Abstract
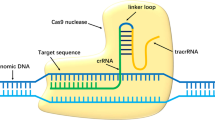
Corneal neovascularization is a sight-threatening condition for which current treatments such as anti-VEGF agents are limited by invasiveness and side effects. We present the first non-viral, CRISPR/Cas9-based gene therapy delivered via topical eye drops that penetrates the cornea and inhibits pathological neovascularization. Cas9 ribonucleoproteins (RNPs) targeting the Vegfa gene were complexed with a liposomal carrier (lipofectamine) and administered to mice after alkali burn injury to the cornea. This approach achieved approximately 2% gene editing at the Vegfa locus in vivo, which significantly reduced local VEGF-A expression. Consequently, treated corneas showed markedly decreased macrophage infiltration and robust suppression of both hemangiogenesis and lymphangiogenesis compared to untreated controls. These findings demonstrate that even modest in vivo gene editing can yield a strong therapeutic effect, highlighting a clinically relevant strategy for controlling corneal angiogenesis. Our study introduces a feasible and safe topical CRISPR therapy for corneal diseases, offering a potential alternative to invasive or virus-based gene delivery methods.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and its extended data and supplemental information files. The datasets generated during or analyzed in the current study are available from the corresponding author upon reasonable request.
References
Taylor AW. Ocular immune privilege. Eye. 2009;23:1885–9.
Cursiefen C, Chen L, Dana MR, Streilein JW. Corneal lymphangiogenesis: evidence, mechanisms, and implications for corneal transplant immunology. Cornea. 2003;22:273–81.
Chames P, Van Regenmortel M, Weiss E, Baty D. Therapeutic antibodies: successes, limitations and hopes for the future. Br J Pharm. 2009;157:220–33.
Roshandel D, Eslani M, Baradaran-Rafii A, Cheung AY, Kurji K, Jabbehdari S, et al. Current and emerging therapies for corneal neovascularization. Ocul Surf. 2018;16:398–414.
Bachmann B, Taylor RS, Cursiefen C. Corneal neovascularization as a risk factor for graft failure and rejection after keratoplasty: an evidence-based meta-analysis. Ophthalmology. 2010;117:1300–5.e7.
Abdelfattah NS, Amgad M, Zayed AA, Hussein H, Abd El-Baky N. Molecular underpinnings of corneal angiogenesis: advances over the past decade. Int J Ophthalmol. 2016;9:768–79.
Philipp W, Speicher L, Humpel C. Expression of vascular endothelial growth factor and its receptors in inflamed and vascularized human corneas. Investig Ophthalmol Vis Sci. 2000;41:2514–22.
Cursiefen C, Chen L, Borges LP, Jackson D, Cao J, Radziejewski C, et al. VEGF-A stimulates lymphangiogenesis and hemangiogenesis in inflammatory neovascularization via macrophage recruitment. J Clin Investig. 2004;113:1040–50.
Kim SW, Ha BJ, Kim EK, Tchah H, Kim T-i. The effect of topical bevacizumab on corneal neovascularization. Ophthalmology. 2008;115:e33–e8.
Benayoun Y, Adenis JP, Casse G, Forte R, Robert PY. Effects of subconjunctival bevacizumab on corneal neovascularization: results of a prospective study. Cornea. 2012;31:937–44.
Sarah B, Ibtissam H, Mohammed B, Hasna S, Abdeljalil M. Intrastromal injection of bevacizumab in the management of corneal neovascularization: about 25 eyes. J Ophthalmol. 2016;2016.
Ferrari G, Dastjerdi MH, Okanobo A, Cheng SF, Amparo F, Nallasamy N, et al. Topical ranibizumab as a treatment of corneal neovascularization. Cornea. 2013;32:992–7.
Mergen B, Dogan C. Intrastromal aflibercept for the treatment of corneal neovascularization. Investig Ophthalmol Vis Sci. 2022;63:908–A0272-908 – A.
Avery RL, Castellarin AA, Steinle NC, Dhoot DS, Pieramici DJ, See R, et al. Systemic pharmacokinetics and pharmacodynamics of intravitreal aflibercept, bevacizumab, and ranibizumab. Retina. 2017;37:1847–58.
Feizi S, Azari AA, Safapour S. Therapeutic approaches for corneal neovascularization. Eye Vis. 2017;4:28.
Cheng HC, Yeh SI, Tsao YP, Kuo PC. Subconjunctival injection of recombinant AAV-angiostatin ameliorates alkali burn induced corneal angiogenesis. Mol Vis. 2007;13:2344–52.
Lai LJ, Xiao X, Wu JH. Inhibition of corneal neovascularization with endostatin delivered by adeno-associated viral (AAV) vector in a mouse corneal injury model. J Biomed Sci. 2007;14:313–22.
Uehara H, Zhang X, Pereira F, Narendran S, Choi S, Bhuvanagiri S, et al. Start codon disruption with CRISPR/Cas9 prevents murine Fuchs’ endothelial corneal dystrophy. eLife. 2021;10.
Hanlon KS, Kleinstiver BP, Garcia SP, Zaborowski MP, Volak A, Spirig SE, et al. High levels of AAV vector integration into CRISPR-induced DNA breaks. Nat Commun. 2019;10:4439.
