Abstract
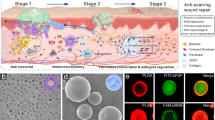
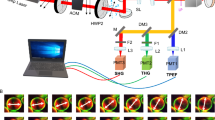
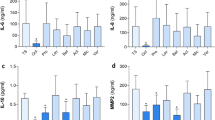
Effective scar control requires selectively suppressing late-stage fibrosis without compromising early wound closure. We developed a localized, time-staged delivery system. A poly-(HA-GMA) hydrogel serves as a short-term depot, loaded with AAV8-sTβRII and applied directly along the wound margin. Materials characterization showed a water-rich porous network that rapidly imbibes and releases vector primarily by diffusion. In vivo, the hydrogel naturally degrades in ~3 days, enabling local enrichment in early healing without excessive retention. In a mouse full-thickness skin-wound model, this approach achieved efficient transduction of the cutaneous and fascial layers while markedly reducing hepatic exposure. From postoperative day 6 onward it accelerated closure and produced a thinner dermis, more orderly collagen organization, and a lower collagen area fraction. Mechanistically, Flag-sTβRII was detected within scar tissue. Phospho-Smad2/3 and α-SMA were reduced, whereas total Smad2/3 was largely unchanged, indicating that inhibition occurs at the activation step of the TGF-β/Smad pathway. Moreover, adding exogenous TGF-β1 reversed the macroscopic and histological benefits, strengthening the evidence for pathway specificity. Compared with direct intradermal injection, hydrogel delivery simultaneously increased local expression and limited systemic spillover. Using the AAV8 capsid provided the most favorable balance—high in skin, low in liver. Safety readouts—including body weight, serum transaminases, and histology of major organs—showed no abnormalities. To our knowledge, the “HA-GMA × AAV8-sTβRII” strategy precisely aligns pathway antagonism with the escalation phase of fibrosis, yielding improvements from molecular and cellular phenotypes to tissue remodeling and healing. It offers a generalizable, materials–biology integrated platform for anti-fibrotic gene therapy at the wound edge.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All data generated and/or analyzed during this study are included in this published article and its supplementary information files. The raw data supporting the findings of this study (including, but not limited to, original Western blot images, histological images, and quantitative analysis data) are available from the corresponding author upon reasonable request.
References
Jeschke MG, Wood FM, Middelkoop E, Bayat A, Teot L, Ogawa R, et al. Scars. Nat Rev Dis Prim. 2023;9:64.
Deng Z, Fan T, Xiao C, Tian H, Zheng Y, Li C, et al. TGF-β signaling in health, disease, and therapeutics. Signal Transduct Target Ther. 2024;9:61.
Massagué J, Sheppard D. TGF-β signaling in health and disease. Cell. 2023;186:4007–37.
Martin P, Pardo-Pastor C, Jenkins RG, Rosenblatt J. Imperfect wound healing sets the stage for chronic diseases. Science. 2024;386:eadp2974.
Reiss UM, Davidoff AM, Tuddenham EGD, Chowdary P, McIntosh J, Muczynski V, et al. Sustained clinical benefit of AAV gene therapy in severe hemophilia B. N Engl J Med. 2025;392:2226–34.
Ponomarev A, Ganiev I, Aimaletdinov A, Mansurova M, Titova A, Rizvanov A, et al. Evaluation of the efficacy of transglutaminase 1 gene delivery by adeno-associated virus into rat and pig skin and safety of ARCI gene therapy. Int J Mol Sci. 2025;26:9976.
Wang JH, Gessler DJ, Zhan W, Gallagher TL, Gao G. Adeno-associated virus as a delivery vector for gene therapy of human diseases. Signal Transduct Target Ther. 2024;9:78.
McCarty DM, Monahan PE, Samulski RJ. Self-complementary recombinant adeno-associated virus (scAAV) vectors promote efficient transduction independently of DNA synthesis. Gene Ther. 2001;8:1248–54.
Correa-Gallegos D, Ye H, Dasgupta B, Sardogan A, Kadri S, Kandi R, et al. CD201(+) fascia progenitors choreograph injury repair. Nature. 2023;623:792–802.
Dabelsteen S, Pallesen EMH, Marinova IN, Nielsen MI, Adamopoulou M, Rømer TB, et al. Essential functions of glycans in human epithelia dissected by a CRISPR-Cas9-engineered human organotypic skin model. Dev Cell. 2020;54:669–84.
Meyer NL, Chapman MS. Adeno-associated virus (AAV) cell entry: structural insights. Trends Microbiol. 2022;30:432–51.
Kato M, Ishikawa S, Shen Q, Du Z, Katashima T, Naito M, et al. In situ-formable, dynamic crosslinked poly(ethylene glycol) carrier for localized adeno-associated virus infection and reduced off-target effects. Commun Biol. 2023;6:508.
Li J, Celiz AD, Yang J, Yang Q, Wamala I, Whyte W, et al. Tough adhesives for diverse wet surfaces. Science. 2017;357:378–81.
Li J, Mooney DJ Designing hydrogels for controlled drug delivery. Nat Rev Mater. 2016;1:16071.
Weng L, Gouldstone A, Wu Y, Chen W. Mechanically strong double network photocrosslinked hydrogels from N,N-dimethylacrylamide and glycidyl methacrylated hyaluronan. Biomaterials. 2008;29:2153–63.
Spearman BS, Agrawal NK, Rubiano A, Simmons CS, Mobini S, Schmidt CE. Tunable methacrylated hyaluronic acid-based hydrogels as scaffolds for soft tissue engineering applications. J Biomed Mater Res A. 2020;108:279–91.
