Abstract
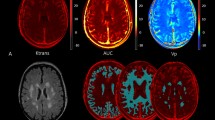
Cerebral small vessel disease (cSVD) is associated with increased blood-brain barrier (BBB) permeability. We sought to evaluate whether arterial stiffness might be associated with BBB permeability in patients with cSVD. We assessed BBB permeability using Dynamic Contrast-Enhanced MRI (DCE-MRI) in 29 patients that had suffered a recent small subcortical infarct (RSSI). BBB permeability in the whole brain (WB), gray matter (GM) and white matter (WM) was assessed with the parameter Ktrans. We used ambulatory blood pressure monitoring to measure 24-h systolic blood pressure (24-h SBP), diastolic blood pressure (24-h DBP), and pulse wave velocity (24-h PWV) both after stroke and following a 2-year follow-up. The differences between both measurements were calculated as Δ24-h SBP, Δ24-h DBP and Δ24-h PWV. DCE-MRI was acquired at a median (IQR) of 24 (19–27) months after stroke. Median age was 66.7 (9.7) years, and 24 (83%) patients were men. Median (IQR) Δ24-h PWV was 0.3 (−0.1, 0.5) m/s. WB-Ktrans, GM-Ktrans, and WM-Ktrans were associated with Δ24-h PWV (Spearman’s, r [95% CI], WB 0.651 [0.363–0.839]; GM 0.657 [0.373–0.845], WM 0.530[0.197–0.777]) but not with Δ24-h SBP or Δ24-h DBP. These associations remained significant after adjustment with linear regression models, controlling for age, sex, body mass index, and Δ24-h SBP (b[95% CI], WB 0.725[0.384–1.127], GM 0.629 [0.316–1.369], WM 0.865 [0.455–0.892]) or Δ24-h DBP (b[95% CI], WM 0.707 [0.370–1.103], GM 0.643 [0.352–1.371], WM 0.772 [0.367–0.834]). Our results suggest that an increment on arterial stiffness in the months following a RSSI might increase BBB permeability.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Pantoni L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010;9:689–701. https://doi.org/10.1016/S1474-4422(10)70104-6.
Duering M, Biessels GJ, Brodtmann A, Chen C, Cordonnier C, de Leeuw FE, et al. Neuroimaging standards for research into small vessel disease—advances since 2013. Lancet Neurol. 2023;22:602–18. https://www.sciencedirect.com/science/article/pii/S147444222300131X.
Wardlaw JM, Doubal F, Armitage P, Chappell F, Carpenter T, Muñoz Maniega S, et al. Lacunar stroke is associated with diffuse blood–brain barrier dysfunction. Ann Neurol. 2009;65:194–202. https://doi.org/10.1002/ana.21549.
Wong SM, Jansen JFA, Zhang CE, Hoff EI, Staals J, van Oostenbrugge RJ, et al. Blood-brain barrier impairment and hypoperfusion are linked in cerebral small vessel disease. Neurology. 2019;92:e1669 LP–e1677. http://n.neurology.org/content/92/15/e1669.abstract.
Muñoz Maniega S, Chappell FM, Valdés Hernández MC, Armitage PA, Makin SD, Heye AK, et al. Integrity of normal-appearing white matter: Influence of age, visible lesion burden and hypertension in patients with small-vessel disease. J Cereb Blood Flow Metab. 2016;37:644–56. https://doi.org/10.1177/0271678X16635657.
Heye AK, Thrippleton MJ, Chappell FM, Valdés Hernández MdelC, Armitage PA, Makin SD, et al. Blood pressure and sodium: Association with MRI markers in cerebral small vessel disease. J Cereb Blood Flow Metab. 2015;36:264–74. https://doi.org/10.1038/jcbfm.2015.64.
Webb AJS, Simoni M, Mazzucco S, Kuker W, Schulz U, Rothwell PM. Increased cerebral arterial pulsatility in patients with leukoaraiosis. Stroke. 2012;43:2631–6. https://doi.org/10.1161/STROKEAHA.112.655837.
Webb AJS, Lawson A, Wartolowska K, Mazzucco S, Rothwell PM. Aortic stiffness, pulse pressure, and cerebral pulsatility progress despite best medical management: the OXVASC cohort. Stroke. 2022;53:1310–7. https://doi.org/10.1161/STROKEAHA.121.035560.