Li C, Samulski RJ. Engineering adeno-associated virus vectors for gene therapy. Nat Rev Genet. 2020;21:255–72.
Wang D, Zhang F, Gao G. CRISPR-based therapeutic genome editing: strategies and in vivo delivery by AAV vectors. Cell. 2020;181:136–50.
Nguyen GN, Everett JK, Kafle S, Roche AM, Raymond HE, Leiby J, et al. A long-term study of AAV gene therapy in dogs with hemophilia A identifies clonal expansions of transduced liver cells. Nat Biotechnol. 2021;39:47–55.
Kim K, Park SW, Kim JH, Lee SH, Kim D, Koo T, et al. Genome surgery using Cas9 ribonucleoproteins for the treatment of age-related macular degeneration. Genome Res. 2017;27:419–26.
Chen G, Abdeen AA, Wang Y, Shahi PK, Robertson S, Xie R, et al. A biodegradable nanocapsule delivers a Cas9 ribonucleoprotein complex for in vivo genome editing. Nat Nanotechnol. 2019;14:974–80.
Wang Y, Shahi PK, Xie R, Zhang H, Abdeen AA, Yodsanit N, et al. A pH-responsive silica–metal–organic framework hybrid nanoparticle for the delivery of hydrophilic drugs, nucleic acids, and CRISPR-Cas9 genome-editing machineries. J Control Release. 2020;324:194–203.
Jang HK, Jo DH, Lee SN, Cho CS, Jeong YK, Jung Y, et al. High-purity production and precise editing of DNA base editing ribonucleoproteins. Sci Adv. 2021;7.
Banskota S, Raguram A, Suh S, Du SW, Davis JR, Choi EH, et al. Engineered virus-like particles for efficient in vivo delivery of therapeutic proteins. Cell. 2022;185:250–65.e16.
Yin D, Ling S, Wang D, Dai Y, Jiang H, Zhou X, et al. Targeting herpes simplex virus with CRISPR-Cas9 cures herpetic stromal keratitis in mice. Nat Biotechnol. 2021;39:567–77.
Wei A, Yin D, Zhai Z, Ling S, Le H, Tian L, et al. In vivo CRISPR gene editing in patients with herpetic stromal keratitis. Mol Ther. 2023;31:3163–75.
Park J, Lim K, Kim JS, Bae S. Cas-analyzer: an online tool for assessing genome editing results using NGS data. Bioinformatics. 2017;33:286–8.
Giacomini C, Ferrari G, Bignami F, Rama P. Alkali burn versus suture-induced corneal neovascularization in C57BL/6 mice: an overview of two common animal models of corneal neovascularization. Exp Eye Res. 2014;121:1–4.
Anderson C, Zhou Q, Wang S. An alkali-burn injury model of corneal neovascularization in the mouse. J Vis Exp. 2014.
Luisi J, Lin JL, Karediya N, Kraft ER, Sharifi A, Schmitz-Brown ME, et al. Concentration-associated pathology of alkali burn in a mouse model using anterior segment optical coherence tomography with angiography. Exp Eye Res. 2022;223:109210.
Lu P, Li L, Liu G, Baba T, Ishida Y, Nosaka M, et al. Critical role of TNF-α-induced macrophage VEGF and iNOS production in the experimental corneal neovascularization. Investig Ophthalmol Vis Sci. 2012;53:3516–26.
Bai Y, Jiao X, Hu J, Xue W, Zhou Z, Wang W. WTAP promotes macrophage recruitment and increases VEGF secretion via N6-methyladenosine modification in corneal neovascularization. Biochim Biophys Acta Mol Basis Dis. 2023;1869:166708.
Kim S, Kim D, Cho SW, Kim J, Kim J-S. Highly efficient RNA-guided genome editing in human cells via delivery of purified Cas9 ribonucleoproteins. Genome Res. 2014;24:1012–9.
Fu YW, Dai XY, Wang WT, Yang ZX, Zhao JJ, Zhang JP, et al. Dynamics and competition of CRISPR-Cas9 ribonucleoproteins and AAV donor-mediated NHEJ, MMEJ and HDR editing. Nucleic Acids Res. 2021;49:969–85.
Amador C, Shah R, Ghiam S, Kramerov AA, Ljubimov AV. Gene therapy in the anterior eye segment. Curr Gene Ther. 2022;22:104–31.
Corliss BA, Azimi MS, Munson JM, Peirce SM, Murfee WL. Macrophages: an inflammatory link between angiogenesis and lymphangiogenesis. Microcirculation. 2016;23:95–121.
Giannaccare G, Pellegrini M, Bovone C, Spena R, Senni C, Scorcia V, et al. Anti-VEGF treatment in corneal diseases. Curr Drug Targets. 2020;21:1159–80.
Bakunowicz-Łazarczyk A, Urban B. Assessment of therapeutic options for reducing alkali burn-induced corneal neovascularization and inflammation. Adv Med Sci. 2016;61:101–12.
