Abstract
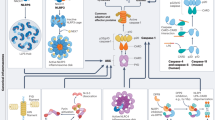
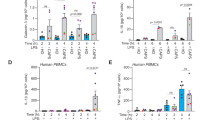
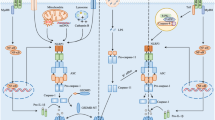
Glioblastoma (GBM) is the deadliest malignant brain tumor, with a survival of less than 14 months after diagnosis. The highly invasive nature of GBM makes total surgical resection challenging, leading to tumor recurrence and declined survival. The heterocellular composition of the GBM reprograms its microenvironment, favoring tumor growth, proliferation, and migration. The innate immune cells in the GBM tumor microenvironment, including microglia, astrocytes, and macrophages, express pattern recognition receptors such as NLRs (Nucleotide-binding domain and leucine-rich repeat-containing) that sense pathogen- and damage-associated molecular patterns initiating inflammation. Upon activation, NLRP3 promotes inflammation by NLRP3 inflammasome formation. Auto-proteolytic cleavage and activation of Caspase-1 within the inflammasome leads to caspase-1-mediated cleavage, activation, and conversion of pro-IL-1ß and pro-IL-18 to IL-1ß and IL-18, leading to pyroptosis. In contrast, NLRP12 downregulates inflammatory responses in microglia and macrophages by regulating the NF-κB pathway. NLRP3 and NLRP12 have been implicated in the disease pathophysiology of several cancers with cell-context-dependent, pro- or anti-tumorigenic roles. In this review, we discuss the current literature on the mechanistic roles of NLRP3 and NLRP12 in GBM and the gaps in the scientific literature in the context of GBM pathophysiology with potential for targeted therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, et al. The 2021 WHO classification of tumors of the Central Nervous System: a summary. Neuro Oncol. 2021;23:1231–51.
Iacob G, Dinca EB. Current data and strategy in glioblastoma multiforme. J Med Life. 2009;2:386–93.
Eckel-Passow JE, Lachance DH, Molinaro AM, Walsh KM, Decker PA, Sicotte H, et al. Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N. Engl J Med. 2015;372:2499–508.
Luo H, Shusta EV. Blood-brain barrier modulation to improve glioma drug delivery. Pharmaceutics. 2020;12:1085.
Holland EC. Glioblastoma multiforme: the terminator. Proc Natl Acad Sci USA. 2000;97:6242–4.
Paw I, Carpenter RC, Watabe K, Debinski W, Lo HW. Mechanisms regulating glioma invasion. Cancer Lett. 2015;362:1–7.
Rong L, Li N, Zhang Z. Emerging therapies for glioblastoma: current state and future directions. J Exp Clin Cancer Res. 2022;41.
Singh S, Deora H, Neyaz A, Das KK, Mehrotra A, Srivastava AK, et al. Trends in clinico-epidemiology profile of surgically operated glioma patients in a tertiary care center over 12 years—through the looking glass! Egypt J Neurosurg. 2021;36.
Tan AC, Ashley DM, López GY, Malinzak M, Friedman HS, Khasraw M. Management of glioblastoma: state of the art and future directions. CA Cancer J Clin. 2020;70:299–312.
Garcia J, Hurwitz HI, Sandler AB, Miles D, Coleman RL, Deurloo R, et al. Bevacizumab (Avastin®) in cancer treatment: a review of 15 years of clinical experience and future outlook. Cancer Treat Rev. 2020;86:102017.
Hardee ME, Zagzag D. Mechanisms of glioma-associated neovascularization. Am J Pathol. 2012;181:1126–41.
Gilbert MR, Dignam JJ, Armstrong TS, Wefel JS, Blumenthal DT, Vogelbaum MA, et al. A randomized trial of bevacizumab for newly diagnosed glioblastoma. N Engl J Med. 2014;370:699–708.
Jaal J, Kase M, Minajeva A, Saretok M, Adamson A, Junninen J, et al. VEGFR-2 expression in glioblastoma multiforme depends on inflammatory tumor microenvironment. Int J Inflam. 2015;2015:1–7.
