Abstract
Aging is accompanied by low-grade intestinal inflammation, shifts in gut microbiota, and impaired oxidative balance. Probiotic supplementation has been proposed to mitigate these processes, yet evidence in elderly populations remains limited. In this pilot trial, older adults received oral Lactiplantibacillus plantarum OL3246 or placebo, with assessments including fecal calprotectin and zonulin as markers of intestinal inflammation, systemic oxidative stress parameters, self-reported quality of life and mood, and gut microbiome composition analyzed by sequencing and functional profiling. L. plantarum OL3246 supplementation was well tolerated and associated with consistent improvements across clinical, biochemical, and microbial measures. Participants reported enhanced quality of life and mood, while fecal calprotectin levels declined, indicating reduced intestinal inflammation. Moreover, oxidative stress markers improved with lower AOPP, stabilization of SOD, and restoration of redox balance. Microbiome analyses showed greater diversity and enrichment of health-associated taxa. These findings indicate that Lactiplantibacillus plantarum OL3246 may support healthy aging.
Similar content being viewed by others
Introduction
In recent decades, the idea of extending lifespan while maintaining good quality of life has gained growing attention across both scientific and commercial fields. This shift is mainly driven by the global aging of populations, a parallel rise in chronic age-related diseases (CARDs), and the mounting costs of elderly healthcare. Forecasts suggest that aging-related health expenditures will increase by 1.8% of GDP in Japan and 1.3% in the EU in the coming decades1. These arguments emphasize the importance of strategies aimed at improving quality of life throughout aging, especially in the context of CARDs, which profoundly influence individual well-being, healthcare costs, and social participation2,3,4,5,6.
As a biological process, aging is characterized by cellular and molecular hallmarks such as genomic instability, mitochondrial dysfunction, telomere shortening, cellular senescence, and chronic low-grade inflammation7. These changes contribute to the development of numerous age-related conditions, including metabolic disorders, sarcopenia, frailty, neurodegeneration, and immune dysregulation8,9. In recent years, increasing attention has been given to the gut microbiome as an important modulator of aging and disease progression, with dysbiosis, defined as functional imbalance in microbial ecosystem10, emerging as a key factor influencing both cellular aging and disease progression across the human lifespan11. Microbial imbalance compromises gut barrier integrity, leading to increased permeability (“leaky gut”), which in turn exposes the gut-associated immune system and systemic circulation to pathogen-associated molecular patterns (PAMPs) and toxic metabolites such as lipopolysaccharides and uraemic toxins. These molecules activate multiple proinflammatory mechanisms, contributing to low-grade systemic inflammation, tissue damage, and heightened vulnerability to chronic diseases12,13,14. Notably, microbial dysbiosis has been linked to the progression of neurodegenerative changes during aging through the gut–brain axis15. Dysbiosis-derived metabolites can adversely modulate neuroimmune signaling and vagal pathways, thereby increasing inflammatory and neurodegenerative processes16,17.
As a consequence, neurodegeneration contributes to a decline in quality of life in older individuals, frequently accompanied by the onset of depressive symptoms and progressive loss of functional independence. Its impact extends beyond cognitive decline, affecting emotional stability, motivation, and the ability to engage in meaningful social relationships. These dimensions (psychological and social well-being) are increasingly recognized as central determinants of healthy aging—understood as developing and maintaining the functional ability that enables well-being in older age18,19,20. High levels of well-being in later life are associated with lower incidence of chronic disease, greater adherence to treatment regimens, improved immune function, and reduced mortality risk. Conversely, disruptions in well-being can exacerbate physical decline, accelerate cognitive deterioration, and diminish quality of life, even in the absence of overt disease. In this context, preserving well-being is not only a marker of resilience, but also a target for preventive strategies aimed at maintaining autonomy and life satisfaction among older individuals21,22,23. Addressing neurodegenerative-related impairments in healthy older adults remains particularly complex, not only due to their multifactorial etiology but also because of the lack of effective therapies that could relieve symptoms. Consequently, there is an urgent need to explore alternative strategies aimed at mitigating or delaying neurodegenerative-related impairments. That approach is integral with the concept of healthy aging, which emphasizes the preservation of quality of life and the attenuation of low-grade inflammation24.
To date, several gut microbiome-targeted interventions have been explored, particularly those aligned with a healthy diet. Promising strategies include high-fiber diets, which enhance short-chain fatty acid (SCFA) production and reshape microbial composition; polyphenol-rich food, known for its antioxidant properties and support of beneficial taxa; and finally, probiotic supplementation. Probiotics have garnered growing attention for their potential to affect a broad range of physiological processes. A growing body of evidence supports their beneficial role in metabolic, cardiovascular, gastrointestinal, and even oncological conditions25. By modulating the microbiome and engaging in both direct and indirect interactions with host cells, probiotics exhibit promising anti-inflammatory and anti-cancerogenic potential, mainly in preliminary studies and in small trials26,27. Notably, emerging research suggests that they may ameliorate neurodegeneration-related symptoms or biomarkers associated with aging through the gut–brain axis28,29.
However, despite this potential, there is a notable lack of clinical studies that address probiotics as a preventive agent or modulator of quality of life in healthy elderly populations. To our knowledge, only a limited number of clinical trials have investigated specific probiotic strains in this context30,31,32,33. The main body of evidence comes from preclinical animal models, and it remains a significant challenge to determine how these findings translate to humans.
Therefore, it is essential to evaluate the mechanisms through which probiotics act on the human gut and gut–brain axis, as well as their clinical effectiveness in supporting well-being in older adults, particularly those living independently, who may be at elevated risk for emotional decline and loss of life satisfaction. To address this gap, we conducted a randomized, double-blind, placebo-controlled, pre–post pilot trial to test the hypothesis that probiotic supplementation could improve intestinal health and contribute to the maintenance of psychological and emotional well-being in healthy older individuals.
Methods
Study design
The study has been conducted as a randomized, pre–post, placebo-controlled trial in healthy elderly adults with no signs of neurodegenerative diseases and in general good health. The main hypothesis was how the administration of the probiotic Lactiplantibacillus plantarum OL3246 (deposit number: KPD 1760) for 3 months would affect quality of life (primary outcome), inflammatory status measured in blood and stool samples, as well as microbiome composition.
Participant enrollment
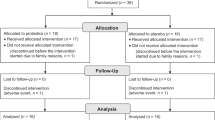
The total number of participants recruited to the study was 15 per group (Table 1). During the study, 4 participants were rolled out, due to external factors (4—COVID infections, 1—resignation). By the end of the trial, overall retention was ~86.6%. Minor differences in sample size across analyses reflect outlier removal and occasional mismatches or missing data between questionnaire, serum, and fecal samples. The study was conducted during the period: June 1–November 15, 2023.
Inclusion criteria: age between 55 and 85 years, generally good overall health, the absence of any exclusion, and MMSE score > 24.
Exclusion criteria: diagnosed dementia, parkinson disease, diabetes mellitus, clinically active depression (BDI > 26 and positive clinical diagnosis), substance use disorders or intoxication, uncontrolled hypertension, unstable ischemic heart disease, allergies, particularly severe allergic reactions, other serious medical conditions that, in the investigator’s opinion, could interfere with test performance or supplement tolerance, use of psychotropic medications, use of antibiotics and probiotics during past 3 months, major changes in diet in last 3 months. The use of prebiotics was not controlled in the protocol. Another limitation of this study is that sleep patterns and physical activity were not specifically controlled or monitored. These lifestyle factors can influence gut microbiota composition and inflammatory markers; however, the study was conducted under free-living conditions to reflect real-world probiotic use.
The qualification and verification of participants have been made by neurologists from the Academy of Physical Education in Katowice and the Medical University of Silesia. The study has been registered under the Resolution No. 6/2022 of the University Bioethics Committee for Scientific Research at the Jerzy Kukuczka Academy of Physical Education in Katowice, dated June 23, 2022, regarding the opinion on the proposed medical experiment.
