, Davis MD. Retinal ischemia in diabetic retinopathy. Arch Ophthalmol. 1975;93:1300–10.
Rajagopal J, Kamath AG, Kamath GG, Solanki N. Foveal neovascularisation in diabetic retinopathy: case report and review of literature. Int Ophthalmol. 2010;30:311–4.
Andreanos K, Rotsos T, Kymionis G, Koutsandrea C, Kotsolis A, Ladas I. Optical coherence tomography angiography of foveal neovascularization in diabetic retinopathy. Case Rep Ophthalmol. 2018;9:93–7.
Murakawa S, Hasegawa T, Koizumi H, Maruko I, Iida T. Foveal retinal neovascularization in proliferative diabetic retinopathy: assessment by optical coherence tomography angiography. Retina. 2017;37:e135–e7.
Kurz PA, Nguyan H, Cooney MJ. Bilateral foveal neovascularization in a patient with insulin-dependent diabetes mellitus. Arch Ophthalmol. 2003;121:1656–7.
Vaz-Pereira S, Proença H. Foveal neovascularization in type 2 diabetes. Ophthalmol Retin. 2019;3:566.
Seth A, Ghosh B, Bhambhwani V, Gupta A, Raina U. Foveal neovascularization in a patient with type 2 diabetes mellitus. Oman J Ophthalmol. 2017;10:255–6.
Tan B, Lim N-A, Tan R, Gan ATL, Chua J, Nusinovici S, et al. Combining retinal and choroidal microvascular metrics improves discriminative power for diabetic retinopathy. Br J Ophthalmol. 2023;107:993–9.
Venkatesh R, Reddy NG, Jayadev C, Mutalik D, Agrawal S, Mishra P, et al. Contributory factors for developing foveal neovascularization in proliferative diabetic retinopathy. Eur J Ophthalmol. 2022;32:3615–21.
Fawzi AA, Fayed AE, Linsenmeier RA, Gao J, Yu F. Improved macular capillary flow on optical coherence tomography angiography after panretinal photocoagulation for proliferative diabetic retinopathy. Am J Ophthalmol. 2019;206:217–27.
Fayed AE, Abdelbaki AM, El Zawahry OM, Fawzi AA. Optical coherence tomography angiography reveals progressive worsening of retinal vascular geometry in diabetic retinopathy and improved geometry after panretinal photocoagulation. PLoS One. 2020;14:e0226629.
Jia Y, Tan O, Tokayer J, Potsaid B, Wang Y, Liu JJ, et al. Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Opt Express. 2012;20:4710–25.
Fayed AE, Menten MJ, Kreitner L, Paetzold JC, Rueckert D, Bassily SM, et al. Retinal vasculature of different diameters and plexuses exhibit distinct vulnerability in varying severity of diabetic retinopathy. Eye. 2024:1-8.
Fayed AE, Gerges TK Optical coherence tomography angiography reveals paradoxically decreasing choroidal thickness & increasing blood flow in remitting Vogt Koyanagi Harada syndrome. Retina. 2022:10-1097.
Wilkinson C, Ferris FL, Klein RE, Lee PP, Agardh CD, Davis M, et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology. 2003;110:1677–82.
Campbell J, Zhang M, Hwang T, Bailey S, Wilson D, Jia Y, et al. Detailed vascular anatomy of the human retina by projection-resolved optical coherence tomography angiography. Sci Rep 2017;7:42201.
Ashraf M, Nesper PL, Jampol LM, Yu F, Fawzi AA. Statistical model of optical coherence tomography angiography parameters that correlate with severity of diabetic retinopathy. Invest Ophthalmol Vis Sci. 2018;59:4292–8.
Rasband WS Imagej, us national institutes of health, Bethesda, Maryland, USA. http://imagej.nih.gov/ij. 2011.
Cai J, Boulton M. The pathogenesis of diabetic retinopathy: old concepts and new questions. Eye. 2002;16:242–60.
Stitt A, Gardiner T, Archer D. Histological and ultrastructural investigation of retinal microaneurysm development in diabetic patients. Br J Ophthalmol. 1995;79:362–7.
Jian B, Jones PL, Li Q, Mohler ER III, Schoen FJ, et al. Matrix metalloproteinase-2 is associated with tenascin-C in calcific aortic stenosis. Am J Ophthalmol. 2001;159:321–7.
Haimovich B, Lipfert L, Brugge J, Shattil S. Tyrosine phosphorylation and cytoskeletal reorganization in platelets are triggered by interaction of integrin receptors with their immobilized ligands. J Biol Chem. 1993;268:15868–77.
Yanoff M. Ocular pathology of diabetes mellitus. Am J Ophthalmol. 1969;67:21–38.
Hidayat AA, Fine BS. Diabetic choroidopathy: light and electron microscopic observations of seven cases. Ophthalmology. 1985;92:512–22.
Shiragami C, Shiraga F, Matsuo T, Tsuchida Y, Ohtsuki H. Risk factors for diabetic choroidopathy in patients with diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol. 2002;240:436–42.
Borrelli E, Palmieri M, Viggiano P, Ferro G, Mastropasqua R. Photoreceptor damage in diabetic choroidopathy. Retina. 2020;40:1062–9.
Tang F, Luenam P, Ran AR, Quadeer AA, Raman R, Sen P, et al. Detection of diabetic retinopathy from ultra-widefield scanning laser ophthalmoscope images: a multicenter deep learning analysis. Ophthalmol Retin. 2021;5:1097–106.
Hayreh SS. Posterior ciliary artery circulation in health and disease the Weisenfeld lecture. Invest Ophthalmol Vis Sci. 2004;45:749–57.
Okamoto M, Matsuura T, Ogata N. Effects of panretinal photocoagulation on choroidal thickness and choroidal blood flow in patients with severe nonproliferative diabetic retinopathy. Retina. 2016;36:805–11.
Yiu G, Manjunath V, Chiu SJ, Farsiu S, Mahmoud TH. Effect of anti–vascular endothelial growth factor therapy on choroidal thickness in diabetic macular edema. Am J Ophthalmol. 2014;158:745–51.e2.
Iglicki M, González DP, Loewenstein A, Zur D. Next-generation anti-VEGF agents for diabetic macular oedema. Eye. 2022;36:273–7.
Author information
Authors and Affiliations
Contributions
RE and AEF formulated the study hypothesis and methodology. MAG, RE and RRF recruited the patients and obtained the imaging. AEF and MAG calculated the measurements. AEF, RRF and RE analysed the data. AEF drafted the manuscript. RE, MAG and RRF revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Watany Eye Hospital research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
An abstract for this manuscript has been presented at the 2024 ARVO annual meeting.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fayed, A.E., Gabra, M.A., Fikry, R.R. et al. Diabetic foveal neovascularization is associated with diminished subfoveal choroidal flow on optical coherence tomography angiography. Eye 39, 2169–2175 (2025). https://doi.org/10.1038/s41433-025-03806-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-025-03806-1