Grossmann P, Narayan V, Chang K, Rahman R, Abrey L, Reardon DA, et al. Quantitative imaging biomarkers for risk stratification of patients with recurrent glioblastoma treated with bevacizumab. Neuro Oncol. 2017;19:1688–97.
Gondhowiardjo S, Christina N, Ganapati NPD, Hawariy S, Radityamurti F, Jayalie VF, et al. Five-year cancer epidemiology at the national referral Hospital: Hospital-Based Cancer Registry data in Indonesia. JCO Glob Oncol. 2021; 190–203.
Hage SCE, Kawtharani MJ, Nabha SM, Saad MH. Epidemiology and distribution of primary brain tumour subtypes in Lebanon: a multicenter eleven-year study. J Neuro-Oncol Neurosci. 6.
Mbi Feh MKN, Lyon KA, Brahmaroutu AV, Tadipatri R, Fonkem E. The need for a central brain tumor registry in Africa: A review of central nervous system tumors in Africa from 1960 to 2017. Neurooncol Pr. 2021;8:337–44.
Mondia MWL, Espiritu AI, Jamora RDG. Primary brain tumor research productivity in southeast Asia and its association with socioeconomic determinants and burden of disease. Front Oncol. 2020;10:607777.
Uwishema O, Frederiksen KS, Badri R, Pradhan AU, Shariff S, Adanur I, et al. Epidemiology and etiology of brain cancer in Africa: A systematic review. Brain Behav. 2023;13.
Girardi F, Rous B, Stiller CA, Gatta G, Fersht N, Storm HH, et al. The histology of brain tumors for 67 331 children and 671 085 adults diagnosed in 60 countries during 2000–2014: a global, population-based study (CONCORD-3). Neuro Oncol. 2021;23:1765–76.
Leece R, Xu J, Ostrom QT, Chen Y, Kruchko C, Barnholtz-Sloan JS. Global incidence of malignant brain and other central nervous system tumors by histology, 2003–2007. Neuro Oncol. 2017;19:1553–64.
Fu P, He YS, Huang Q, Ding T, Cen YC, Zhao HY, et al. Bevacizumab treatment for newly diagnosed glioblastoma: Systematic review and meta-analysis of clinical trials. Mol Clin Oncol. 2016;4:833–8
Xiao D, Yan C, Li D, Xi T, Liu X, Zhu D, et al. National Brain Tumour Registry of China (NBTRC) statistical report of primary brain tumours diagnosed in China in years 2019–2020. Lancet Reg Health West Pac. 2023;34:100715.
The Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature. 2008;455:1061–8.
Pamies D, Zurich MG, Hartung T. Organotypic models to study human glioblastoma: Studying the beast in its ecosystem. iScience. 2020;23:101633.
Azzarelli R. Organoid models of glioblastoma to study brain tumor stem cells. Front Cell Dev Biol. 2020;8:220.
Gilazieva Z, Ponomarev A, Rutland C, Rizvanov A, Solovyeva V. Promising applications of tumor spheroids and organoids for personalized medicine. Cancers Basel. 2020;12:2727.
Joseph JV, Blaavand MS, Daubon T, Kruyt FAE, Thomsen MK. Three-dimensional culture models to study glioblastoma — current trends and future perspectives. Curr Opin Pharm. 2021;61:91–7.
Miller KD, Ostrom QT, Kruchko C, Patil N, Tihan T, Cioffi G, et al. Brain and other central nervous system tumor statistics, 2021. CA Cancer J Clin. 2021;71:381–406.
Ostrom QT, Price M, Neff C, Cioffi G, Waite KA, Kruchko C, et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2015–2019. Neuro-Oncol. 2022;24:v1–95.
Jalali R, Datta D. Prospective analysis of incidence of central nervous tumors presenting in a tertiary cancer hospital from India. J Neurooncol. 2008;87:111–4.
Manoharan N, Julka PK, Rath GK. Descriptive epidemiology of primary brain and CNS tumors in Delhi, 2003–2007. Asian Pac J Cancer Prev. 2012;13:637–40.
Santosh V, Jaiswal J, Shastry A, Ramesh A, Chickabasaviah Y, Arimappamagan A. Spectrum of primary intracranial tumors at a tertiary care neurological institute: a hospital-based brain tumor registry. Neurol India. 2016;64:494.