Randomization and blinding
Permuted-block randomization with a fixed block size of four (2:2 ratio) was generated using a computer algorithm. Both participants and researchers were blinded to the group allocation. Additionally, the randomization sequence was kept confidential until study completion and implemented by a trained assistant, who assigned participants to the intervention or Placebo group strictly according to the pre-generated sequence.
-
1.
Intervention group/probiotic group: Tested formulation had a pharmaceutical form of capsules, containing Lactiplantibacillus plantarum OL3246 (deposit number: KPD 1760) lyophilizate (further: OL3246) and excipients such as maltodextrin, magnesium stearate, and silicon oxide (1.4 × 1010 CFU per day; taken orally as a 370 mg capsule containing 7 × 109 CFU/capsule twice a day: morning and evening).
-
2.
Placebo group: Placebo capsules contained an equal mass of fulfillment, while the probiotic was substituted by maltodextrin (taken orally as a 370 mg capsule twice a day: morning and evening).
The study product was developed and manufactured in-house at Olimp Laboratories Sp. z o.o. Dębica, Poland, by the research and development team, who are co-authors of this study, in compliance with Good Manufacturing Practice (GMP) guidelines and all relevant quality assurance standards. The appearance (shape, size, and packaging) of both products was identical. Before administration to participants, both products were examined to align with local guidelines of food and dietary supplement manufacturing to confirm the safety and quality of the product.
Study capsules were maintained in cool storage (4 °C). Participants were instructed to store capsules in their home fridge, and to contact the study centre if they were accidentally left at room temperature for more than 2 days, or left in the hot sun for 30 min or more; under these circumstances, replacement capsules were issued.
Short Form Health Survey (SF-36)
Quality of life was assessed using the Short Form Health Survey (SF-36), a widely used, multidimensional instrument developed to evaluate health-related quality of life (Ware and Sherbourne, 1992). For the purposes of this study, quality of life was assessed using the Polish version of the SF-36 questionnaire, adapted by Tylka and Piotrowicz34. This version yields a total score ranging from 0 to 171 points, where higher scores indicate a lower quality of life. The maximum possible scores for the physical and mental components are 103 and 68 points, respectively. The questionnaire was applied at baseline and post-intervention to detect changes in perceived health status.
Beck’s Depression Inventory scale (BDI)
Depressive symptoms were assessed using the Beck Depression Inventory (BDI), a widely validated self-report questionnaire commonly used to monitor both the severity and changes in depressive symptomatology. In line with current knowledge, the BDI is suitable for evaluating depressive symptoms across various populations, including older adults. The version with 21 questions was used. Scores range from 0 (no depression symptoms) to 63 (severe depression symptoms)35. A cut-off score of 11 points has been recommended for identifying clinically relevant depressive symptoms in several populations, including the Polish population. Scores above this threshold are indicative of depressive disturbances, with a reported sensitivity of 82.5%36,37.
Safety assessment
Safety was monitored throughout the study. At the initial visit, participants received a diary and were instructed to record any adverse effects (solicited and unsolicited) occurring during the 12-week supplementation and the subsequent 2-week follow-up. Participants were also instructed to promptly report any adverse event to the principal investigator. Additionally, possible adverse effects were assessed during each study visit. The predefined grading system included three levels of severity (mild, moderate, severe), and each event, if reported, was to be classified as related, possibly related, or unrelated to the study product. No adverse events (AEs) or serious adverse events (SAEs) were reported in either study arm. No participant discontinued the intervention due to safety reasons.
Analysis of α-synuclein level
Total human α-synuclein concentrations in serum samples were determined using Human α-Synuclein ELISA Kit (Invitrogen™, Thermo Fisher Scientific, Waltham, MA, USA #KHB0061) according to the manufacturer’s instructions. In short, 50 µL of standards, controls, or serum samples were pipetted into appropriate wells pre-coated with an anti-α-synuclein antibody. Subsequently, 50 µL of HRP-conjugated detection antibody was added to each well. Plates were incubated for 3 h at room temperature and then washed four times with a washing buffer. Next, 100 µL of freshly prepared Anti-Rabbit IgG-HRP solution was added to each well and incubated for 30 min at room temperature. After a second wash step, 100 µL of TMB substrate solution was added and allowed to develop in the dark for 30 min. The enzymatic reaction was terminated by the addition of 100 µL Stop Solution, and absorbance was measured at 450 nm using a microplate reader.
Analysis of advanced oxidation protein products (AOPP)
The level of advanced oxidation protein products (AOPP) was determined using the AOPP Assay Kit (#ab242295; Abcam, Cambridge, UK) according to the manufacturer’s instructions. Serum samples and chloramine standards were loaded in a volume of 200 µL per well in a 96-well plate. Subsequently, 10 µL of Chloramine Reaction Initiator was added to each well, followed by incubation on a shaker for 5 min at room temperature. The reaction was stopped by adding 20 µL of Stop Solution. Absorbance was measured immediately at 340 nm using a microplate reader. AOPP concentrations were calculated based on a standard curve generated from chloramine solutions of known concentrations.
Evaluation of superoxide dismutase (SOD) level
Total superoxide dismutase (SOD) concentration was quantified using the Human Total Superoxide Dismutase ELISA Kit (#A247278; antibodies.com, Stockholm, Sweden) according to the manufacturer’s protocol. Briefly, 100 µL of standards and approximately 1:2-diluted samples were added to wells of a microtiter plate and incubated at 37 °C for 90 min. After washing the plate twice with Wash Buffer, 100 µL of biotinylated detection antibody was added to each well, followed by a 60 min incubation at 37 °C. Plates were washed three times, then incubated with 100 µL of HRP–streptavidin conjugate (SABC) at 37 °C for another 60 min. After five additional washes, 90 µL of TMB substrate was added and the plate was incubated in the dark at 37 °C for 10–20 min. The reaction was stopped by adding 50 µL of Stop Solution to each well. Absorbance was measured immediately at 450 nm using a microplate reader.
Stool samples collection
Fecal samples were collected at the end of the intervention period. Participants received detailed verbal and written instructions and were provided with a dedicated sampling kit comprising a sterile stool collection tool (Kałszyk, Kosowski, Wąchock, Poland) and a short questionnaire to document participant ID, date, time of collection, and relevant metadata. Samples were transported at 4 °C and frozen at −80 °C within 2 h of delivery to ensure preservation of microbial integrity. Frozen samples were subsequently used for biomarker and microbiota analyses.
Evaluation of zonulin level
Fecal zonulin levels were quantified using a competitive IDK® Zonulin ELISA (#K5600; Immundiagnostik AG, Bensheim, Germany). At first, stool samples were extracted using a dilution buffer and yielding a 1:50 dilution. Then, extracted stool samples, standards, and controls were mixed with biotinylated ZFP tracer. After vortexing, 100 µL of each mixture was transferred to wells pre-coated with polyclonal anti-ZFP antibodies and incubated for 1 h at room temperature. Following five washes, 100 µL of peroxidase-conjugated streptavidin was added and incubated for another hour under identical conditions. After washing, 100 µL of TMB substrate was added and incubated in the dark for 25 min. The reaction was stopped by adding 100 µL of stop solution, and absorbance was measured at 450 nm with a reference at 620 nm.
Analysis of calprotectin level
Fecal calprotectin (MRP8/14) levels were measured using the IDK® Calprotectin ELISA kit (#6927, Immundiagnostik AG, Bensheim, Germany), strictly following the manufacturer’s instructions. At the beginning, stool samples were extracted using an extraction buffer and yielding a final dilution factor of 1:2500. Then, standards, controls, and diluted stool extracts were pipetted into wells of a microtiter plate pre-coated with a monoclonal anti-calprotectin antibody. The plate was incubated for 30 min at room temperature 22 °C. After five washes with 250 µL of wash buffer, 100 µL of HRP-conjugated detection antibody was added to each well and incubated for another 30 min. Following another wash step, 100 µL of TMB substrate was added and incubated for 25 min in the dark. The enzymatic reaction was stopped by adding 100 µL of stop solution, and absorbance was measured at 450 nm with a 620 nm reference wavelength.
