Abstract
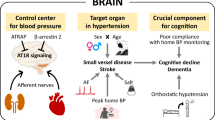
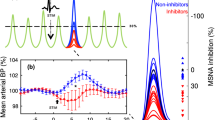
Hypertension is both a driver and a consequence of brain dysfunction. The brain regulates circulation via control of autonomic nervous system tone by integrating neural and humoral signals. It is also a vulnerable target of blood pressure (BP)–related injury, ranging from overt stroke to covert small-vessel disease. Recognizing this bidirectional relationship is essential for advancing precision in prevention and treatment. On the mechanistic side, recent work has clarified how the brain renin–angiotensin system, sodium–glucose cotransporter 2, the melanocortin system, and the gut–brain axis shape autonomic output. In addition, renewed attention has been given to centrally acting sympatholytics, imidazoline receptor agonists, which demonstrate antihypertensive efficacy and metabolic neutrality in contemporary trials. Collectively, these studies reinforce that central pathways remain viable therapeutic targets for modulating sympathetic activity. Clinically, multiple investigations highlight that cerebrovascular outcomes depend not only on mean BP but also on patterns and cumulative injury. Total small-vessel disease burden integrates lifetime vascular damage and predicts prognosis after stroke. Nocturnal BP surges and visit-to-visit variability further stratify cerebrovascular risk, while beat-to-beat fluctuations after reperfusion influence recovery. Pulse pressure after intracerebral hemorrhage links systemic hemodynamics with renal and neurological outcomes, and prevention gaps such as untreated hypertension remain striking, especially in younger patients. Together, these advances emphasize that brain health and BP regulation are inseparable. This review highlights recent advances in both central mechanisms of sympathoexcitation and clinical perspectives on cerebrovascular outcomes in hypertension.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hirooka Y, Kishi T, Ito K, Sunagawa K. Potential clinical application of recently discovered brain mechanisms involved in hypertension. Hypertension. 2013;62:995–1002.
Nakagawa P, Sigmund CD. How is the brain renin–angiotensin system regulated?. Hypertension. 2017;70:10–18.
Fekete ÉM, Gomez J, Ghobrial M, Kaminski K, Muskus PC, Boychuk CR, et al. Definitive evidence for the identification and function of renin-expressing cholinergic neurons in the nucleus ambiguus. Hypertension. 2025;82:282–92.
Elgazzaz M, Lakkappa N, Berdasco C, Mohan UP, Nuzzo A, Restivo L, et al. UBR1 promotes sex-dependent ACE2 ubiquitination in hypertension. Hypertension. 2025;82:84–95.
Ahwin P, Martinez D. The relationship between SGLT2 and systemic blood pressure regulation. Hypertens Res. 2024;47:2094–2103.
Oshima N, Onimaru H, Yamashiro A, Goto H, Tanoue K, Fukunaga T, Sato H, Uto A, Matsubara H, Imakiire T, Kumagai H. SGLT2 and SGLT1 inhibitors suppress the activities of the RVLM neurons in newborn Wistar rats. Hypertens Res. 2024;47:46–54.
Zhang S, Huang Y, Han C, Wang F, Chen M, Yang Z, et al. Central SGLT2 mediate sympathoexcitation in hypertensive heart failure via attenuating subfornical organ endothelial cGAS ubiquitination to amplify neuroinflammation: molecular mechanism behind sympatholytic effect of empagliflozin. Int Immunopharmacol. 2025;145:113711.
Kim DH, Lee MJ, Kang D, Lee JY, Park S, Khang AR, et al. Empagliflozin attenuates high-glucose-induced astrocyte activation and inflammation via NF-κB pathway. Curr Issues Mol Biol. 2024;46:12417–12427.
Elsaafien K, de Koet AD, Krause EG, Sumners C. Brain angiotensin type-1 and type-2 receptors in physiological and hypertensive conditions: focus on neuroinflammation. Curr Hypertens Rep. 2020;22:48.
Katsurada K. Interaction between SGLT2 and the sympathetic nervous system in normal and various cardiovascular metabolic disease states. Hypertens Res. 2025;48:2072–8.
do Carmo JM, da Silva AA, Wang Z, Fang T, Aberdein N, Perez de Lara CE, et al. Role of the brain melanocortins in blood pressure regulation. Biochim Biophys Acta Mol Basis Dis. 2017;1863:2508–14.
Guo DF, Williams PA, Olson A, Morgan DA, Herz H, Resch J, et al. Loss of melanocortin receptor accessory protein 2 in melanocortin-4 receptor neurons protect from obesity-associated autonomic and cardiovascular dysfunctions. Cardiovasc Res. 2025:121:1929–40.
Jama HA, Rhys-Jones D, Nakai M, Yao CK, Climie RE, Sata Y, et al. Prebiotic intervention with HAMSAB in untreated essential hypertensive patients assessed in a phase II randomized trial. Nat Cardiovasc Res. 2023;2:35–43.