Krishnatreya M, Kataki AC, Sharma JD, Bhattacharyya M, Nandy P, Hazarika M. Brief descriptive epidemiology of primary malignant brain tumors from North-East India. Asian Pac J Cancer Prev. 2014;15:9871–3.
Pal S, Mondal S, Pradhan R, Biswas B, Banerjee A, Bhattacharyya D. Clinicopathological pattern of brain tumors: a 3-year study in a tertiary care hospital in India. Clin Cancer Investig J. 2016;5:437.
Meel M, Choudhary N, Kumar M, Mathur K. Epidemiological profiling and trends of primary intracranial tumors: a hospital-based brain tumor registry from a tertiary care center. J Neurosci Rural Pr. 2021;12:145–52.
GenomeAsia100K Consortium. The GenomeAsia 100K Project enables genetic discoveries across Asia. Nature. 2019;576:106–11.
Majumder PP, Basu A. A genomic view of the peopling and population structure of India: Fig. 1. Cold Spring Harb Perspect Biol. 2015;7:a008540.
Becker A, Sells B, Haque S, Chakravarti A. Tumor Heterogeneity in glioblastomas: From light microscopy to molecular pathology. Cancers Basel. 2021;13:761.
Gularyan SK, Gulin AA, Anufrieva KS, Shender VO, Shakhparonov MI, Bastola S, et al. Investigation of inter- and intratumoral heterogeneity of glioblastoma using TOF-SIMS. Mol Cell Proteom. 2020;19:960–70.
Chonan Y, Yamashita T, Sampetrean O, Saya H, Sudo R. Spatial heterogeneity of invading glioblastoma cells regulated by paracrine factors. Tissue Eng Part A. 2022;28:573–85.
Zhao X, Chen R, Liu M, Feng J, Chen J, Hu K. Remodeling the blood–brain barrier microenvironment by natural products for brain tumor therapy. Acta Pharm Sin B. 2017;7:541–53.
Morantz RA, Wood GW, Foster M, Clark M, Gollahon K. Macrophages in experimental and human brain tumors. J Neurosurg. 1979;50:298–304.
Navone SE, Marfia G, Invernici G, Cristini S, Nava S, Balbi S, et al. Isolation and expansion of human and mouse brain microvascular endothelial cells. Nat Protoc. 2013;8:1680–93.
Hattermann K, Sebens S, Helm O, Schmitt AD, Mentlein R, Mehdorn HM, et al. Chemokine expression profile of freshly isolated human glioblastoma-associated macrophages/microglia. Oncol Rep. 2014;32:270–6.
Annovazzi L, Mellai M, Bovio E, Mazzetti S, Pollo B, Schiffer D Microglia immunophenotyping in gliomas. Oncol Lett 2017;15:998–1006 [Internet].
Matias D, Balça-Silva J, da Graça GC, Wanjiru CM, Macharia LW, Nascimento CP, et al. Microglia/astrocytes–glioblastoma crosstalk: crucial molecular mechanisms and microenvironmental factors. Front Cell Neurosci. 2018;12.
Hambardzumyan D, Gutmann DH, Kettenmann H. The role of microglia and macrophages in glioma maintenance and progression. Nat Neurosci. 2016;19:20–7.
Junttila MR, de Sauvage FJ. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature. 2013;501:346–54.
Ma J, Chen CC, Li M. Macrophages/microglia in the glioblastoma tumor microenvironment. Int J Mol Sci. 2021;22:5775.
Tirosh I, Suvà ML. Dissecting human gliomas by single-cell RNA sequencing. Neuro Oncol. 2018;20:37–43.
Huber JD, Campos CR, Mark KS, Davis TP. Alterations in blood-brain barrier ICAM-1 expression and brain microglial activation after λ-carrageenan-induced inflammatory pain. Am J Physiol Heart Circ Physiol. 2006;290:H732–40.
Perelroizen R, Philosof B, Budick-Harmelin N, Chernobylsky T, Ron A, Katzir R, et al. Astrocyte immunometabolic regulation of the tumour microenvironment drives glioblastoma pathogenicity. Brain. 2022;145:3288–307.