Microbiome analysis
Microbial DNA was extracted from stool samples using the Genomic Mini AX Stool Kit (#065-60, A&A Biotechnology, Gdańsk, Poland) following the manufacturer’s protocol. Briefly, ~100 mg of stool was lysed in the presence of proteinase K, and DNA was purified using silica spin columns with precipitation and ethanol wash steps. DNA was eluted in Tris buffer and stored at −20 °C until further processing. Library preparation was carried out using a two-step PCR protocol. The first PCR involved amplification of the target 16S rRNA gene region with primers containing adapter overhangs. In the second PCR, sample-specific indices and Illumina sequencing adapters were added. Sequencing was performed on an Illumina platform using paired-end reads with a minimum of 2 × 300 bp read length. Microbiome analysis was performed using QIIME2 (version 2022.11)38. Demultiplexed paired-end reads were processed with the DADA2 plugin for quality filtering, denoising, chimera removal, and inference of amplicon sequence variants (ASVs)39. Taxonomic classification of ASVs was conducted using a pre-trained Naïve Bayes classifier specific to the 515F/806R region, trained on the SILVA 138.2 reference database40. Beta-diversity was assessed using UniFrac distance metrics41, and group differences were tested with PERMANOVA42. Differential abundance analysis was performed with the Analysis of Compositions of Microbiomes with Bias Correction (ANCOM-BC) method43.
Prediction of metabolic changes in microbiome (picrust2 + LinDA)
Functional pathway prediction was performed using PICRUSt2 (Phylogenetic Investigation of Communities by Reconstruction of Unobserved States44), based on 16S rRNA gene amplicon data. Raw sequencing data were processed using QIIME2 (version 2022.1138), following standard workflows for denoising (via DADA2), taxonomic assignment, and phylogenetic tree construction. The resulting feature table and phylogeny were used as inputs to the PICRUSt2 pipeline to infer MetaCyc pathway abundances. This approach has been widely adopted in similar microbiome studies to estimate community-level functional potential from marker gene data45,46,47.
The predicted pathway abundance table (compositional) was analyzed using LinDA48, a statistical framework designed for differential analysis of microbiome functions, which accounts for compositionality and sparsity in relative abundance data. Pathways were considered differentially abundant at an unadjusted p-value < 0.05 and absolute log₂-fold change > 1, acknowledging the exploratory nature of the study. Visualization of pathway-level results, including volcano plots and category-based summaries, was performed using ggplot249 in R.
Due to the exploratory and pilot nature of this study, with a total sample size of n = 21, we did not expect to detect statistically significant differences after multiple testing corrections. Accordingly, we report results based on unadjusted p-values to retain sensitivity and avoid type II errors associated with overcorrection in small-sample, high-dimensional datasets. This approach is in line with prior recommendations for pilot microbiome research, where the focus is on hypothesis generation rather than formal inference50. Pathways with unadjusted p-values below 0.05 and an absolute log2-fold change > 1 were considered differentially abundant for exploratory purposes. These results should be interpreted as putative signals warranting follow-up in larger, confirmatory studies.
Statistical analysis
Statistical analyses were conducted using a linear mixed-effects model with restricted maximum likelihood estimation (REML), specified as: SF36 ~ Group* Time + baseline+(1|Patient_ID), where Group (Placebo, L.plantarum OL3246) and Time (Pre, Post) were fixed effects, and subject ID was included as a random intercept to account for repeated measures. Prior to model fitting, assumptions were tested by: detecting outliers using the interquartile range (IQR) method; assessing homogeneity of variance using Levene’s test; evaluating normality of residuals via Q–Q plots.
Estimated marginal means (EMMs) and pairwise contrasts were obtained using the emmeans package with Kenward–Roger approximation for degrees of freedom. Effect size (Hedges’s g) was computed using the eff_size() function. Significance was set at p < 0.05. All analyses were performed in R (version 2024.12.1 + 563).
The same linear mixed-effects modeling strategy described for SF-36 was applied to the BDI scale and biochemical parameters using the model: Variable (BDI/Biochemical parameter) ~ Group* Time + baseline +(1|Patient_ID). Diagnostic checks and statistical procedures were applied identically. Estimated marginal means were derived using emmeans, and group-by-time interactions were interpreted with pairwise contrasts. For transparency, all Group × Time interaction estimates are provided in Supplementary Table S1. Models were additionally adjusted for sex as a sensitivity check; inclusion of sex did not alter the direction or interpretation of effects. Given the exploratory nature of this pilot study, the main text focuses on simple effects (within- and between-group contrasts) to illustrate observed trends.
Results
Baseline characteristics of the study population
The baseline demographic and clinical characteristics of the participants are shown in Table 1. Taken together, these data indicate that the recruited cohort was generally healthy, without serious liver, kidney, and metabolic diseases. Throughout the study period, no adverse events (AEs) or serious adverse events (SAEs) were observed in either study arm during the 12-week intervention and the 2-week follow-up period.
Lactiplantibacillus plantarum OL3246 improves quality of life and depressive symptoms
Aging is frequently accompanied by a gradual decline in perceived quality of life and a higher prevalence of mood-related disturbances, including depressive symptoms. These psychological dimensions are not only critical indicators of individual well-being, but also correlate with physiological aging and disease burden51. To evaluate the potential impact of L. plantarum OL3246 on subjective health perception and emotional status, we assessed participants using the SF-36 health survey and the Beck Depression Inventory (BDI) at baseline and following the intervention. Both instruments are well-established tools for quantifying multidimensional aspects of quality of life and depressive symptomatology, respectively.
As shown in Fig. 1, the linear mixed-effects model revealed statistically significant changes in SF-36 scores in the L. plantarum OL3246 group compared to Placebo at post-intervention (estimate = 2.547, p = 0.039). No significant differences were observed at baseline between the groups (p = 0.612). Within-group comparisons showed a clinically meaningful and statistically significant decrease in SF-36 scores from pre- to post-intervention in the L. plantarum OL3246 group (estimate = −4.930, p < 0.001), while changes in the Placebo group were not significant (p = 0.183) (Fig. 1; Supplementary Fig. S1). The Hedges’s g effect size was 0.76, indicating a medium magnitude of improvement, although the 95% confidence interval [−0.113, 1.631] suggests some uncertainty due to sample size. These findings suggest that the probiotic intervention may positively impact patient-reported quality of life in the selected cohort, as measured by SF-36.
Pre/Post comparisons of SF-36 (a, blue-pre, orange-post) and BDI (b, green-pre, orange-post) between the Placebo and Intervention group have been made using REML ANOVA with Tukey post-hoc comparison; outliers have been filtered using the IQR method. For individual pre-to-post (Δ) changes in each scale, refer to Supplementary Fig. S1. Significance code: ns non-significant, *p < 0.05, **p < 0.01, *** p < 0.001, ****p < 0.0001.
In accordance with Beck’s depression inventory results, we observed the same pattern as in SF-36. The L. plantarum OL3246 group improved by 1.29 points in BDI (p = 0.0009), whereas the Placebo group showed no significant changes over time. The between-group contrasts at post-intervention were marginally significant (p = 0.050), suggesting a trend toward probiotic-associated benefit (Fig. 1; Supplementary Fig. S1). Effect size estimates indicated a small, but biologically meaningful effect due to low baseline levels (g = 0.36), with wide confidence intervals, which reflected sample-related uncertainty.
Administration of Lactiplantibacillus plantarum OL3246 improves specific markers of protein oxidation and lowers levels of alpha-synuclein
One of the few blood-based biomarkers that worsens over time and may contribute to decreased quality of life and increased disease risk is disrupted redox homeostasis. Excessive production and accumulation of free radicals, coupled with inadequate antioxidant defenses, can result in extensive molecular damage, including protein oxidation and misfolding, DNA strand breaks, and lipid peroxidation52. These deleterious processes are recognized contributors to the progression of neurodegenerative diseases and reduced well-being53. Given this knowledge, we pre-selected two redox markers in the study design—advanced oxidation protein products (AOPP) and superoxide dismutase (SOD)—to monitor inflammatory and oxidative status across the study period. Additionally, we correlated the pre–post changes in SOD with those in α‑synuclein levels to explore whether modulation of oxidative defense aligns with alterations in a protein involved in neurodegenerative processes.
