Abstract
Aim
To evaluate clinical outcomes, treatment intervals, and safety outcomes of brolucizumab (BRO) treatment in a national neovascular age-related macular degeneration (nAMD) database.
Methods
Multicentre, national, routine clinical care database study of nAMD eyes receiving ≥1 BRO injection. Demographics, visual acuity (VA) measured in logMAR letters, macular neovascularization (MNV) activity, number of injections, visit data and information on any adverse events were collected at baseline and at 3, 6, 9 and 12 months after BRO initiation for each patient/eye.
Results
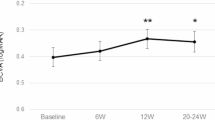
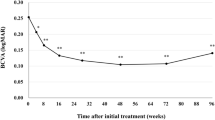
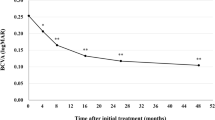
A total of 305 eyes received \(\ge \)1 BRO injection and 214 eyes (14% naïve, 86% switchers) completed \(\ge \)3 months follow-up. In switchers, the percentage of eyes extended to ≥8 week intervals at 3/6/9 months was 43.2%/45.7%/54.5% and to ≥10 week intervals was 12.9%/18.5%/13.6%, respectively. Eyes with VA ≥ 70 increased from 36% at baseline to 48% at 3 months and 50% at 9 months. MNV lesion activity status decreased from 94% (active/active-only SRF, 46/48%) at baseline to 56% (21/35%), 61% (23/38%), 76% (27/49%) and 65% (24/41%) at months 3/6/9 and 12, respectively. Adverse effects were observed in 6.5% of the treated eyes, being the most prevalent anterior uveitis (3.2%), vitritis (4.5%) and vasculitis (2.2%).
Conclusion
In this series BRO achieves an extension in the treatment intervals in half of the patients which require frequent reinjections (<8 weekly), reducing MNV activity in a third of this specific difficult-to-treat subgroup. The adverse event rates described are consistent with other cohorts and need to be considered to inform treatment decisions in case-by-case discussions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Dugel PU, Koh A, Ogura Y, Jaffe GJ, Schmidt-Erfurth U, Brown DM, et al. HAWK and HARRIER: phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;12:772–84.
Dugel PU, Singh RP, Koh A, Ogura Y, Weissgerber G, Gedif K, et al. HAWK and HARRIER: ninety-six-week outcomes from the phase 3 trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2021;128:89–99.
Bilgic A, Kodjikian L, de Ribot FM, Spitzer MS, Vasavada V, Gonzalez-Cortes JH, et al. Real-world experience with brolucizumab in neovascular age-related macular degeneration over 2 years: the REBA extension study. Graefe’s Arch Clin Exp Ophthalmol. 2024;262:1161–7.
Kim DJ, Kim DG, Kwak HD, Jang JY, Ji YS, Lee SH, et al. Long-term efficacy and safety of brolucizumab in neovascular age-related macular degeneration: a multicentre retrospective real-world study. Acta Ophthalmol. 2024;102:e1018–28.
Kim DJ, Jin KW, Han JM, Lee SH, Park YS, Lee JY, et al. Short-term safety and efficacy of intravitreal brolucizumab injections for neovascular age-related macular degeneration: a multicenter retrospective real-world study. Ophthalmologica. 2023;246:192–202.
Chakraborty D, Maiti A, Sheth JU, Mondal S, Boral S, Nandi K, et al. Brolucizumab in neovascular age-related macular degeneration - indian real-world experience: the braille study - fifty-two- week outcomes. Clinical Ophthalmol. 2022;16:4303–13.
MacCumber MW, Wykoff CC, Karcher H, Adiguzel E, Sinha SB, Vishwakarma S, et al. One-year brolucizumab outcomes in neovascular age-related macular degeneration from a large United States cohort in the IRIS® registry. Ophthalmology. 2023;130:937–46.
Witkin AJ, Hahn P, Murray TG, Arevalo JF, Blinder KJ, Choudhry N, et al. Occlusive retinal vasculitis following intravitreal brolucizumab. J Vitreoretin Dis. 2020;4:269–79.