McCoy MG, Nyanyo D, Hung CK, Goerger JP, R Zipfel W, Williams RM, et al. Endothelial cells promote 3D invasion of GBM by IL-8-dependent induction of cancer stem cell properties. Sci Rep. 2019;9.
Rosińska S, Gavard J. Tumor vessels fuel the fire in glioblastoma. Int J Mol Sci. 2021;22:6514.
Truffi M, Sorrentino L, Corsi F. Fibroblasts in the Tumor Microenvironment. In: Advances in Experimental Medicine and Biology. Cham: Springer International Publishing; 2020. p. 15–29. (Advances in experimental medicine and biology).
Galbo PM Jr, Madsen AT, Liu Y, Peng M, Wei Y, Ciesielski MJ, et al. Functional contribution and clinical implication of cancer-associated fibroblasts in glioblastoma. Clin Cancer Res. 2024;30:865–76.
Trylcova J, Busek P, Smetana K, Balaziova E, Dvorankova B, Mifkova A, et al. Effect of cancer-associated fibroblasts on the migration of glioma cells in vitro. Tumour Biol. 2015;36:5873–9.
Arrieta VA, Dmello C, McGrail DJ, Brat DJ, Lee-Chang C, Heimberger AB, et al. Immune checkpoint blockade in glioblastoma: from tumor heterogeneity to personalized treatment. J Clin Invest. 2023;133.
Inda MdelM, Bonavia R, Mukasa A, Narita Y, Sah DWY, Vandenberg S, et al. Tumor heterogeneity is an active process maintained by a mutant EGFR-induced cytokine circuit in glioblastoma. Genes Dev. 2010;24:1731–45.
Jacob F, Salinas RD, Zhang DY, Nguyen PTT, Schnoll JG, Wong SZH, et al. A patient-derived glioblastoma organoid model and biobank recapitulates inter- and intra-tumoral heterogeneity. Cell. 2020;180:188–204.e22.
Lam KHB, Leon AJ, Hui W, Lee SCE, Batruch I, Faust K, et al. Topographic mapping of the glioblastoma proteome reveals a triple-axis model of intra-tumoral heterogeneity. Nat Commun. 2022;13.
Ting JPY, Lovering RC, Alnemri ES, Bertin J, Boss JM, Davis BK, et al. The NLR gene family: a standard nomenclature. Immunity. 2008;28:285–7.
Harton JA, Linhoff MW, Zhang J, Ting JPY. Cutting edge: CATERPILLER: a large family of mammalian genes containing CARD, Pyrin, nucleotide-binding, and leucine-rich repeat domains. J Immunol. 2002;169:4088–93.
Chou WC, Jha S, Linhoff MW, Ting JPY. The NLR gene family: from discovery to present day. Nat Rev Immunol. 2023;23:635–54.
Agostini L, Martinon F, Burns K, McDermott MF, Hawkins PN, Tschopp J. NALP3 forms an IL-1β-processing inflammasome with increased activity in muckle-wells autoinflammatory disorder. Immunity. 2004;20:319–25.
Davis BK, Roberts RA, Huang MT, Willingham SB, Conti BJ, Brickey WJ, et al. Cutting edge: NLRC5-dependent activation of the inflammasome. J Immunol. 2011;186:1333–7.
Martinon F, Burns K, Tschopp J. The inflammasome. Mol Cell. 2002;10:417–26.
Wlodarska M, Thaiss CA, Nowarski R, Henao-Mejia J, Zhang JP, Brown EM, et al. NLRP6 inflammasome orchestrates the colonic host-microbial interface by regulating goblet cell mucus secretion. Cell. 2014;156:1045–59.
Hornung V, Ablasser A, Charrel-Dennis M, Bauernfeind F, Horvath G, Caffrey DR, et al. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature. 2009;458:514–8.
Allen IC, Moore CB, Schneider M, Lei Y, Davis BK, Scull MA, et al. NLRX1 protein attenuates inflammatory responses to infection by interfering with the RIG-I-MAVS and TRAF6-NF-κB signaling pathways. Immunity. 2011;34:854–65.
