Abstract
Background
There is a growing burden that highlights the need for ophthalmology research to improve treatment outcomes and patient care. By promoting transparency, reproducibility, and collaboration, Data Sharing Statements (DSS) provide a formal declaration of whether and how data can be accessed, reused, or shared, ensuring that research findings can be validated. This study examines the prevalence, content, and implementation of DSS in ophthalmology journals from 2018 to 2023.
Methods
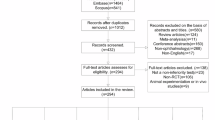
A comprehensive literature search in MEDLINE (PubMed) was conducted to evaluate clinical studies published in ten leading ophthalmology journals between January 1, 2018, and December 14, 2023. Data extraction was conducted via a standardized form in a masked, duplicate fashion. A hierarchical logistic regression was used to assess factors with potential influence on DSS inclusion. A qualitative analysis was performed to identify common themes in DSS.
Results
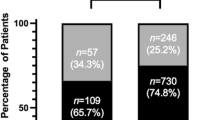
Of the 1385 articles analyzed, 326 (23.54%) included a DSS, with notable variability in DSS inclusion between journals. The British Journal of Ophthalmology had the highest DSS rate (152/228, 66.67%), while Investigative Ophthalmology and Visual Science had the lowest (4/103, 3.88%). Clinical trials were the most observed study design; however, DSS rates were low (183/667; 27%). Factors including study design, impact factor, funding type, and article access did not significantly influence DSS inclusion. The most common DSS themes were Conditional Data Availability (199/302, 65.89%) and Gatekeeper Role (87/302, 28.81%).
Conclusion
Implementing stronger mandates and adopting standardized data-sharing policies could address barriers to data-sharing practices, improving transparency and reproducibility in ophthalmology research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
All versions of our protocol, data, analysis scripts, and other study artifacts have been uploaded to Open Science Framework to promote open science practices and transparency (https://osf.io/jqmn8/).
References
Swenor B, Varadaraj V, Lee MJ, Whitson H, Ramulu P. World Health report on vision: aging implications for global vision and eye health. Innov Aging. 2020;4:807–8.
Lanza M, Incagli F, Ceccato C, Reffo ME, Mercuriali E, Parmeggiani F, et al. Quality of life, functioning and participation of children and adolescents with visual impairment: a scoping review. Res Dev Disabil. 2024;151:104772.
Kee QT, Abd Rahman MH, Mohamad Fadzil N, Mohammed Z, Shahar S. The impact of near visual impairment on instrumental activities of daily living among community-dwelling older adults in Selangor. BMC Res Notes. 2021;14:395.
CITI Program. The Benefits of Data Sharing and Its Contribution to Research Transparency. CITI Program. September 12, 2024. Accessed May 8, 2025. https://about.citiprogram.org/blog/the-benefits-of-data-sharing-and-its-contribution-to-research-transparency/
Committee on Strategies for Responsible Sharing of Clinical Trial Data, Board on Health Sciences Policy, Institute of Medicine. Guiding Principles for Sharing Clinical Trial Data. Washington: National Academies Press(US); 2015.
Institute of Medicine (US). Sharing Clinical Research Data: Workshop Summary. Washington (DC): National Academies Press (US); 2013 Mar 29. Available from: https://www.ncbi.nlm.nih.gov/books/NBK131772/https://doi.org/10.17226/18267
ICMJE. Accessed November 25, 2023. https://www.icmje.org/recommendations/browse/publishing-and-editorial-issues/clinical-trial-registration.html
Martone ME, Garcia-Castro A, VandenBos GR. Data sharing in psychology. Am Psychol. 2018;73:111–25.
Flanagin A, Curfman G, Bibbins-Domingo K. Data sharing and the growth of medical knowledge. JAMA. 2022;328:2398–9.
Nakayama LF, de Matos JCRG, Stewart IU, Mitchell WG, Martinez-Martin N, Regatieri C, et al. Retinal scans and data sharing: the privacy and scientific development equilibrium. Mayo Clin Proc Digit Health. 2023;1:67–74.
Ruamviboonsuk V, Thinkhamrop B, Kulvichit K, Tulvatana W. Data sharing implementation in top 10 ophthalmology journals in 2021. BMJ Open Ophthalmol. 2023;8. https://doi.org/10.1136/bmjophth-2023-001276
PRISMA 2020. PRISMA statement. Accessed September 12, 2024. https://www.prisma-statement.org/prisma-2020
The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Accessed September 12, 2024. https://www.equator-network.org/reporting-guidelines/strobe/
OSF. Accessed October 3, 2024. https://osf.io/jqmn8/
Statement DA. Data Availability and Policy. Accessed September 1, 2024. https://www.nature.com/eye/authors-and-referees/data-availability-and-policy
Authors. British Journal of Ophthalmology. Accessed September 1, 2024. https://bjo.bmj.com/pages/authors
Instructions for Authors–JOVD. J Vet Dent. 2023;40:94-98.