Haug SJ, Hien DL, Uludag G, Ngoc TTT, Lajevardi S, Halim MS, et al. Retinal arterial occlusive vasculitis following intravitreal brolucizumab administration. Am J Ophthalmol Case Rep. 2020;18:100680.
Monés J, Srivastava SK, Jaffe GJ, Tadayoni R, Albini TA, Kaiser PK, et al. Risk of inflammation, retinal vasculitis, and retinal occlusion–related events with brolucizumab: post Hoc review of HAWK and HARRIER. Ophthalmology. 2021;128:1050–9.
Singer M, Albini TA, Seres A, Baumal CR, Parikh S, Gale R, et al. Clinical Characteristics and outcomes of eyes with intraocular inflammation after brolucizumab: post hoc analysis of HAWK and HARRIER. Ophthalmol Retin. 2022;6:97–108.
Khanani AM, Zarbin MA, Barakat MR, Albini TA, Kaiser PK, Guruprasad B, et al. Safety outcomes of brolucizumab in neovascular age-related macular degeneration: results from the Iris Registry and Komodo Healthcare Map. JAMA Ophthalmol. 2022;140:20–28.
Radke NV, Mohamed S, Brown RB, Ibrahim I, Chhablani J, Amin HV, et al. Review on the safety and efficacy of brolucizumab for neovascular age-related macular degeneration from major studies and real-world data. Asia Pac J Ophthalmol. 2023;12:168–83.
Writing Committee for the UK Age-Related Macular Degeneration EMR Users Group. The neovascular age-related macular degeneration database: multicenter study of 92,976 ranibizumab injections: report 1: visual acuity. Ophthalmology. 2014;121:1092–101. https://doi.org/10.1016/j.ophtha.2013.11.031.
Zarranz-Ventura J, Liew G, Johnston RL, Xing W, Akerele T, McKibbin M, et al. The neovascular age-related macular degeneration database: report 2: incidence, management, and visual outcomes of second-treated eyes. Ophthalmology. 2014;121:1966–75.
Rao P, Lum F, Wood K, Salman C, Burugapalli B, Hall R, et al. Real-world vision in age-related macular degeneration patients treated with single anti–VEGF drug type for 1 year in the Iris Registry. Ophthalmology. 2018;125:522–8.
Gillies MC, Walton R, Liong J, Arnold JJ, McAllister I, Morlet N, et al. Efficient capture of high-quality data on outcomes of treatment for macular diseases: the fight retinal blindness! project. Retina. 2014;34:188–95.
Zarranz-Ventura J, Parrado-Carrillo A, Nguyen V, Sararols L, Garay-Aramburu G, Puzo M, et al. Creation of a neovascular age-related macular degeneration national database using a web-based platform: fight retinal blindness Spain. Report 1: Visual Outcomes. Clin Exp Ophthalmol. 2022;50:312–24.
Schmidt-Erfurth U, Mulyukov Z, Gerendas BS, Reiter GS, Lorand D, Weissgerber G, et al. Therapeutic response in the HAWK and HARRIER trials using deep learning in retinal fluid volume and compartment analysis. Eye. 2023;37:1160–9.
Tadayoni R, Jaffe GJ, Holz FG, Schmidt-Erfurth U, Takahashi K, Cheung CMG, et al. Potential for treatment interval extension in eyes with namd disease activity post loading phase in HAWK and HARRIER. Ophthalmol Ther. 2023;12:2209–16.
Chaudhary V, Matonti F, Zarranz-Ventura J, Stewart MW. Impact of fluid compartments on functional outcomes for patients with neovascular age-related macular degeneration. Retina. 2022;42:589–606.
Kodjikian L, Parravano M, Clemens A, Dolz-Marco R, Holz FG, Munk MR, et al. Fluid as a critical biomarker in neovascular age-related macular degeneration management: literature review and consensus recommendations. Eye. 2021;35:2119–35.
Martin-Pinardel R, Izquierdo-Serra J, De Zanet S, Parrado-Carrillo A, Garay-Aramburu G, Puzo M, et al. Artificial intelligence-based fluid quantification and associated visual outcomes in a real-world, multicentre neovascular age-related macular degeneration national database. British J Ophthalmol. 2024;108:253–62.