Conti BJ, Davis BK, Zhang J, O’Connor W Jr, Williams KL, Ting JPY. CATERPILLER 16.2 (CLR16.2), a novel NBD/LRR family member that negatively regulates T cell function. J Biol Chem. 2005;280:18375–85.
Cui J, Zhu L, Xia X, Wang HY, Legras X, Hong J, et al. NLRC5 negatively regulates the NF-κB and type I interferon signaling pathways. Cell. 2010;141:483–96.
Lich JD, Williams KL, Moore CB, Arthur JC, Davis BK, Taxman DJ, et al. Cutting edge: Monarch-1 suppresses non-canonical NF-κB activation and p52-dependent chemokine expression in monocytes. J Immunol. 2007;178:1256–60.
Duncan JA, Bergstralh DT, Wang Y, Willingham SB, Ye Z, Zimmermann AG, et al. Cryopyrin/NALP3 binds ATP/dATP, is an ATPase, and requires ATP binding to mediate inflammatory signaling. Proc Natl Acad Sci U A. 2007;104:8041–6.
Hochheiser IV, Pilsl M, Hagelueken G, Moecking J, Marleaux M, Brinkschulte R, et al. Structure of the NLRP3 decamer bound to the cytokine release inhibitor CRID3. Nature. 2022;604:184–9.
Sharif H, Wang L, Wang WL, Magupalli VG, Andreeva L, Qiao Q, et al. Structural mechanism for NEK7-licensed activation of NLRP3 inflammasome. Nature. 2019;570:338–43.
Jin T, Huang M, Jiang J, Smith P, Xiao TS. Crystal structure of human NLRP12 PYD domain and implication in homotypic interaction. PLoS One. 2018;13:e0190547.
Sharma BR, Kanneganti TD. NLRP3 inflammasome in cancer and metabolic diseases. Nat Immunol. 2021;22:550–9.
Subramanian N, Natarajan K, Clatworthy MR, Wang Z, Germain RN. The adaptor MAVS promotes NLRP3 mitochondrial localization and inflammasome activation. Cell. 2013;153:348–61.
Hagar JA, Powell DA, Aachoui Y, Ernst RK, Miao EA. Cytoplasmic LPS activates caspase-11: implications in TLR4-independent endotoxic shock. Science. 2013;341:1250–3.
Juliana C, Fernandes-Alnemri T, Kang S, Farias A, Qin F, Alnemri ES. Non-transcriptional priming and deubiquitination regulate NLRP3 inflammasome activation. J Biol Chem. 2012;287:36617–22.
Py BF, Kim MS, Vakifahmetoglu-Norberg H, Yuan J. Deubiquitination of NLRP3 by BRCC3 critically regulates inflammasome activity. Mol Cell. 2013;49:331–8.
Gurung P, Malireddi RKS, Anand PK, Demon D, Walle LV, Liu Z, et al. Toll or interleukin-1 receptor (TIR) domain-containing adaptor inducing interferon-β (TRIF)-mediated caspase-11 protease production integrates toll-like receptor 4 (TLR4) protein- and Nlrp3 inflammasome-mediated host defense against enteropathogens. J Biol Chem. 2012;287:34474–83.
Gurung P, Anand PK, Malireddi RKS, Vande Walle L, Van Opdenbosch N, Dillon CP, et al. FADD and caspase-8 mediate priming and activation of the canonical and noncanonical Nlrp3 inflammasomes. J Immunol. 2014;192:1835–46.
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol. 2010;11:136–40.
Albalawi F, Lu W, Beckel JM, Lim JC, McCaughey SA, Mitchell CH. The P2X7 receptor primes IL-1β and the NLRP3 inflammasome in astrocytes exposed to mechanical strain. Front Cell Neurosci. 2017;11.
He Y, Zeng MY, Yang D, Motro B, Núñez G. NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux. Nature. 2016;530:354–7.
Schmid-Burgk JL, Chauhan D, Schmidt T, Ebert TS, Reinhardt J, Endl E, et al. A genome-wide CRISPR (clustered regularly interspaced short palindromic repeats) screen identifies NEK7 as an essential component of NLRP3 inflammasome activation. J Biol Chem. 2016;291:103–9.
