Abstract
Background/objectives
To investigate if early detection of neovascular age-related macular degeneration (nAMD) in the second eye is associated with a reduced number of intravitreal treatments compared with the first eye using data from the EDNA/FASBAT trial.
Subjects/methods
Post-hoc analysis of 117 participants receiving standard clinical care during the FASBAT study. Assessments were at enrolment and at an average of 18.9 (SD = 10.2) and 30.5 (SD = 9.7) months in the first eye. Assessment of the second eye was made at the onset of nAMD (as participants were monitored for conversion) and at 12- and 24-months. The annualised injection rate for first eyes and the actual rate for second eyes are reported alongside visual acuity (VA) and optical coherence tomography (OCT) characteristics.
Results
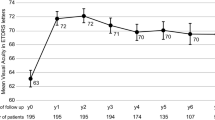
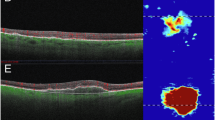
In second eyes the annualised number of treatments was lower both in year 1 (second eyes mean = 6.4, SD = 3.4 vs mean = 7.2, SD = 1.9 in first eyes) and in year 2 (second eyes mean = 5.8, SD = 2.9; versus mean = 6.2, SD = 2.7 in first eyes). Second eyes had better VA at the point of conversion to nAMD (mean = 74.1, SD = 9.9) compared with the first eyes at baseline (mean = 55.6, SD = 15.3) which was maintained until 24 months (second eye: mean = 73.6, SD = 9.9; first eye: mean = 53.2, SD = 19.3). Highly reflective material was detected less frequently in second eyes compared to first eyes at clinical visits.
Conclusions
Compared to first eyes, early detection of nAMD in the second eye is beneficial in terms of better maintenance of visual acuity, reduced intravitreal treatment burden, and improved anatomical findings after 2 years of treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
NICE. Age-related macular degeneration NICE guidelines. 2018. https://www.nice.org.uk/guidance/ng82/resources/agerelated-macular-degeneration-pdf-1837691334853.
Lee AY, Lee CS, Butt T, Xing W, Johnston RL, Chakravarthy U, et al. UK AMD EMR USERS GROUP REPORT V: Benefits of initiating ranibizumab therapy for neovascular AMD in eyes with vision better than 6/12. Br J Ophthalmol. 2015;99:1045–50.
Sivaprasad S, Banister K, Azuro-Blanco A, Goulao B, Cook JA, Hogg R, et al. Diagnostic accuracy of monitoring tests of fellow eyes in patients with unilateral neovascular age-related macular degeneration: early detection of neovascular age-related macular degeneration study. Ophthalmology. 2021;128:1736–47. https://doi.org/10.1016/j.ophtha.2021.07.025.
Gale RP, Airody A, Sivaprasad S, Hanson RLW, Allgar V, McKibbin M, et al. Improved structure and function in early detected second eye neovascular age-related macular degeneration; FASBAT/EDNA report 1. Ophthalmol Retina. 2024. https://doi.org/10.1016/j.oret.2023.12.012.
Norridge CFE, Gruszka-Goh MH, Mckibbin M, Henry P, Donachie J. The first report of age-related macular degeneration audit (AMD) patients starting treatment for neovascular AMD. 2023. The Royal College of Ophthalmologists, National Ophthalmology Database Audit. https://nodaudit.org.uk/node/4174.
Starr MR, Xu D, Boucher N, Saroj N, Patel LG, Ammar M, et al. Characterizing progression to neovascular amd in fellow eyes of patients treated with intravitreal anti-vegf injections. Ophthalmic Surg Lasers Imaging Retin. 2021;52:123–8.
Zarranz-Ventura J, Liew G, Johnston RL, Xing W, Akerele T, McKibbin M, et al. The neovascular age-related macular degeneration database: report 2: incidence, management, and visual outcomes of second treated eyes. Ophthalmology. 2014;121:1966–75.
Hanson RLW, Airody A, Sivaprasad S, Gale RP. Optical coherence tomography imaging biomarkers associated with neovascular age-related macular degeneration: a systematic review. Eye. 2022. https://www.nature.com/articles/s41433-022-02360-4.
Acknowledgements
The authors would like to acknowledge with thanks support from all members of the FASBAT study group across the UK, the EDNA study group, Augusto Azuara-Blanco, Networc UK, the Central Angiographic Resource Facility and the Research and Development team at York and Scarborough Teaching Hospitals NHS Foundation Trust.
Funding
Funding was received from Novartis Pharmaceuticals UK Ltd to RPG via an Investigator Initiated Trial (OAP030A2401T). The funding organisation had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Consortia
Contributions
Conception and Design: RPG, AA, MM, SS, UC. Data Collection: RPG, AA, SS, UC, MM, the EDNA Study group and the FASBAT Study group. Analysis and Interpretation: RPG, AA, RLH, ABM, SS, UC, MM, TP and the FASBAT Study group. Obtained Funding: RPG. Overall Responsibility: RPG for FASBAT and UC for EDNA.
Corresponding author
Ethics declarations
Competing interests
RPG: Received honorarium for advisory board meetings and speaker fees from Abbvie, Allergan, Alimera, Apellis, Bayer, Heidelberg Engineering, Lux Biosciences, Novartis, Notal Vision and Roche; Institutional grants from Novartis Pharmaceuticals UK Ltd and Bayer Pharmaceuticals. SS: Member of the Eye editorial board. Institutional research grants and received support for publication from Novartis, Bayer, Allergan/Abbvie and Boehringer Ingleheim; Attended advisory board meetings of Allergan, Apellis, Bayer, Boehringer Ingelheim, Heidelberg Engineering, Novartis, Oxurion, Oculis, Optos, Ophthea and Roche. UC: Personal fees from Novartis, Iveric, Roche, Boehringer Ingleheim, Apellis and Alimera; Other: Bayer, Gyroscope. TP: Member of the Eye editorial board.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hanson, R.L.W., Airody, A., Sivaprasad, S. et al. Early detection of neovascular age-related macular degeneration in the second eye reduces intravitreal treatment burden: FASBAT report 2. Eye 40, 493–497 (2026). https://doi.org/10.1038/s41433-025-04210-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-025-04210-5