The intervention group exhibited a statistically significant reduction in advanced oxidation protein products (AOPP) compared to the Placebo group (p < 0.001) and in pre-post comparison (p < 0.0001), indicating a lower burden of systemic oxidative damage (Fig. 2). This reduction suggests that the L. plantarum OL3246 effectively modulated redox homeostasis, potentially attenuating consequences of increased oxidation. Regarding superoxide dismutase (SOD), we observed a significant pre-to-post increase in circulating SOD levels only in the Placebo group, whereas levels remained stable in the Probiotic group over time. Additionally, when adjusted for baseline values, endpoint SOD concentrations did not differ significantly between groups, suggesting that the intervention maintained redox stability, while the Placebo group may have mounted a compensatory antioxidant response to persistent oxidative stress. To further investigate this relationship, we analyzed the correlation between changes in SOD (ΔSOD) and α-synuclein (Δα-synuclein), one of the important proteins associated with proteotoxicity and neurodegeneration. In the Placebo group, a strong positive correlation was identified (r = 0.77, p = 0.015), indicating that individuals with greater increases in SOD levels also exhibited an increase in α-synuclein. Conversely, the L. plantarum OL3246 group showed a significant inverse correlation (r = –0.68, p = 0.044), suggesting that the treatment may have disrupted the oxidative stress–α-synuclein axis. Comparison of pre- and post-change of α-synuclein in serum between the Placebo and L. plantarum OL3246 group can be found in Fig. S2 and Table S2. Collectively, these findings support the hypothesis that the intervention may beneficially modulate oxidative status, as evidenced by lower AOPP concentrations and more stable SOD levels. While these changes are intriguing from a mechanistic perspective, they should be regarded as preliminary and exploratory. Overall, the results suggest a potential link between redox homeostasis, improved quality of life (SF-36), and mood-related outcomes (BDI), which merits further investigation in larger and mechanistically focused studies.
Pre/Post comparisons of AOPP (a; orange-pre, blue-post) and SOD (b; orange-pre, purple-post) between the Placebo and Intervention group have been made using REML ANOVA with Tukey post-hoc comparison; outliers have been filtered using the IQR method. Delta—change of ∝-synuclein (c) has been compared using the Welch t-test. Delta’s of SOD and ∝-syn have been correlated (d; grey—placebo; green—L. plantarum OL3246) by the Pearson method—R and p values are presented in the plot. Significance code: ns—non-significant, *p < 0.05, **p < 0.01, *** p < 0.001, ****p < 0.0001.
Improved inflammatory markers in intestine after Lactiplantibacillus plantarum OL3246 administration
One of the best-described mechanisms and effects of probiotics in the elderly population is the modification of the gut microbiome and inflammation-related markers30. Given that the probiotic formulation was administered orally, its primary site of action was presumed to be the gastrointestinal tract. Accordingly, we assessed gut-related outcomes by analyzing differences in microbiota composition between study groups and across timepoints. In parallel, we measured fecal concentrations of zonulin and calprotectin, two well-established biomarkers associated with intestinal barrier function and mucosal inflammation. Both markers are frequently elevated in older adults and may reflect age-related increases in intestinal permeability and low-grade gut-derived inflammation54. Mentioned changes could be a seminal point of the inflammatory cascade, which propagates to the blood and then to the brain, leading to increased protein misfolding and neurodegeneration55.
We observed significant differences in fecal Calprotectin levels in comparison between Placebo and Intervention at the end of the experiment (Fig. 3). In the Placebo group, the mean concentration was approximately 104 µg/g, with a median of 109 µg/g. In contrast, the intervention group demonstrated substantially lower values, with a mean of 54 µg/g and a median of 30 µg/g, placing them around or below the reference threshold. Despite the lack of pre-intervention data about fecal biomarkers, this pattern indicates a beneficial effect of the intervention on fecal Calprotectin levels. The difference between the Placebo and intervention groups was statistically significant (p = 0.023). The analysis of zonulin levels did not reveal any changes due to the probiotic treatment.
The levels of zonulin (a) and calprotectin (b) have been analyzed at the end of the experiment. Statistical comparison has been made using the Welch t-test. Significance code: ns non-significant, * p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
Administration of Lactiplantibacillus plantarum OL3246 modulates microbiome in healthy older people
Aging-related changes in the gut microbiome have been increasingly linked to systemic inflammation, oxidative stress, and neurodegeneration—all of which were reflected in the physiological and psychological parameters evaluated in this study. To better understand the possible microbial underpinnings of the observed improvements in quality of life, mood, and redox status following L. plantarum OL3246 administration, we next examined whether the intervention affected the global structure of gut microbiota. We focused on beta diversity as a metric of between-sample dissimilarity in microbial community structure, providing insight into ecological variation across individuals and experimental conditions. Changes in beta diversity can reflect dynamic restructuring of the gut microbiome in response to external interventions. Increases in beta diversity are often interpreted as indicators of different microbial community structure, potentially signifying a shift toward a more balanced and health-associated community configuration56.
We selected weighted UniFrac as the primary beta-diversity metric because it captures differences in both taxonomic abundance and phylogenetic relationships, which is essential in interventions expected to modulate rather than replace existing microbial communities (Fig. 4). This makes it better suited to detect shifts in dominant lineages that may impact microbial function or host interaction. To statistically verify significance, we performed the PERMANOVA test and obtained results (pseudo-F = 2.35, p = 0.021).
Three two-dimensional PCoA plots depict the spatial clustering of samples along the first three principal coordinates: a PCo2 vs. PCo3 (explaining 16.17% and 13.56% of variation, respectively), b PCo1 vs. PCo3 (37.32% and 13.56%), c PCo1 vs. PCo2 (37.32% and 16.17%). Samples are color-coded by group: blue for Placebo, and red for L. plantarum OL3246. Group separation was statistically evaluated using PERMANOVA (n = 21 samples, 2 groups, 999 permutations), suggesting a significant difference in beta diversity between groups (pseudo-F = 2.354254, p-value = 0.028, q-value = 0.028).
A statistically significant difference in weighted UniFrac beta diversity was observed between the Intervention and Placebo groups, indicating distinct microbial community structures. The calculated effect size (pseudo-F ≈ 2.35) reflects a modest, yet biologically relevant shift in overall composition, suggesting partial overlap between groups alongside a measurable intervention-induced divergence. This indicates that the two groups differ in their phylogenetic composition and in the relative abundance of taxa, not just in presence/absence. Further ANCOM-BC analysis on the family level revealed enrichment in Ruminococcaceae in the Probiotic group (p = 0.03) (Fig. 5). Deeper analysis uncovered that Fecalibacterium (p = 0.035) and Subdoligranulum (p = 0.083) were responsible for differences in beta-diversity. On the species level, we observed enrichment of Fecalibacterium prausnitzii and undefined species of Subdoligranulum and Dialister genus and Clostridia class. All of the mentioned changes had an effect size > 0.4. Additionally, we observed three other switches, whose log-fold change was >0.4: Bacteroides uniformis (lfc = 0.4), genus Eubacterium coprostanoligenes group (lfc = 0.52), and Akkermansia muciniphila (lfc = −0.49), notwithstanding that these results were not statistically significant.
Each point represents a microbial feature (taxon). The x-axis displays the estimated log-fold change (LFC) in relative abundance between the Placebo and L. plantarum OL3246 groups, while the y-axis represents the corresponding statistical significance as −log₁₀ of the raw p-value. Features with nominal significance (p < 0.05) are highlighted in green; non-significant in red. Features with moderate or large effect sizes (|LFC| > 0.4) are emphasized with a bold outline; those with |LFC| < 0.4 are emphasized with a narrow outline. Labeled points indicate features with p < 0.05, annotated with their taxonomic identity and corresponding FDR-adjusted q-value. Dashed vertical lines denote thresholds for moderate effect size (LFC = ±0.4), and the horizontal dashed line indicates the nominal significance threshold (p = 0.05). ANCOM-BC43 has been performed using a dedicated Qiime2 plugin. To improve clarity, only the last assigned taxonomic level has been displayed on the chart.