Mascarenhas J, Migliaccio AR, Kosiorek H, Bhave R, Palmer J, Kuykendall A, et al. A phase Ib trial of AVID200, a TGFβ 1/3 Trap, in patients with myelofibrosis. Clin Cancer Res. 2023;29:3622–32.
Schirmer L, Atallah P, Freudenberg U, Werner C. Chemokine-capturing wound contact layer rescues dermal healing. Adv Sci. 2021;8:e2100293.
Liu X, Zhuang Y, Huang W, Wu Z, Chen Y, Shan Q, et al. Interventional hydrogel microsphere vaccine as an immune amplifier for activated antitumour immunity after ablation therapy. Nat Commun. 2023;14:4106.
Fajardo AR, Fávaro SL, Rubira AF, Muniz EC. Dual-network hydrogels based on chemically and physically crosslinked chitosan/chondroitin sulfate. React Funct Polym. 2013;73:1662–71.
Penaud-Budloo M, François A, Clément N, Ayuso E. Pharmacology of recombinant adeno-associated virus production. Mol Ther Methods Clin Dev. 2018;8:166–80.
Wang H, Li R, Sadekar S, Kamath AV, Shen BQ. A novel approach to quantitate biodistribution and transduction of adeno-associated virus gene therapy using radiolabeled AAV vectors in mice. Mol Ther Methods Clin Dev. 2024;32:101326.
Correa S, Grosskopf AK, Lopez Hernandez H, Chan D, Yu AC, Stapleton LM, et al. Translational applications of hydrogels. Chem Rev. 2021;121:11385–457.
Richbourg NR, Peppas NA. The swollen polymer network hypothesis: quantitative models of hydrogel swelling, stiffness, and solute transport. Prog Polym Sci. 2020;105:101243.
Wu F, Pang Y, Liu J. Swelling-strengthening hydrogels by embedding with deformable nanobarriers. Nat Commun. 2020;11:4502.
Kim D, Lu N, Ma R, Kim Y, Kim R, Wang S, et al. Epidermal electronics. Science. 2011;333:838–43.
Chu M, Nguyen T, Pandey V, Zhou Y, Pham HN, Bar-Yoseph R, et al. Respiration rate and volume measurements using wearable strain sensors. npj Digit Med. 2019;2:8.
Jensen KH, Kim W, Chang S. Dynamics of water imbibition through hydrogel-coated capillary tubes. Phys Rev Fluids. 2022;7:64301.
Huang X, Brazel CS. On the importance and mechanisms of burst release in matrix-controlled drug delivery systems. J Control Release. 2001;73:121–36.
Ashcroft GS, Dodsworth J, van, Boxtel E, Tarnuzzer RW, Horan MA, et al. Estrogen accelerates cutaneous wound healing associated with an increase in TGF-beta1 levels. Nat Med. 1997;3:1209–15.
Butzelaar L, Ulrich MM, Mink van der Molen AB, Niessen FB, Beelen RH. Currently known risk factors for hypertrophic skin scarring: a review. J Plast Reconstr Aesthet Surg. 2016;69:163–9.
Lim CH, Sun Q, Ratti K, Lee SH, Zheng Y, Takeo M, et al. Hedgehog stimulates hair follicle neogenesis by creating inductive dermis during murine skin wound healing. Nat Commun. 2018;9:4903.
Nuutila K, Eriksson E. Moist wound healing with commonly available dressings. Adv Wound Care. 2021;10:685–98.
Chen JH, Zhan LJ, Lv CM, Teng JB, Cao CY. Advances and challenges in adeno-associated virus gene therapy applications of localized delivery strategies. Curr Med Sci. 2025;45:683–98.
Walkey CJ, Snow KJ, Bulcha J, Cox AR, Martinez AE, Ljungberg MC, et al. A comprehensive atlas of AAV tropism in the mouse. Mol Ther. 2025;33:1282–99.
Yang Y, Chu C, Liu L, Wang C, Hu C, Rung S, et al. Tracing immune cells around biomaterials with spatial anchors during large-scale wound regeneration. Nat Commun. 2023;14:5995.
Rajendran V, Ramesh P, Dai R, Kalgudde Gopal S, Ye H, Machens HG, et al. Therapeutic silencing of p120 in fascia fibroblasts ameliorates tissue repair. J Investig Dermatol. 2023;143:854–63.
Greig JA, Martins KM, Breton C, Lamontagne RJ, Zhu Y, He Z, et al. Integrated vector genomes may contribute to long-term expression in primate liver after AAV administration. Nat Biotechnol. 2024;42:1232–42.
Acknowledgements
This work is supported by The central government guides the special funds for the development of local science and technology (2024BSB012) and the National Natural Science Foundation of China (81772833) and Hubei Provincial Natural Science Foundation of China (2024AFB585). The authors would like to thank Shuchu Shen for helping with the modification of the figure.
Author information
Authors and Affiliations
Contributions
JC conceived the study and contributed to methodology, data curation, and writing of the original draft. LZ, JD, and TW contributed to software and writing of the original draft. ZM., QW, and JY contributed to visualization, software, and investigation. XH contributed to data curation. YL contributed to supervision, software, and resources. XS and CC contributed to writing—review and editing, supervision, and funding acquisition. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
All animal experiments in this study were approved by the Animal Ethics Committee of China Three Gorges University (Approval No. 2024020H1). All experimental procedures were performed in strict accordance with relevant institutional and national guidelines and regulations for the care and use of laboratory animals. All methods were carried out in accordance with relevant guidelines and regulations.
Competing interests
The authors have declared that no competing interests exist.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Zhan, L., Duan, J. et al. The poly-(HA-GMA) hydrogel carrying AAV8-sTβRII alleviates scar formation in mice skin wound healing by inhibiting fibrosis. Gene Ther (2026). https://doi.org/10.1038/s41434-026-00608-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41434-026-00608-2