Shi Y, Thrippleton MJ, Blair GW, Dickie DA, Marshall I, Hamilton I, et al. Small vessel disease is associated with altered cerebrovascular pulsatility but not resting cerebral blood flow. J Cereb Blood Flow Metab. 2018;40:85–99. https://doi.org/10.1177/0271678X18803956.
Garcia-Polite F, Martorell J, Del Rey-Puech P, Melgar-Lesmes P, O’Brien CC, Roquer J, et al. Pulsatility and high shear stress deteriorate barrier phenotype in brain microvascular endothelium. J Cereb Blood Flow Metab. 2016;37:2614–25. https://doi.org/10.1177/0271678X16672482.
CJ A, Patrick S, Timothy H, Raymond T. Large-artery stiffness in health and disease. J Am Coll Cardiol. 2019;74:1237–63. https://doi.org/10.1016/j.jacc.2019.07.012.
Vilar-Bergua A, Riba-Llena I, Penalba A, Cruz LM, Jiménez-Balado J, Montaner J, et al. N-terminal pro-brain natriuretic peptide and subclinical brain small vessel disease. Neurology. 2016;87:2533 LP–2539. http://n.neurology.org/content/87/24/2533.abstract.
Gómez-Choco M, Mena L, Font MÀ, Mengual JJ, Garcia-Sanchez SM, Avellaneda C, et al. NT-proBNP, cerebral small vessel disease and cardiac function in patients with a recent lacunar infarct. J Hum Hypertens. 2023;37:62–7. https://doi.org/10.1038/s41371-021-00648-8.
Bohara M, Kambe Y, Nagayama T, Tokimura H, Arita K, Miyata A. C-type natriuretic peptide modulates permeability of the blood–brain barrier. J Cereb Blood Flow Metab. 2014;34:589–96. https://doi.org/10.1038/jcbfm.2013.234.
Thrippleton MJ, Backes WH, Sourbron S, Ingrisch M, van Osch MJP, Dichgans M, et al. Quantifying blood-brain barrier leakage in small vessel disease: review and consensus recommendations. Alzheimer’s Dement. 2019;15:840–58. https://www.sciencedirect.com/science/article/pii/S1552526019300457.
Mena L, Mengual JJ, García-Sánchez SM, Avellaneda-Gómez C, Font MÀ, Montull C, et al. Relationship of arterial stiffness and baseline vascular burden with new lacunes and microbleeds: a longitudinal cohort study. Eur Stroke J. 2023;23969873231207764. Available from: https://doi.org/10.1177/23969873231207764.
Wardlaw JM, Smith EE, Biessels GJ, Cordonnier C, Fazekas F, Frayne R, et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 2013;12:822–38. https://doi.org/10.1016/S1474-4422(13)70124-8.
Fernández-Llama P, Pareja J, Yun S, Vázquez S, Oliveras A, Armario P, et al. Cuff-based oscillometric central and brachial blood pressures obtained through ABPM are similarly associated with renal organ damage in arterial hypertension. Kidney Blood Press Res. 2017;42:1068–77. https://doi.org/10.1159/000485595.
Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M, et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur Heart J. 2018;39:3021–104. https://doi.org/10.1093/eurheartj/ehy339.
Hametner B, Wassertheurer S, Kropf J, Mayer C, Eber B, Weber T. Oscillometric estimation of aortic pulse wave velocity: comparison with intra-aortic catheter measurements. Blood Press Monit. 2013;18. Available from: https://journals.lww.com/bpmonitoring/fulltext/2013/06000/oscillometric_estimation_of_aortic_pulse_wave.9.aspx.
Klarenbeek P, van Oostenbrugge RJ, Rouhl RPW, Knottnerus ILH, Staals J. Ambulatory blood pressure in patients with lacunar stroke. Stroke. 2013;44:2995–9. https://doi.org/10.1161/STROKEAHA.113.002545.
Barnes SR, Ng TSC, Santa-Maria N, Montagne A, Zlokovic BV, Jacobs RE. ROCKETSHIP: a flexible and modular software tool for the planning, processing and analysis of dynamic MRI studies. BMC Med Imaging. 2015;15:19 https://doi.org/10.1186/s12880-015-0062-3.