Given that compositional shifts in the gut microbiome can influence its functional output, we next examined predicted metabolic pathway profiles to assess whether the observed taxonomic differences translated into changes in microbial metabolic potential. Functional prediction was performed using PICRUSt2, which infers community-level pathway abundance from 16S rRNA gene data44. This approach allows for the estimation of biologically meaningful alterations in metabolic activity, including those relevant to host–microbe interactions, immune modulation, and redox homeostasis. The following results present key pathway-level differences between the intervention and placebo groups (Fig. 6).
Volcano plot showing log₂-fold changes (x-axis) and statistical significance (−log₁₀(p-value); y-axis) for predicted MetaCyc pathways inferred using PICRUSt2 from 16S rRNA gene data. Each point represents a single metabolic pathway. Pathways were stratified by functional category and colored accordingly (red—carbohydrate metabolism, blue—polyamine/nucleotide metabolism, green—vitamin metabolism). Only pathways with an unadjusted p < 0.05 and |log₂FC| > 1 (dashed lines) were considered differentially abundant in this exploratory analysis. Gray points denote pathways that did not meet these thresholds. Statistical testing was performed using LinDA, and visualization was created in R using ggplot2.
Differential abundance analysis revealed several microbial metabolic pathways with unadjusted p-values < 0.05 and absolute log2-fold changes > 1. Notably, pathways related to carbohydrate metabolism (e.g., GALACT-GLUCUROCAT-PWY, GLUCUROCAT-PWY), vitamin biosynthesis (COBALSYN-PWY, FOLSYN-PWY), and polyamine/nucleotide biosynthesis (POLYAMINSYN3-PWY) were consistently upregulated in the probiotic group. Although none of the pathways remained significant after false discovery rate (FDR) adjustment, these uncorrected results offer biologically plausible leads that align with prior findings in gut microbiome research and justify further targeted investigation. A volcano plot illustrating these changes highlights the functional categories with the strongest exploratory signals and possible microbiome-related mechanisms modulated by L. plantarum OL3246 administration. Positive influence on the mentioned pathways may be a possible explanation for how OL3246 modulates microbial composition. Exact laboratory verification, whether OL3246 stimulates synthesis in the microbiome (via Clostridia enrichment) or can produce particular vitamins on its own, should be a target of future studies.
Discussion
Maintaining gastrointestinal health becomes increasingly challenging with age, as the elderly typically exhibit a decline in beneficial taxa such as Bifidobacterium and Lactobacillus, accompanied by an expansion of pro-inflammatory groups, including Enterobacteriaceae57. Such dysbiotic alterations are associated with a range of health complications that negatively impact quality of life58. Probiotic supplementation offers a strategy to re-establish a more balanced microbial ecosystem, thereby potentially counteracting some of these age-related shifts.
Probiotics have shown considerable promise in improving the quality of life among patients with gastrointestinal diseases59,60. Comparable benefits have also been observed in the older population, where probiotic supplementation alleviated age-associated constipation and, in turn, contributed to enhanced quality of life61,62. However, studies on the influence of probiotics on QoL in healthy elderly populations, where gastrointestinal diseases and symptoms are less prevalent, do not provide clear and consistent results. The lack of consistent findings largely reflects heterogeneity in study design, including the use of different assessment tools and methodological approaches30,31,33. In our study, the L. plantarum OL3246 group showed a statistically significant improvement in QoL assessment, while the Placebo group did not. Post-intervention comparison between groups, adjusted for baseline using a REML model, also favored the intervention. Although the observed change was slightly below the commonly referenced MCID thresholds (4–7 points for 0–100 tests), even modest improvements may carry clinical and practical significance in generally healthy, high-functioning older adults, where baseline impairment is minimal. In this context, subtle shifts in QoL measures may reflect enhanced well-being and resilience, which are particularly valuable outcomes in preventive interventions. Importantly, the SF-36 results paralleled the improvements on the BDI scale, strengthening the validity of the observed effects across distinct yet complementary domains of health. According to Button et al.63, a reduction of ~17.5% from baseline constitutes a clinically meaningful improvement from the patient’s perspective. In our study, the mean BDI score in pre–post comparison for the L. plantarum OL3246 group decreased from 7.18 to 5.88, corresponding to a reduction of 18.0%. Although both values remained below the clinical cut-off (scores < 13), the observed relative change surpassed the established MCID threshold, indicating a potentially meaningful enhancement in mood and emotional well-being, relevant in a healthy, community-dwelling female-predominant older population. These findings underscore the potential value of interventions designed to promote emotional stability, even among individuals without clinically manifest symptomatology64. Epidemiologic data provide a nuanced picture of mood health in older adults. Large-scale surveys, such as the 2021–2023 U.S. National Health and Nutrition Examination Survey, showed that the prevalence of major depressive disorder tends to decline with age, from 19.2% in adolescents to 8.7% among adults aged ≥60 years65. However, this pattern coexists with a substantial burden of subthreshold depressive symptoms and psychological distress later in life. In the pan-European SHARE cohort (n ≈ 45,600; ≥65 years), clinically relevant depressive symptoms (EURO-D ≥ 4) affected 35.1% of women and 21.5% of men66, and meta-analyses report pooled prevalence estimates of 19–32% among older adults globally67,68. Mentioned numbers are much more alarming when compared to estimates from 1990 and to recent numbers of depressive symptoms in older adults, which have increased by 136.1%69. Such findings indicate that while the incidence of major depression may be lower than in younger cohorts, mood-related disturbances, reduced psychological well-being, and stress vulnerability remain highly relevant in aging populations. These conditions often accompany multimorbidity, neurobiological aging, and social isolation, underscoring the need for interventions that can modulate neuropsychological resilience and emotional health in later life—such as the probiotic strategies evaluated in the present study. Additionally, the presented results are in line with actual knowledge about probiotics and their anti-depressive effects70. However, there is still a lack of studies where the effectiveness of a single probiotic strain has been evaluated using the BDI scale. In all studies included in the meta-analysis, probiotics were administered in multistrain preparations70. This limited the possibility of direct comparisons, as potential synergistic interactions between strains obscured the specific contribution of L. plantarum within the tested formulations71. The only studies employing single-strain formulations of L. plantarum are those conducted with strains PS12872 and 299v73,74. PS128 has demonstrated efficiency in reducing depressive symptoms in individuals with insomnia, while 299v has shown benefits in patients diagnosed with major depressive disorder. Notably, however, there is no data that L. plantarum strains exert positive effects on mood or depressive symptoms in healthy elderly populations. Collectively, this suggests that the tested L. plantarum OL3246 strain could act as a positive modulator of mood and/or depressive symptoms in the female predominant group of older adults.