Chen J, Chen ZJ. PtdIns4P on dispersed trans-Golgi network mediates NLRP3 inflammasome activation. Nature. 2018;564:71–6.
Broz P, von Moltke J, Jones JW, Vance RE, Monack DM. Differential requirement for caspase-1 autoproteolysis in pathogen-induced cell death and cytokine processing. Cell Host Microbe. 2010;8:471–83.
Ataide MA, Andrade WA, Zamboni DS, Wang D, Souza M do C, Franklin BS, et al. Malaria-induced NLRP12/NLRP3-dependent caspase-1 activation mediates inflammation and hypersensitivity to bacterial superinfection. PLoS Pathog. 2014;10:e1003885.
Platnich JM, Chung H, Lau A, Sandall CF, Bondzi-Simpson A, Chen HM, et al. Shiga toxin/lipopolysaccharide activates caspase-4 and gasdermin D to trigger mitochondrial reactive oxygen species upstream of the NLRP3 inflammasome. Cell Rep. 2018;25:1525–36.e7.
Allen IC, Lich JD, Arthur JC, Jania CM, Roberts RA, Callaway JB, et al. Characterization of NLRP12 during the development of allergic airway disease in mice. PLoS One. 2012;7:e30612.
Lupfer C, Kanneganti TD. Unsolved mysteries in NLR biology. Front Immunol. 2013;4:285.
Vladimer GI, Weng D, Paquette SWM, Vanaja SK, Rathinam VAK, Aune MH, et al. The NLRP12 Inflammasome Recognizes Yersinia pestis. Immunity. 2012;37:96–107.
Sundaram B, Pandian N, Mall R, Wang Y, Sarkar R, Kim HJ, et al. NLRP12-PANoptosome activates PANoptosis and pathology in response to heme and PAMPs. Cell. 2023;186:2783–801.e20.
Kostik MM, Suspitsin EN, Guseva MN, Levina AS, Kazantseva AY, Sokolenko AP, et al. Multigene sequencing reveals heterogeneity of NLRP12-related autoinflammatory disorders. Rheumatol Int. 2018;38:887–93.
Borte S, Celiksoy MH, Menzel V, Ozkaya O, Ozen FZ, Hammarström L, et al. Novel NLRP12 mutations associated with intestinal amyloidosis in a patient diagnosed with common variable immunodeficiency. Clin Immunol. 2014;154:105–11.
Hoffman HM, Mueller JL, Broide DH, Wanderer AA, Kolodner RD. Mutation of a new gene encoding a putative pyrin-like protein causes familial cold autoinflammatory syndrome and Muckle–Wells syndrome. Nat Genet. 2001;29:301–5.
Jéru I, Duquesnoy P, Fernandes-Alnemri T, Cochet E, Yu JW, Lackmy-Port-Lis M, et al. Mutations in NALP12 cause hereditary periodic fever syndromes. Proc Natl Acad Sci U A. 2008;105:1614–9.
Allen IC, TeKippe EM, Woodford RMT, Uronis JM, Holl EK, Rogers AB, et al. The NLRP3 inflammasome functions as a negative regulator of tumorigenesis during colitis-associated cancer. J Exp Med. 2010;207:1045–56.
Khan S, Zaki H. Crosstalk between NLRP12 and JNK during hepatocellular carcinoma. Int J Mol Sci. 2020;21:496.
Xu Y, Li H, Chen W, Yao X, Xing Y, Wang X, et al. Mycoplasma hyorhinis activates the NLRP3 inflammasome and promotes migration and invasion of gastric cancer cells. PLoS One. 2013;8:e77955.
Pan G, Zhu X, Yao Y, Zhang H, Zhan S, Sun L, Yang X, et al. Downregulation of NLRP12 enhances the proliferation, migration and drug-resistance of colorectal cancer cells by modulating MEK/ERK/GLI1 signaling pathway. Int J Clin Exp Med. 2018;11:5332–42.
Udden SMN, Kwak YT, Godfrey V, Khan MAW, Khan S, Loof N, et al. NLRP12 suppresses hepatocellular carcinoma via downregulation of cJun N-terminal kinase activation in the hepatocyte. Elife. 2019;8.
