Abstract
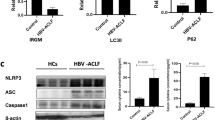
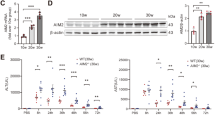
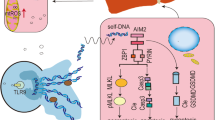
Autoimmune hepatitis (AIH) involves chronic liver injury from abnormal immune responses, leading to hepatocyte death and inflammation. Currently, the role of pyroptosis in AIH has not been fully elucidated. Emerging evidence indicates that Nlrp12, a cytosolic sensor of damage-associated molecular patterns (DAMPs), participates in inflammasome assembly and pyroptotic signalling. Therefore, this study aims to investigate the role of Nlrp12 in AIH. Using a concanavalin A (ConA)-induced AIH mouse model, we observed that ConA challenge significantly upregulated Nlrp12 expression in liver tissues. Genetic ablation of Nlrp12 improved survival rates, lowered serum aminotransferase levels, reduced pro-inflammatory cytokine production, and ameliorated histological liver damage. Further studies revealed that Nlrp12 drives the assembly of PANoptosomes (including ASC, RIPK3, and Caspase-8), promoting the release of PANoptosis-related proteins and inflammatory factors. The RIPK3 inhibitor GSK-872 blocked PANoptosome formation, mitigating liver injury and inflammation. These findings unveil a previously unrecognized role for Nlrp12 in aggravating immune-mediated liver injury via PANoptosis activation, highlighting the Nlrp12-PANoptosis axis as a promising therapeutic target for AIH.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data from this study were made available through reasonable requests to the corresponding author. No datasets were generated or analysed during the current study.
References
Mieli-Vergani G, Vergani D, Czaja AJ, Manns MP, Krawitt EL, Vierling JM, et al. Autoimmune hepatitis. Nat Rev Dis Prim. 2018;4:18017.
Muratori L, Lohse AW, Lenzi M. Diagnosis and management of autoimmune hepatitis. BMJ. 2023;380:e070201.
Sirbe C, Simu G, Szabo I, Grama A, Pop TL. Pathogenesis of autoimmune hepatitis-cellular and molecular mechanisms. Int J Mol Sci. 2021;22:13578.
Wang H, Yan W, Feng Z, Gao Y, Zhang L, Feng X, et al. Plasma proteomic analysis of autoimmune hepatitis in an improved AIH mouse model. J Transl Med. 2020;18:3.
Wang L, Yan F, Zhang J, Xiao Y, Wang C, Zhu Y, et al. Cornuside improves murine autoimmune hepatitis through inhibition of inflammatory responses. Phytomedicine. 2023;120:155077.
Wang J, Sun Z, Xie J, Ji W, Cui Y, Ai Z, et al. Inflammasome and pyroptosis in autoimmune liver diseases. Front Immunol. 2023;14:1150879.
An R, Zhu Z, Chen Y, Guan W, Wang J, Ren H. MSCs Suppress Macrophage Necroptosis and Foster Liver Regeneration by Modulating SP1/SK1 Axis in Treating Acute Severe Autoimmune Hepatitis. Adv Sci (Weinh). 2025;12:e2408974.
Sun X, Yang Y, Meng X, Li J, Liu X, Liu H. PANoptosis: mechanisms, biology, and role in disease. Immunol Rev. 2024;321:246–62.
Qi Z, Zhu L, Wang K, Wang N. PANoptosis: emerging mechanisms and disease implications. Life Sci. 2023;333:122158.
Pandeya A, Kanneganti TD. Therapeutic potential of PANoptosis: innate sensors, inflammasomes, and RIPKs in PANoptosomes. Trends Mol Med. 2024;30:74–88.
Sundaram B, Pandian N, Kim HJ, Abdelaal HM, Mall R, Indari O, et al. NLRC5 senses NAD(+) depletion, forming a PANoptosome and driving PANoptosis and inflammation. Cell. 2024;187:4061–77.e17.
Yang B, Hu A, Wang T, Chen X, Ma C, Yang X, et al. SARS-CoV-2 infection induces ZBP1-dependent PANoptosis in bystander cells. Proc Natl Acad Sci USA. 2025;122:e2500208122.
Liu K, Wang M, Li D, Duc Duong NT, Liu Y, Ma J, et al. PANoptosis in autoimmune diseases interplay between apoptosis, necrosis, and pyroptosis. Front Immunol. 2024;15:1502855.
Huang L, Tao Y, Wu X, Wu J, Shen M, Zheng Z. The role of Nlrp12 in inflammatory diseases. Eur J Pharm. 2023;956:175995.