The positive clinical effects observed in our study may be mechanistically linked to the regulation of inflammatory pathways and/or redox homeostasis. A growing body of evidence indicates that probiotics can modulate host inflammatory response75, and may serve as adjunctive interventions for a wide spectrum of inflammatory diseases, including those located in the gastrointestinal tract76 and far beyond77,78,79. L. plantarum strains have emerged as one of the most comprehensively studied candidates for therapeutic application from a vast array of probiotics80, with well-established evidence of anti-oxidant properties81,82,83,84,85. However, as in the case of mood and QoL outcomes, evidence supporting the effectiveness of L. plantarum strains in older people remains limited, yet encouraging. For example, short-term supplementation with L. plantarum HEAL9 in healthy individuals over 70 years old was shown to reduce low-grade inflammation, as reflected by decreased fecal calprotectin levels86. In our study, we observed a pronounced effect of L. plantarum OL3246 on redox homeostasis, specifically through the stabilization of SOD and a reduction in AOPP levels. Given that SOD represents a primary enzymatic defense against reactive oxygen species, SOD activity is widely regarded as a key indicator of antioxidant capacity and cellular protection against oxidative stress87,88. In mammalian cells, both mitochondrial MnSOD and soluble Cu/ZnSOD genes are regulated by multiple signaling pathways, including up-regulation in response to oxidative stress89,90. In our study, we observed increased levels of SOD in pre-post comparison only in the Placebo group, whereas in the intervention group, SOD levels remained stable. These findings suggest an elevation of oxidative stress in the Placebo group and, conversely, indicate the capacity of L. plantarum OL3246 to mitigate this effect. This conclusion was further supported by the beneficial effect of L. plantarum OL3246 on AOPP levels. AOPP is considered one of the universal markers of oxidative stress with broad diagnostic and prognostic utility across various diseases, including chronic kidney disease and renal failure91,92, prostate and breast cancer93,94, cardiovascular95,96, as well as autoimmune and inflammatory diseases97. In healthy elderly individuals, AOPP levels are generally higher than in younger adults, reflecting an age-associated increase in oxidative protein damage and systemic oxidative stress98,99. Previous studies have reported mean AOPP concentrations in healthy older adults of approximately 80 μmol/L100, although values vary considerably depending on the studied population and analytical methodology. In our cohort, mean levels were around 50 μmol/L, placing them in the lower quartile of the reported 40–200 μmol/L range, which may be interpreted as relatively low oxidative damage. Nevertheless, the improvements observed both within and between groups in our trial suggest that L. plantarum OL3246 contributed to maintaining redox equilibrium and protecting against protein oxidation. These findings indicate that the tested strain may serve as a valuable protective agent even in subclinical or otherwise healthy elderly individuals, particularly in the context of age-related accumulation of AOPP as part of the physiological aging process, which needs further investigation.
Protein accumulation and impaired mechanisms of clearance are fundamental processes underlying numerous pathologies, particularly neurodegeneration, but they also play a critical role in physiological aging101. Given the close interconnection between redox homeostasis, protein misfolding, and aggregation, we assessed plasma levels of α-synuclein, one of the hallmark proteins associated with neurodegeneration, especially Parkinson’s disease (PD)102. Meta-analyses have clearly demonstrated that an increase in total α-syn in plasma correlates with the early stages of PD103, underscoring the importance of preventive strategies and the need to maintain a balanced interplay between oxidative stress and α-syn regulation. Our study provides exploratory evidence suggesting a potential relationship between oxidative stress markers (SOD) and α-synuclein expression. Although preliminary, this observation raises an interesting hypothesis that redox imbalance might influence α-synuclein dynamics, which in turn could be associated with aspects of mood and quality of life. Mechanistically, increased oxidative damage to neural tissue may upregulate α-synuclein as a compensatory response, since this presynaptic protein plays a key role in neurotransmitter release and synaptic stability. However, during sustained oxidative stress, excessive α-synuclein expression combined with oxidative modification may promote misfolding and aggregation, processes that can impair cognition, affect mood regulation, and ultimately contribute to neurodegenerative pathways. These findings are hypothesis-generating and require confirmation in larger mechanistic studies designed to clarify the interplay between redox homeostasis, α-synuclein regulation, and quality-of-life outcomes. To date, there is no clear evidence that other strains of L. plantarum influence AOPP, SOD, and α-syn levels, particularly in healthy populations. Existing clinical studies with L. plantarum remain scarce and typically report effects on single oxidative parameters, such as SOD activity104 or other redox single biomarker104,105. Our study, conducted with a single-strain formulation, provides novel insights into the relationship between temporal changes in α-syn and SOD. In the Placebo group, SOD levels positively correlated with rising levels of α-syn, suggesting co-activation of gene expression for both proteins in response to oxidative stress. In contrast, supplementation of L. plantarum decoupled this association, maintaining stable levels of SOD and preventing α-syn accumulation. This effect may reflect reduced ROS production and protein oxidation, thereby attenuating oxidative damage and preserving protein homeostasis. Importantly, disturbances in proteostasis and oxidative stress have been strongly implicated in the pathophysiology of mood disorders, including depression, where elevated oxidative markers are frequently reported. Thus, stabilization of redox balance and protein integrity may not only mitigate neurodegenerative risk but also contribute to improved emotional well-being and quality of life during physiological aging, as reflected in our findings.
The health-promoting effects of probiotics are largely attributed to their activity within the gastrointestinal tract (GI). A wide range of direct and indirect mechanisms of action has been described. Direct interactions with the GI tract include strengthening of the intestinal barrier, attenuation of inflammatory responses, and stimulation of mucin production106. Indirectly, probiotics may influence host physiology by modulating both the composition and metabolic activity of the gut microbiota, thereby altering the production of bioactive metabolites and signaling molecules. These mechanisms, whether acting individually or synergistically, are considered central to improvements in systemic health, including metabolic regulation, immune modulation, and overall well-being107. Importantly, such activities provide the rationale for evaluating biomarkers that reflect gut barrier function and inflammation. In this context, fecal levels of calprotectin and zonulin are particularly informative, even as collected only at the end of the experiment, as they represent sensitive indicators of intestinal permeability and mucosal immune activation.
Fecal calprotectin is a well-established marker of intestinal inflammation108. In healthy adults, concentrations below 50 µg/g are generally considered normal; however, levels tend to increase with age. Accordingly, higher reference values have been proposed for older individuals, with some suggesting a threshold of 112 µg/g for individuals over 60 years of age109,110,111. In the present study, calprotectin levels in the Placebo group exceeded the conventional adult threshold but remained at or slightly below the higher values regarded as physiologically normal in the context of aging and its associated low-grade inflammation111. By contrast, participants in the L. plantarum OL3246 group exhibited markedly lower mean and median values compared with Placebo, with the median falling well within the generally accepted normal range (<50 µg/g)111,112,113. In summary, these findings suggest that although calprotectin levels in the Placebo group may primarily reflect age-related, sub-clinical inflammation, L. plantarum OL3246 supplementation effectively reduced this marker to a range consistent with intestinal homeostasis. This outcome underscores the anti-inflammatory potential of the intervention and aligns with previous clinical trials reporting probiotic-mediated reductions in fecal calprotectin, particularly in populations with low-grade or subclinical intestinal inflammation86.
Zonulin, besides calprotectin, is increasingly recognized as a key regulator of intestinal permeability and has been implicated in the age-related phenomenon commonly referred to as “leaky gut.” However, the interpretation of absolute zonulin concentrations remains a matter of debate, as no universally accepted threshold reliably distinguishes between physiological and pathological permeability114,115,116. Some studies suggest that zonulin levels may rise with advancing age. For instance117, reported that serum zonulin concentrations were 22% higher in older adults compared with younger individuals. Conversely, other investigations, including that of Seethaler et al.114, found no significant association between zonulin levels and age. These inconsistencies highlight both the complexity of zonulin biology and the methodological variability across studies114,116,118,119,120. In the present study, zonulin levels fell within the range previously reported for healthy individuals114 and remained unaffected by probiotic supplementation. This finding suggests that L. plantarum OL3246 did not modify intestinal permeability through modulation of zonulin. Nevertheless, it cannot be excluded that other tight junction proteins, such as occludins or cadherins, may have been influenced. Moreover, a longer intervention period or higher dosage may be required to fully reveal potential probiotic effects on barrier function. Despite the study limitations (single-end measurement of fecal biomarkers) and the lack of change in zonulin, the observed reduction in calprotectin levels and alterations in gut microbiota composition suggest that the intervention could bring a beneficial influence on intestinal inflammatory status.