Peng S, Lü B, Ruan W, Zhu Y, Sheng H, Lai M. Genetic polymorphisms and breast cancer risk: evidence from meta-analyses, pooled analyses, and genome-wide association studies. Breast Cancer Res Treat. 2011;127:309–24.
Li Y, Zhao W, Zhao Z, Wu J, Chen L, Ma Y, et al. IL1B gene polymorphisms, age and the risk of non-small cell lung cancer in a Chinese population. Lung Cancer Amst Neth. 2015;89:232–7.
Santos Freire M, Victor de Oliveira Monteiro A, Moura Martins T, Socorro Silva Lima Duarte M, Carlos Lima A, Luiz Araújo Bentes Leal A, et al. Genetic variations in immune mediators and prostate cancer risk: a field synopsis with Bayesian calculations. Cytokine. 2024;179:156630.
Gunter MJ, Canzian F, Landi S, Chanock SJ, Sinha R, Rothman N. Inflammation-related gene polymorphisms and colorectal adenoma. Cancer Epidemiol Biomark Prev Publ Am Assoc Cancer Res Cosponsored Am Soc Prev Oncol. 2006;15:1126–31.
Nardin A, Abastado JP. Macrophages and cancer. Front Biosci. 2008;13:494–505.
Bunt SK, Yang L, Sinha P, Clements VK, Leips J, Ostrand-Rosenberg S. Reduced inflammation in the tumor microenvironment delays the accumulation of myeloid-derived suppressor cells and limits tumor progression. Cancer Res. 2007;67:10019–26.
Miskiewicz A, Szparecki G, Durlik M, Rydzewska G, Ziobrowski I, Górska R. The Q705K and F359L single-nucleotide polymorphisms of NOD-like receptor signaling pathway: Association with chronic pancreatitis, pancreatic cancer, and periodontitis. Arch Immunol Ther Exp Warsz. 2015;63:485–94.
Karan D, Tawfik O, Dubey S. Expression analysis of inflammasome sensors and implication of NLRP12 inflammasome in prostate cancer. Sci Rep. 2017;7.
Jiao J, Zhao G, Wang Y, Ren P, Wu M. MCC950, a selective inhibitor of NLRP3 inflammasome, reduces the inflammatory response and improves neurological outcomes in mice model of spinal cord injury. Front Mol Biosci. 2020;7:37.
Oronsky B, Takahashi L, Gordon R, Cabrales P, Caroen S, Reid T. RRx-001: a chimeric triple action NLRP3 inhibitor, Nrf2 inducer, and nitric oxide superagonist. Front Oncol. 2023;13:1204143.
Parveen R, Kashif M, Srinivasan H, Khan J, Yousif A, et al. An In Silico Investigation of Pharmacological Modulators and Inflammasomes in Glioblastoma Multiforme. Appl Biochem Biotechnol. 2024;196:2771–97.
Shang S, Wang L, Zhang Y, Lu H, Lu X. The beta-hydroxybutyrate suppresses the migration of glioma cells by inhibition of NLRP3 inflammasome. Cell Mol Neurobiol. 2018;38:1479–89.
Yin XF, Zhang Q, Chen ZY, Wang HF, Li X, Wang HX, et al. NLRP3 in human glioma is correlated with increased WHO grade, and regulates cellular proliferation, apoptosis and metastasis via epithelial-mesenchymal transition and the PTEN/AKT signaling pathway. Int J Oncol. 2018;53:973–86.
Xue L, Lu B, Gao B, Shi Y, Xu J, Yang R, et al. NLRP3 promotes glioma cell proliferation and invasion via the interleukin-1β/NF-κB p65 signals. Oncol Res. 2019;27:557–64.
Demuth T, Reavie LB, Rennert JL, Nakada M, Nakada S, Hoelzinger DB, et al. MAP-ing glioma invasion: Mitogen-activated protein kinase kinase 3 and p38 drive glioma invasion and progression and predict patient survival. Mol Cancer Ther. 2007;6:1212–22.
Yeung YT, Bryce NS, Adams S, Braidy N, Konayagi M, McDonald KL, et al. p38 MAPK inhibitors attenuate pro-inflammatory cytokine production and the invasiveness of human U251 glioblastoma cells. J Neurooncol. 2012;109:35–44.