Guymer RH, Markey CM, McAllister IL, Gillies MC, Hunyor AP, Arnold JJ, et al. Tolerating subretinal fluid in neovascular age-related macular degeneration treated with ranibizumab using a treat-and-extend regimen: FLUID study 24-month results. Ophthalmology. 2019;126:723–34.
Zarbin MA, MacCumber MW, Karcher H, Adiguzel E, Mayhook A, LaPrise A, et al. Real-world safety outcomes with brolucizumab in neovascular age-related macular degeneration: findings from the IRIS® Registry. Ophthalmol Ther. 2024;13:1357–68.
Bulirsch LM, Saßmannshausen M, Nadal J, Liegl R, Thiele S, Holz FG. Short-term real-world outcomes following intravitreal brolucizumab for neovascular AMD: SHIFT study. British J Ophthalmol. 2022;106:1288–94.
Enríquez AB, Baumal CR, Crane AM, Witkin AJ, Lally DR, Liang MC, et al. Early experience with brolucizumab treatment of neovascular age-related macular degeneration. JAMA Ophthalmol. 2021;139:441–8.
Nguyen HV, Li AS, Silva AR, Leng T. Ocular adverse events following intravitreal brolucizumab for neovascular age-related macular degeneration at a single tertiary care center. Eur J Ophthalmol. 2022;32:2747–51. https://doi.org/10.1177/11206721211059332.
Izquierdo-Serra J, Martin-Pinardel R, Moll-Udina A, Bernal-Morales C, Garay-Aramburu G, Sanchez-Monroy J, et al. Macular neovascularization type influence on anti-VEGF intravitreal therapy outcomes in age-related macular degeneration. Ophthalmol Retin. 2024;8:350–9.
Zarranz-Ventura J, Escobar-Barranco JJ, Gómez-Baldó L, Gallego-Pinazo R. Reasons for delayed Anti-VEGF treatment during COVID-19 lockdown and clinical impact in neovascular age-related macular degeneration. Ophthalmol Ther. 2023;12:2537–55.
Acknowledgements
The authors would like to thank Sonia de Orte Seemann and Cristina Martinez García from Syneos Health for their invaluable help in the overall management of this study; Guillem Masdeu, Miguel Moratalla, Judith Martinez, Daniel Garcés, Maria Jose Gonzalez and Antonia Soriano from Fundació Clínic per a la Recerca Biomedica (FCRB-IDIBAPS) for the administrative support with contracts and legal issues; and Laia Gomez-Baldo and Carmen Navarro from Novartis Spain for the external support during the development of this study.
Funding
This work was supported in part by an independent research collaboration from Novartis Pharmaceuticals. No member of Novartis participated in the analysis of the presented data.
Author information
Authors and Affiliations
Consortia
Contributions
Conception and design, obtained funding and overall responsibility: JZV; data collection: JZV, SMP, MFB, BPM, EC, CARF, CB, APC, IAV, CB, ECi, JAC, FJAP, PC, MPRT, MFR, and CBM; analysis, interpretation and writing: JZV, SMP, RMP, and CBM; manuscript revision: JZV, SMP, RMP, MFB, BPM, EC, CARF, APC, IAV, CB, ECi, JAC, FJAP, PC, MPRT, GSG, MFR, RPCM, and CBM.
Corresponding author
Ethics declarations
Competing interests
Javier Zarranz-Ventura is a grant holder for Novartis Pharmaceuticals, Bayer, Allergan/Abbvie and Roche, and a consultant for Novartis Pharmaceuticals, Bayer, Allergan/Abbvie, Alcon, Alimera Sciences, Bausch and Lomb, Brill Pharma, DORC, Preceyes, Roche, Topcon, and Zeiss.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zarranz-Ventura, J., Marías-Pérez, S., Martin-Pinardel, R. et al. Brolucizumab clinical and safety outcomes in a neovascular age-related macular degeneration national database: Fight Retinal Blindness Spain (FRB Spain). Eye 39, 2407–2414 (2025). https://doi.org/10.1038/s41433-025-03871-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-025-03871-6
This article is cited by
-
Establishing Expert Consensus on a Unified Decision Framework for Retina Patient Care
Ophthalmology and Therapy (2025)