Continuing, although changes in zonulin were not detected, the intervention exerted measurable effects at the microbial level. Shifts in community composition, particularly the enrichment of health-associated taxa, may provide a mechanistic explanation for the observed improvements in inflammatory status. Observed taxonomic differences in gut microbial composition between Placebo and L. plantarum OL3246 groups may underlie the reductions in fecal calprotectin and improvements in oxidative homeostasis. Among the taxa enriched in the L. plantarum OL3246 group, Fecalibacterium prausnitzii is particularly noteworthy. This species is widely recognized for its anti-inflammatory properties121 and its capacity to synthesize butyrate in situ122, a short-chain fatty acid known to modulate inflammatory responses, maintain gut barrier integrity, and influence host metabolism123,124,125. Reduced levels of F. prausnitzii have been negatively associated with several age-related diseases, including cancer126, diabetes127, and cardiovascular diseases128. Its higher abundance in the L. plantarum OL3246 group, therefore, suggests increased resilience and a more stable microbial ecosystem. Importantly, Fecalibacterium species is also a strong ecological competitor that can suppress colonization by members of the Enterobacteriaceae family (E. coli, Klebsiella), which tend to increase with age14,129. In line with this, age-associated declines in F. prausnitzii abundance have been documented across the lifespan130. Only two prior studies involving L. plantarum reported a comparable positive influence on F. prausnitzii: first conducted by Ma et al.131 in stressed patients and another in malnourished participants receiving L. plantarum Dad-13132. Additionally, our intervention promoted enrichment at the family level, with a higher relative abundance of Ruminococcaceae, a key butyrate-producing family alongside Lachnospiraceae that plays a central role in gut fermentation and SCFA production133. Notably, many of these taxa are auxotrophic for vitamins, especially thiamine and folate134. Recent evidence suggests that enhanced vitamin availability, whether through dietary supplementation or microbial production, can increase the abundance of SCFA-producing phyla in the gut microbiome135,136,137. In this regard, the enrichment of unclassified Clostridia in the L. plantarum OL3246 group is particularly relevant, as members of this class are important contributors to microbial vitamin biosynthesis. This observation is in line with the parallel increase in abundance of metabolic pathways related to vitamins and polyamine biosynthesis. Finally, growing evidence suggests that cross-feeding interactions between microbial taxa represent one of the most important ecological mechanisms for stabilizing gut communities compositionally138,139. Taken together, our findings suggest that L. plantarum OL3246 has the potential to reshape gut microbial structures not only through direct modulation of key taxa but also indirectly by enhancing the community’s metabolic capacity, particularly with respect to vitamin biosynthesis.
In conclusion, this pilot study provides a comprehensive assessment of the influence of L. plantarum OL3246 on the health outcomes in a female predominant older population (Fig. 7). The observed pre-post changes suggest not only a positive impact on quality of life and mood but also measurable benefits at both systemic and local levels of host physiology. Specifically, reductions in AOPP and stabilization of SOD levels indicate a restored balance between free radical production and antioxidant defenses. These systemic effects may be mediated, at least in part, by the local action of L. plantarum OL3246 within the gastrointestinal tract. Orally administered, L. plantarum OL3246 was associated with reduced fecal calprotectin levels and increased microbial diversity, including enrichment of SCFA-producing bacterial families. Such microbial shifts are likely to contribute to the observed improvements in oxidative homeostasis. Furthermore, the enrichment of taxa with vitamin and polyamine biosynthetic potential points toward an additional mechanism of action that warrants further investigation. Collectively, these findings highlight the multifaceted effects of L. plantarum OL3246 and support its potential role in promoting healthy aging. These preliminary results should be confirmed in larger, sex-balanced cohort studies and in trials directly comparing single-strain and multi-strain probiotic formulations.
Created in BioRender. Mytych, J. (2025) https://BioRender.com/yydja2p.
Data availability
The datasets generated and analyzed during the current study contain sensitive medical information. In accordance with patient confidentiality, GDPR regulations, and the requirements of the local ethics committee, these data cannot be made publicly available. Importantly, the data have been presented in the manuscript in the form of dot plots, ensuring transparency and allowing independent assessment of the findings without access to raw medical records. For ethically justified and scientifically sound requests, limited access to anonymized datasets may be considered upon reasonable request to the corresponding author (J.M.), under conditions that guarantee data protection and confidentiality.
References
Williams, G.A., Cylus, J. & Roubal. T. Sustainable Health Financing with an Ageing Population (European Observatory on Health Systems and Policies, 2019).
Aiyar, S. & Ebeke, C. H. The Impact of Workforce Aging on European Productivity (International Monetary Fund, 2017).
Kalfoss, M. H., Reidunsdatter, R. J., Klöckner, C. A. & Nilsen, M. Validation of the WHOQOL-Bref: psychometric properties and normative data for the Norwegian general population. Health Qual. Life Outcomes 19, 13 (2021).
Mosaad Ali, M., Kamel Mosbah, S. & Mahmoud Abo El-Fadl, N. Factors affecting quality of life and work productivity among patients with gout. Am. J. Nurs. Res. 7, 128–135 (2019).
Gonzalez, L. A. D. et al. Health-related quality of life, financial toxicity, productivity loss and catastrophic health expenditures after lung cancer diagnosis in Argentina. arXiv [econ.GN] https://doi.org/10.21203/rs.3.rs-2365239/v1 (2022).
Yoshino, O., Takahashi, N. & Suzukamo, Y. Menstrual symptoms, health-related quality of life, and work productivity in Japanese women with dysmenorrhea receiving different treatments: prospective observational study. Adv. Ther. 39, 2562–2577 (2022).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: an expanding universe. Cell 186, 243–278 (2023).
Kaeberlein, M. Longevity and aging. F1000Prime Rep. 5, 5 (2013).
Guo, J. et al. Aging and aging-related diseases: from molecular mechanisms to interventions and treatments. Signal Transduct. Target. Ther. 7, 391 (2022).
Petersen, C. & Round, J. L. Defining dysbiosis and its influence on host immunity and disease. Cell. Microbiol. 16, 1024–1033 (2014).
Conway, J. & A Duggal, N. Ageing of the gut microbiome: potential influences on immune senescence and inflammageing. Ageing Res. Rev. 68, 101323 (2021).
Golshany, H. et al. The gut microbiome across the lifespan: how diet modulates our microbial ecosystem from infancy to the elderly. Int. J. Food Sci. Nutr. 76, 95–121 (2025).
Tavella, T. et al. Elevated gut microbiome abundance of Christensenellaceae, Porphyromonadaceae and Rikenellaceae is associated with reduced visceral adipose tissue and healthier metabolic profile in Italian elderly. Gut Microbes 13, 1–19 (2021).
Boopathi, S. et al. Gut Enterobacteriaceae and uraemic toxins—perpetrators for ageing. Exp. Gerontol. 173, 112088 (2023).
Iwaniak, P., Owe-Larsson, M. & Urbańska, E. M. Microbiota, tryptophan and aryl hydrocarbon receptors as the target triad in Parkinson’s disease—a narrative review. Int. J. Mol. Sci. 25, 2915 (2024).
Marć, M. A., Jastrząb, R. & Mytych, J. Does the gut microbial metabolome really matter? The connection between GUT metabolome and neurological disorders. Nutrients 14, 3967 (2022).
Kadyan, S. et al. Resistant starches from dietary pulses improve neurocognitive health via gut–microbiome–brain axis in aged mice. Front. Nutr. 11, 1322201 (2024).
World Report on Ageing and Health https://www.who.int/publications/i/item/9789241565042 (2015).
Capuron, L. & Miller, A. H. Immune system to brain signaling: neuropsychopharmacological implications. Pharmacol. Ther. 130, 226–238 (2011).
Dantzer, R., O’Connor, J. C., Freund, G. G., Johnson, R. W. & Kelley, K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat. Rev. Neurosci. 9, 46–56 (2008).
Lee, J. W. et al. Higher patient activation is associated with lower odds of functional limitation in older adults with chronic diseases. Geriatr. Nurs. 61, 157–161 (2025).
Religioni, U., Barrios-Rodríguez, R., Requena, P., Borowska, M. & Ostrowski, J. Enhancing therapy adherence: impact on clinical outcomes, healthcare costs, and patient quality of life. Medicina (Kaunas) 61, 153 (2025).
Moskalev, A., Stambler, I. & Caruso, C. Innate and adaptive immunity in aging and longevity: the foundation of resilience. Aging Dis 11, 1363–1373 (2020).
Zhang, W., Xiao, D., Mao, Q. & Xia, H. Role of neuroinflammation in neurodegeneration development. Signal Transduct. Target. Ther. 8, 267 (2023).
Jastrząb, R. et al. The strain-dependent cytostatic activity of Lactococcus lactis on CRC cell lines is mediated through the release of arginine deiminase. Microb. Cell Fact. 23, 82 (2024).