Wardlaw JM, Makin SJ, Valdés Hernández MC, Armitage PA, Heye AK, Chappell FM, et al. Blood-brain barrier failure as a core mechanism in cerebral small vessel disease and dementia: evidence from a cohort study. Alzheimer’s Dement. 2017;13:634–43. https://doi.org/10.1016/j.jalz.2016.09.006.
Kerkhofs D, Wong SM, Zhang E, Staals J, Jansen JFA, van Oostenbrugge RJ, et al. Baseline blood-brain barrier leakage and longitudinal microstructural tissue damage in the periphery of white matter hyperintensities. Neurology. 2021;96:e2192 LP–e2200. http://n.neurology.org/content/96/17/e2192.abstract.
Muhire G, Iulita MF, Vallerand D, Youwakim J, Gratuze M, Petry FR, et al. Arterial stiffness due to carotid calcification disrupts cerebral blood flow regulation and leads to cognitive deficits. J Am Heart Assoc. 2019;8:e011630. https://doi.org/10.1161/JAHA.118.011630.
Verheggen ICM, de Jong JJA, van Boxtel MPJ, Gronenschild EHBM, Palm WM, Postma AA, et al. Increase in blood–brain barrier leakage in healthy, older adults. GeroScience. 2020;42:1183–93. https://doi.org/10.1007/s11357-020-00211-2.
Group TSMI for the SR. Association of intensive vs standard blood pressure control with cerebral white matter lesions. JAMA. 2019;322:524–34. https://doi.org/10.1001/jama.2019.10551.
White WB, Wakefield DB, Moscufo N, Guttmann CRG, Kaplan RF, Bohannon RW, et al. Effects of intensive versus standard ambulatory blood pressure control on cerebrovascular outcomes in older people (INFINITY). Circulation. 2019;140:1626–35. https://doi.org/10.1161/CIRCULATIONAHA.119.041603.
Miyagi T, Ishida A, Shinzato T, Ohya Y. Arterial stiffness is associated with small vessel disease irrespective of blood pressure in stroke-free individuals. Stroke. 2023;54:2814–21. https://doi.org/10.1161/STROKEAHA.123.042512.
Goldstein ED, Wolcott Z, Garg G, Navarro K, Delic A, Yaghi S, et al. Effect of antihypertensives by class on cerebral small vessel disease: a post hoc analysis of SPRINT-MIND. Stroke 2022;53:2435–40. https://doi.org/10.1161/STROKEAHA.121.037997.
Kopczak A, Stringer MS, van den Brink H, Kerkhofs D, Blair GW, van Dinther M, et al. The effects of amlodipine and other blood pressure lowering agents on microvascular function in small vessel diseases (TREAT-SVDs) trial: study protocol for a randomised crossover trial. Eur Stroke J. 2022;8:387–97. https://doi.org/10.1177/23969873221143570. Available from.
Bernardo-Castro S, Sousa JA, Brás A, Cecília C, Rodrigues B, Almendra L, et al. Pathophysiology of blood–brain barrier permeability throughout the different stages of ischemic stroke and its implication on hemorrhagic transformation and recovery. Front Neurol. 2020. Available from: https://www.frontiersin.org/articles/10.3389/fneur.2020.594672
Acknowledgements
We thank CERCA Programme / Generalitat de Catalunya for institutional support. We thank all the personnel at Alomar center for the technical support.
Funding
This work was supported by a grant from the Fondo de Investigaciones Sanitarias of the Spanish Ministry of Health (PI17/1188). MGC was funded by the Department of Health of the Generalitat de Catalunya (grant SLT008/18/00032) by the call “Acció instrumental: Intensificació de professionals de la salut, modalitat de professionals sanitaris (infermeria I fisioteràpia)”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mena Romo, L., Mengual, J.J., Avellaneda-Gómez, C. et al. Association between blood-brain barrier permeability and changes in pulse wave velocity following a recent small subcortical infarct. Hypertens Res 47, 2495–2502 (2024). https://doi.org/10.1038/s41440-024-01764-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-024-01764-x