Chen R, Zou J, Chen J, Zhong X, Kang R, Tang D. Pattern recognition receptors: function, regulation and therapeutic potential. Signal Transduct Target Ther. 2025;10:216.
Jiang J, Shen W, He Y, Liu J, Ouyang J, Zhang C, et al. Overexpression of Nlrp12 enhances antiviral immunity and alleviates herpes simplex keratitis via pyroptosis/IL-18/IFN-γ signaling. Int Immunopharmacol. 2024;137:112428.
Li X, Zhou G, Sun X, Qu S, Lai H, Wu Y, et al. Nlrp12 Senses the SARS-CoV-2 Membrane Protein and Promotes an Inflammatory Response. J Infect Dis. 2024;229:660–70.
Khan S, Kwak YT, Peng L, Hu S, Cantarel BL, Lewis CM, et al. Nlrp12 downregulates the Wnt/β-catenin pathway via interaction with STK38 to suppress colorectal cancer. J Clin Invest. 2023;133:e166295.
Sundaram B, Pandian N, Mall R, Wang Y, Sarkar R, Kim HJ, et al. Nlrp12-PANoptosome activates PANoptosis and pathology in response to heme and PAMPs. Cell. 2023;186:2783–801.e20.
Bernard EM, Broz P. Nlrp12 senses heme and PAMPs to drive necrotic cell death and inflammation. Mol Cell. 2023;83:2621–3.
Tuladhar S, Kanneganti TD. Nlrp12 in innate immunity and inflammation. Mol Asp Med. 2020;76:100887.
Wang J, He W, Li C, Ma Y, Liu M, Ye J, et al. Focus on negatively regulated NLRs in inflammation and cancer. Int Immunopharmacol. 2024;136:112347.
Babamale AO, Chen ST. Nod-like receptors: critical intracellular sensors for host protection and cell death in microbial and parasitic infections. Int J Mol Sci. 2021; 22:11398.
Yue Y, Shi M, Song X, Ma C, Li D, Hu X, et al. Lycopene ameliorated DSS-induced colitis by improving epithelial barrier functions and inhibiting the escherichia coli adhesion in mice. J Agric Food Chem. 2024;72:5784–96.
Ma W, Wang Y, Liu J. NLRP3 inflammasome activation in liver disorders: from molecular pathways to therapeutic strategies. J Inflamm Res. 2025;18:8277–94.
Shi C, Cao P, Wang Y, Zhang Q, Zhang D, Wang Y, et al. PANoptosis: a cell death characterized by pyroptosis, apoptosis, and necroptosis. J Inflamm Res. 2023;16:1523–32.
Wang L, Zhu Y, Zhang L, Guo L, Wang X, Pan Z, et al. Mechanisms of PANoptosis and relevant small-molecule compounds for fighting diseases. Cell Death Dis. 2023;14:851.
Acknowledgements
We acknowledge all individuals who contributed to this research and appreciate the funding that supported this research.
Funding
This study was supported by the National Natural Science Foundation of China (No.82170645), Department of Science and Technology of Sichuan Province (CN) (No.2024NSFSC0735), CMC Excellent-talent Programme (No.2024kjTzn04), the Scientific Research and Innovation Fund for Postgraduate of Chengdu Medical College (No. YCX2025-01-71) and the Research Fund of Chengdu Medical College (No. CYZYB25-10).
DeclarationsAll authors have made significant contributions to the conception, design, analysis, and/or interpretation of the work presented in this manuscript. Furthermore, all authors have thoroughly reviewed and approved the final version of the manuscript. They fully agree with its content and the decision to submit it for consideration.
Author information
Authors and Affiliations
Contributions
JL: Investigation, Data curation, Methodology, Writing-original draft. KYF: Investigation, Formal analysis, Methodology, Writing-original draft. FYW: Investigation, Formal analysis, Methodology. ZQZ: Validation, Methodology. XYL: Writing-review & editing, Project administration, Conceptualization. CFM: Writing-review & editing, Supervision, Funding acquisition, Conceptualization. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All animal experiments were conducted following the Guide for the Care and Use of Laboratory Animals and approved by the Ethics Committee of Chengdu Medical College (Approval No.CMC-IACUC-2022052).
Informed consent
All authors have read and approved the final version of the manuscript and agree to its publication. Informed consent was obtained from all participants involved in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Associated Data This section collects any data citations, data availability statements, or supplementary materials included in this article.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, J., Feng, K., Wang, F. et al. Nlrp12-driven PANoptosis exacerbates liver injury in ConA-induced autoimmune hepatitis. Genes Immun (2026). https://doi.org/10.1038/s41435-026-00386-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41435-026-00386-2