Verhaar BJH, Wijdeveld M, Wortelboer K, Rampanelli E, Levels JHM, Collard D, et al. Effects of oral butyrate on blood pressure in patients with hypertension: a randomized, placebo-controlled trial. Hypertension. 2024;81:2124–36.
Welathanthree M, Keating DJ, Macefield VG, Carnevale D, Marques FZ, Muralitharan R. R. Cross-talk between microbiota-gut-brain axis and blood pressure regulation. Clin Sci. 2025;139:431–47.
Blanch GT, Freiria-Oliveira AH, Speretta GFF, Carrera EJ, Li H, Speth RC, et al. Increased expression of angiotensin II type 2 receptors in the solitary-vagal complex blunts renovascular hypertension. Hypertension. 2014;64:777–83.
Arnold AC, Gallagher PE, Diz DI. Brain renin–angiotensin system in the nexus of hypertension and aging. Hypertens Res. 2013;36:5–13.
Basalay MV, Korsak A, He Z, Gourine AV, Davidson SM, Yellon DM. SGLT2 inhibition induces cardioprotection by increasing parasympathetic activity. Circ Res. 2025;136:229–231.
Bousquet P, Hudson A, García-Sevilla JA, Li JX. Imidazoline receptor system: the past, the present, and the future. Pharmacol Rev. 2020;72:50–79.
Valensi P, Jambart S. Marriage: a randomized trial of moxonidine versus ramipril or in combination with ramipril in overweight patients with hypertension and impaired fasting glucose or diabetes mellitus. Impact on blood pressure, heart rate and metabolic parameters. J Cardiovasc Pharmacol Ther. 2024;29:10742484241258381.
Érszegi A, Viola R, Bahar MA, Tóth B, Fejes I, Vágvölgyi A, et al. Not first-line antihypertensive agents, but still effective—the efficacy and safety of imidazoline receptor agonists: a network meta-analysis. Pharmacol Res Perspect. 2024;12:e1215.
Schlaich MP, Tsioufis K, Taddei S, Ferri C, Cooper M, Sindone A, et al. Targeting the sympathetic nervous system with the selective imidazoline receptor agonist moxonidine for the management of hypertension: an international position statement. J Hypertens. 2024;42:2025–40.
Kitagawa K, Toi S, Hosoya M, Seki M, Yamagishi S, Hoshino T, et al. Small vessel disease burden predicts incident stroke and all-cause death, but not acute coronary event. Hypertens Res. 2024;47:3001–3009.
Ikeda S, Yakushiji Y, Tanaka J, Nishihara M, Ogata A, Eriguchi M, et al. Cerebral small vessel disease markers and long-term prognosis in spontaneous intracerebral hemorrhage: the HAGAKURE-ICH study. Hypertens Res. 2025;48:233–243.
Kario K, Okawara Y, Kanegae H, Tomitani N, Hoshide S. Peak nocturnal home blood pressure as an early and strong novel risk factor for stroke: the practitioner-based nationwide J-HOP Nocturnal BP study. Hypertens Res. 2025;48:622–631.
Oba Y, Kabutoya T, Kohro T, Imai Y, Kario K, Sato H, et al. Achievement of guideline-recommended target blood pressure is associated with reducing the risk of hemorrhagic and ischemic stroke in Japanese coronary artery disease patients -the CLIDAS study. Hypertens Res. 2025;48:632–639.
Ohya Y, Irie F, Nakamura K, Kiyohara T, Wakisaka Y, Ago T, et al. Association between pulse pressure and risk of acute kidney injury after intracerebral hemorrhage. Hypertens Res. 2025;48:939–49.
Ozawa T, Suzuki H, Miyata T, Kameda T, Kobari T, Tetsuka M, et al. Untreated and uncontrolled hypertension in Japanese patients with spontaneous intracerebral hemorrhage. Hypertens Res. 2025;48:1575–85.
Proietti R, Palazzolo MG, Ruff CT, Lip GYH, Giugliano RP. Long-term visit-to-visit blood pressure variability and risk of cardiovascular and bleeding events: insights from the ENGAGE AF-TIMI 48 trial. Hypertens Res. 2025;48:1613–8.
Sun YY, Pang SY, Qu Y, Wang SJ, Zhu HJ, Yin WJ, et al. Prognostic value of beat-to-beat blood pressure variability parameters in patients after mechanical thrombectomy. Hypertens Res. 2025;48:1155–62.
Acknowledgements
We appreciate the editors of Hypertension Research for giving us the opportunity to contribute to this mini-review article.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ikeda, S., Shinohara, K. Brain and hypertension: from sympathetic outflow to brain-focused blood pressure management. Hypertens Res 49, 749–755 (2026). https://doi.org/10.1038/s41440-026-02548-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-026-02548-1