Yoshino Y, Aoyagi M, Tamaki M, Duan L, Morimoto T, Ohno K. Activation of p38 MAPK and/or JNK contributes to increased levels of VEGF secretion in human malignant glioma cells. Int J Oncol. 2006;29:981–7.
Sasaki A, Tamura M, Hasegawa M, Ishiuchi S, Hirato J, Nakazato Y. Expression of lnterleukin-1 β mRNA and protein in human gliomas assessed by RT-PCR and immunohistochemistry. J Neuropathol Exp Neurol. 1998;57:653–63.
Tarassishin L, Casper D, Lee SC. Aberrant expression of interleukin-1β and inflammasome activation in human malignant gliomas. PLoS One. 2014;9:e103432.
Chen P, Li Y, Li N, Shen L, Li Z. Comprehensive analysis of pyroptosis-associated in molecular classification, immunity and prognostic of glioma. Front Genet. 2021;12:781538.
Hübner M, Effinger D, Wu T, Strauß G, Pogoda K, Kreth FW, et al. The IL-1 antagonist anakinra attenuates glioblastoma aggressiveness by dampening tumor-associated inflammation. Cancers Basel. 2020;12:433.
Tuladhar S, Kanneganti TD. NLRP12 in innate immunity and inflammation. Mol Asp Med. 2020;76:100887.
Sharma N, Saxena S, Agrawal I, Singh S, Srinivasan V, Arvind S, et al. Differential expression profile of NLRs and AIM2 in glioma and implications for NLRP12 in glioblastoma. Sci Rep. 2019;9.
Cheng YW, Chen YY, Lin CJ, Chen YT, Lieu AS, Tsai HP, et al. High expression of NLRP12 predicts poor prognosis in patients with intracranial glioma. J Chin Med Assoc. 2023;86:88–97.
Tang F, Kunder R, Chu T, Hains A, Nguyen A, McBride JM, et al. First‐in‐human phase 1 trial evaluating safety, pharmacokinetics, and pharmacodynamics of NLRP3 inflammasome inhibitor, GDC‐2394, in healthy volunteers. Clin Transl Sci. 2023;16:1653–66.
MCC950 directly targets the NLRP3 ATP-hydrolysis motif for inflammasome inhibition | Nat Chem Biol [Internet]. [cited 2024 Feb 22]. Available from: https://www.nature.com/articles/s41589-019-0277-7
Frontiers | Tranilast directly targets NLRP3 to protect melanocytes from keratinocyte-derived IL-1β under oxidative stress [Internet]. [cited 2024 Feb 22]. Available from: https://www.frontiersin.org/articles/10.3389/fcell.2020.00588/full
Jayabalan N, Oronsky B, Cabrales P, Reid T, Caroen S, Johnson AM, et al. A Review of RRx-001: a late-stage multi-indication inhibitor of NLRP3 activation and chronic inflammation. Drugs. 2023;83:389–402.
Coleman MP, Quaresma M, Berrino F, Lutz JM, De Angelis R, Capocaccia R, et al. Cancer survival in five continents: a worldwide population-based study (CONCORD). Lancet Oncol. 2008;9:730–56.
Mendiratta J, Pillamarapu M, Chakraborty I, Vaswani R, Kapoor M, Vadlamani S, et al. Ethnic representation in interventional clinical trials run in India. Lancet Reg Health Southeast Asia. 2023;15:100230.
Acknowledgements
We would like to acknowledge Durgesh Meena for proofreading the manuscript.
Funding
The Inflammation Immunity and Tumor Biology Lab is supported by the Indian Institute of Technology Jodhpur, and SR is supported by the Ministry of Human Resource and Development funded GATE fellowships for pursuing a PhD.
Author information
Authors and Affiliations
Contributions
SR wrote the manuscript after a literature study with inputs from SJ. The table data are compiled by SR, and the figures are created by SR using biorender.com upon the supervision of SJ.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rajkhowa, S., Jha, S. The role of NLRP3 and NLRP12 inflammasomes in glioblastoma. Genes Immun 25, 541–551 (2024). https://doi.org/10.1038/s41435-024-00309-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41435-024-00309-z