What IOVS Authors Should Know. Investig Ophthalmol Vis Sci. 2002;43:i–vi.
The Ocular Surface. Accessed September 12, 2024. https://www.sciencedirect.com/journal/the-ocular-surface/publish/guide-for-authors
Hendrickson F, Hossain MD, Duncan J, Dennis B, Hagood A, Gardner T, et al. Evaluating data sharing statements in leading emergency medicine journals: a mixed methods review. Am J Emerg Med. 2025;89:159–68.
Major J, Wild K, Elfar A, Dennis B, Oldham E, Hagood A, et al. Evaluating data-sharing policies and author compliance in leading orthopaedic journals. J Bone Joint Surg Am. 2025;107:1024–33.
Moore J, Nguyen K, Dennis B, Chaudhry M, Elfar A, Oldham E, et al. Assessing the prevalence, quality and compliance of data-sharing statements in gastroenterology publications: a cross-sectional analysis. BMJ Open. 2025;15:e092490.
Pham M, Bagaruka E, Oldham E, Chaudhry M, Duncan J, Gardner T, et al. The state of data sharing in rheumatology: a systematic review of top journal practices. Rheumatology (Oxford). 2025;64:4113–24.
Gabelica M, Bojčić R, Puljak L. Many researchers were not compliant with their published data sharing statement: a mixed-methods study. J Clin Epidemiol. 2022;150:33–41. https://doi.org/10.1016/j.jclinepi.2022.05.019.
McDonald L, Schultze A, Simpson A, Graham S, Wasiak R, Ramagopalan SV. A review of data sharing statements in observational studies published in the BMJ: a cross-sectional study. F1000Res. 2017;6. https://doi.org/10.12688/f1000research.12673.2
Gim N, Wu Y, Blazes M, Lee CS, Wang RK, Lee AY. A clinician’s guide to sharing data for ai in ophthalmology. Investig Ophthalmol Vis Sci. 2024;65:21.
Tom E, Keane PA, Blazes M, Pasquale LR, Chiang MF, Lee AY, et al. Protecting data privacy in the age of AI-enabled ophthalmology. Transl Vis Sci Technol. 2020;9:36.
Wu C, Yang X, Gilkes EG, Cui H, Choi J, Sun N, et al. De-identification and obfuscation of gender attributes from retinal scans. In Proceedings Clinical Image-Based Procedures, Fairness of AI in Medical Imaging, and Ethical and Philosophical Issues in Medical Imaging. Lecture notes in computer science. Switzerland: Springer Nature; 2023.
Wilkinson MD, Dumontier M, Aalbersberg IJ, Appleton G, Axton M, Baak A, et al. The FAIR guiding principles for scientific data management and stewardship. Sci Data. 2016;3:160018.
Center for Open Science. TOP Guidelines. Accessed July 23, 2024. https://www.cos.io/initiatives/top-guidelines
NOT-OD-21-013: Final NIH Policy for Data Management and Sharing. Accessed May 11, 2025. https://grants.nih.gov/grants/guide/notice-files/NOT-OD-21-013.html
Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev. 2019;10:ED000142.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work and for the final approval of the version to be published. Authors MC, CB, EO, EP were involved in drafting/revision of the article for content, including medical writing for content; curation of data; screening and extraction of data; study concept or design; analysis or interpretation of the data; writing of the original draft and copy edits. Authors MC, EP, AE, and MV were involved in drafting/revision of the article for content, including medical writing for content; study concept or design and conceptualization; analysis or interpretation of data, and curation of data. All authors are to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
MV reports receipt of funding from the National Institute on Drug Abuse, the National Institute on Alcohol Abuse and Alcoholism, the U.S. Office of Research Integrity, Oklahoma Center for Advancement of Science and Technology, and internal grants from Oklahoma State University Center for Health Sciences—all outside of the present work. AF reports receipt of funding from the Center for Integrative Research on Childhood Adversity, the Oklahoma Shared Clinical and Translational Resources, and internal grants from Oklahoma State University and Oklahoma State University Center for Health Sciences—all outside of the present work. All other authors have nothing to report.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chaudhry, M., Bratten, C., Paul, E. et al. Evaluating the prevalence and application of data sharing statements in high-impact ophthalmology journals. Eye 40, 540–549 (2026). https://doi.org/10.1038/s41433-025-04214-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-025-04214-1