Mukwaya A, Mirabelli P, Lennikov A, Thangavelu M, Jensen L, Peebo B, et al. Repeat corneal neovascularization is characterized by more aggressive inflammation and vessel invasion than in the initial phase. Investig Ophthalmol Vis Sci. 2019;60:2990–3001.
Bastola P, Song L, Gilger BC, Hirsch ML. Adeno-associated virus mediated gene therapy for corneal diseases. Pharmaceutics. 2020;12:767.
Miyadera K, Conatser L, Llanga TA, Carlin K, O’Donnell P, Bagel J, et al. Intrastromal gene therapy prevents and reverses advanced corneal clouding in a canine model of mucopolysaccharidosis I. Mol Ther. 2020;28:1455–63.
Su W, Sun S, Tian B, Tai PWL, Luo Y, Ko J, et al. Efficacious, safe, and stable inhibition of corneal neovascularization by AAV-vectored anti-VEGF therapeutics. Mol Ther Methods Clin Dev. 2021;22:107–21.
Chew WL, Tabebordbar M, Cheng JK, Mali P, Wu EY, Ng AH, et al. A multifunctional AAV-CRISPR-Cas9 and its host response. Nat Methods. 2016;13:868–74.
Wang D, Tai PWL, Gao G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat Rev Drug Discov. 2019;18:358–78.
Saika S, Ikeda K, Yamanaka O, Flanders KC, Nakajima Y, Miyamoto T, et al. Therapeutic effects of adenoviral gene transfer of bone morphogenic protein-7 on a corneal alkali injury model in mice. Lab Investig. 2005;85:474–86.
Mohan RR, Sinha S, Tandon A, Gupta R, Tovey JC, Sharma A. Efficacious and safe tissue-selective controlled gene therapy approaches for the cornea. PLoS One. 2011;6:e18771.
Gupta S, Rodier JT, Sharma A, Giuliano EA, Sinha PR, Hesemann NP, et al. Targeted AAV5-Smad7 gene therapy inhibits corneal scarring in vivo. PLoS ONE. 2017;12:e0172928.
Mohan RR, Martin LM, Sinha NR. Novel insights into gene therapy in the cornea. Exp Eye Res. 2021;202:108361.
Ge Q, Zhang H, Hou J, Wan L, Cheng W, Wang X, et al. VEGF secreted by mesenchymal stem cells mediates the differentiation of endothelial progenitor cells into endothelial cells via paracrine mechanisms. Mol Med Rep. 2018;17:1667–75.
Ogunshola OO, Antic A, Donoghue MJ, Fan SY, Kim H, Stewart WB, et al. Paracrine and autocrine functions of neuronal vascular endothelial growth factor (VEGF) in the central nervous system. J Biol Chem. 2002;277:11410–5.
Maruyama K, Ii M, Cursiefen C, Jackson DG, Keino H, Tomita M, et al. Inflammation-induced lymphangiogenesis in the cornea arises from CD11b-positive macrophages. J Clin Investig. 2005;115:2363–72.
Detry B, Blacher S, Erpicum C, Paupert J, Maertens L, Maillard C, et al. Sunitinib inhibits inflammatory corneal lymphangiogenesis. Investig Ophthalmol Vis Sci. 2013;54:3082–93.
Cursiefen C, Cao J, Chen L, Liu Y, Maruyama K, Jackson D, et al. Inhibition of hemangiogenesis and lymphangiogenesis after normal-risk corneal transplantation by neutralizing VEGF promotes graft survival. Investig Ophthalmol Vis Sci. 2004;45:2666–73.
Acknowledgements
This research was supported by Seoul National University Hospital Research Grant (18-2023-0010), National Research Council of Science & Technology (NST) grant by the Korea government (MSIT) (GTL24021-000 to J.H.K.), grants from the National Research Foundation of Korea (NRF) (2022M3A9E4017127, RS-2023-00260351, and RS-2024-00467177 to J.H.K.), and Korean Fund for Regenerative Medicine(KFRM) grant (21A0202L1-11 to S.B.).
Author information
Authors and Affiliations
Contributions
Seok Jae Lee: in vivo experiments, Writing-Original draft. Bae-Geun Nam: in vitro experiments, Writing-Reviewing and Editing. Sung-A Hong: in vitro experiments, Writing-Reviewing and Editing. Dong Hyun Jo: Writing-Reviewing and Editing. Sang-Mok Lee: Supervision, Writing-Reviewing, and Editing. Sangsu Bae: Funding acquisition, Supervision, Writing- Reviewing and Editing. Jeong Hun Kim: Conceptualization, Funding acquisition, Supervision, Writing- Reviewing and Editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflicts of interest in relation to this article.
Ethics approval and consent to participate
All animal procedures and related protocols were conducted in accordance with the guidelines of the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research and were approved by the Seoul National University Animal Care and Use Committee. This study was conducted exclusively on mice; therefore, obtaining consent to participate was not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, S.J., Nam, BG., Hong, SA. et al. Topical application of Cas9 ribonucleoproteins inhibits corneal neovascularization in a mouse model of alkali burn injury. Gene Ther (2026). https://doi.org/10.1038/s41434-026-00607-3
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41434-026-00607-3