Snigdha, S. et al. Probiotics: potential novel therapeutics for microbiota–gut–brain axis dysfunction across gender and lifespan. Pharmacol. Ther. 231, 107978 (2022).
Kumar, A. et al. Probiotics as modulators of gut–brain axis for cognitive development. Front. Pharmacol. 15, 1348297 (2024).
Ojha, S., Patil, N., Jain, M., Kole, C. & Kaushik, P. Probiotics for neurodegenerative diseases: a systematic review. Microorganisms 11, 1083 (2023).
Castelli, V. et al. The emerging role of probiotics in neurodegenerative diseases: new hope for Parkinson’s disease?. Neural Regen. Res. 16, 628–634 (2021).
Hutchinson, A. N. et al. The effect of probiotics on health outcomes in the elderly: a systematic review of randomized, placebo-controlled studies. Microorganisms 9, 1344 (2021).
Östlund-Lagerström, L. et al. Probiotic administration among free-living older adults: a double blinded, randomized, placebo-controlled clinical trial. Nutr. J. 15, 80 (2016).
Nyangale, E. P. et al. Bacillus coagulans GBI-30, 6086 modulates Faecalibacterium prausnitzii in older men and women. J. Nutr. 145, 1446–1452 (2015).
Kim, C.-S. et al. Probiotic supplementation improves cognitive function and mood with changes in gut microbiota in community-dwelling older adults: a randomized, double-blind, placebo-controlled, multicenter trial. J. Gerontol. A Biol. Sci. Med. Sci. 76, 32–40 (2021).
Tylka, J. & Piotrowicz, R. Quality of life questionnaire SF-36—Polish version. Kardiol. Pol. 67, 1166–1169 (2009).
Aalto, A.-M., Elovainio, M., Kivimäki, M., Uutela, A. & Pirkola, S. The Beck Depression Inventory and General Health Questionnaire as measures of depression in the general population: a validation study using the Composite International Diagnostic Interview as the gold standard. Psychiatry Res. 197, 163–171 (2012).
Suija, K. et al. Validation of the Whooley questions and the Beck Depression Inventory in older adults. Scand. J. Prim. Health Care 30, 259–264 (2012).
Segal, D. L., Coolidge, F. L., Cahill, B. S. & O’Riley, A. A. Psychometric properties of the Beck Depression Inventory II (BDI-II) among community-dwelling older adults. Behav. Modif. 32, 3–20 (2008).
Bolyen, E. et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 37, 852–857 (2019).
Callahan, B. J. et al. DADA2: high-resolution sample inference from Illumina amplicon data. Nat. Methods 13, 581–583 (2016).
Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41, D590–D596 (2013).
Lozupone, C. & Knight, R. UniFrac: a new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 71, 8228–8235 (2005).
Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 26, 32–46 (2001).
Lin, H. & Peddada, S. D. Analysis of compositions of microbiomes with bias correction. Nat. Commun. 11, 3514 (2020).
Douglas, G. M. et al. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 38, 685–688 (2020).
Fan, Z. et al. Lactobacillus casei CCFM1074 alleviates collagen-induced arthritis in rats via balancing Treg/Th17 and modulating the metabolites and gut microbiota. Front. Immunol. 12, 680073 (2021).
Zabolotneva, A. A. et al. The obesogenic gut microbiota as a crucial factor defining the depletion of predicted enzyme abundance for vitamin B12 synthesis in the mouse intestine. Biomedicines 12, 1280 (2024).
Mihailović, M. et al. Beneficial effects of probiotic Lactobacillus paraplantarum BGCG11 on pancreatic and duodenum function in diabetic rats. Int. J. Mol. Sci. 25, 7697 (2024).
Zhou, H., He, K., Chen, J. & Zhang, X. LinDA: linear models for differential abundance analysis of microbiome compositional data. Genome Biol 23, 95 (2022).
Wickham, H. Ggplot2 (Springer International Publishing, 2016).
Knight, R. et al. Best practices for analysing microbiomes. Nat. Rev. Microbiol. 16, 410–422 (2018).
Hussenoeder, F. S. et al. Depression and quality of life in old age: a closer look. Eur. J. Ageing 18, 75–83 (2021).
Jomova, K. et al. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging. Arch. Toxicol. 97, 2499–2574 (2023).
Rahman, M. A. et al. Oxidative stress in DNA damage and neurodegenerative diseases: unveiling the mechanisms and therapeutic opportunities. Cell Biochem. Biophys. https://doi.org/10.1007/s12013-025-01845-9 (2025).
Ahmad Fadzuli, N. I. et al. Faecal intestinal permeability and intestinal inflammatory markers in older adults with age-related disorders: a systematic review and meta-analysis. Ageing Res. Rev. 101, 102506 (2024).
Padhi, P. et al. Mechanistic insights into gut microbiome dysbiosis-mediated neuroimmune dysregulation and protein misfolding and clearance in the pathogenesis of chronic neurodegenerative disorders. Front. Neurosci. 16, 836605 (2022).
Zhou, H., Tang, L., Fenton, K. A. & Song, X. Exploring and evaluating microbiome resilience in the gut. FEMS Microbiol. Ecol. 101, fiaf046 (2025).
Kossowska, M. et al. The interplay between gut microbiota and cognitive functioning in the healthy aging population: a systematic review. Nutrients 16, 852 (2024).
Foster, J. A. & McVey Neufeld, K.-A. Gut–brain axis: how the microbiome influences anxiety and depression. Trends Neurosci. 36, 305–312 (2013).
Le Morvan de Sequeira, C., Kaeber, M., Cekin, S. E., Enck, P. & Mack, I. The effect of probiotics on quality of life, depression and anxiety in patients with irritable bowel syndrome: a systematic review and meta-analysis. J. Clin. Med. 10, 3497 (2021).
van der Waal, M. B. et al. Probiotics for improving quality of life in ulcerative colitis: exploring the patient perspective. PharmaNutrition 7, 100139 (2019).
Recharla, N., Choi, J., Puligundla, P., Park, S.-J. & Lee, H.-J. Impact of probiotics on cognition and constipation in the elderly: a meta-analysis. Heliyon 9, e18306 (2023).
Moludi, J. et al. The efficacy of probiotics supplementation on the quality of life of patients with gastrointestinal disease: a systematic review of clinical studies. Prev. Nutr. Food Sci. 29, 237–255 (2024).
Button, K. S. et al. Minimal clinically important difference on the Beck Depression Inventory-II according to the patient’s perspective. Psychol. Med. 45, 3269–3279 (2015).
Wang, C. et al. Effectiveness of psychological interventions among community-dwelling older adults with subthreshold depression: a systematic review and meta-analysis. J. Affect. Disord. 354, 368–375 (2024).
Brody, D. J. & Hughes, J. P. Depression prevalence in adolescents and adults: United States, August 2021–August 2023. NCHS Data Brief 1, (2025).
Melo, D. et al. Prevalence and determinants of depressive symptoms in older adults across Europe: evidence from SHARE Wave 9. J. Clin. Med. 14, 5340 (2025).
Zenebe, Y., Akele, B., W/Selassie, M. & Necho, M. Prevalence and determinants of depression among old age: a systematic review and meta-analysis. Ann. Gen. Psychiatry 20, 55 (2021).
Jalali, A. et al. Global prevalence of depression, anxiety, and stress in the elderly population: a systematic review and meta-analysis. BMC Geriatr. 24, 809 (2024).
Wang, B. et al. Global, regional, and national burden and attributable risk factors of depressive disorders among older adults, 1990–2021. Int. Psychogeriatr. 37, 100069 (2025).
Rahmannia, M. et al. Strain-specific effects of probiotics on depression and anxiety: a meta-analysis. Gut Pathog. 16, 46 (2024).
Saccarello, A. et al. Oral administration of S-adenosylmethionine (SAMe) and Lactobacillus plantarum HEAL9 improves the mild-to-moderate symptoms of depression: a randomized, double-blind, placebo-controlled study. Prim. Care Companion CNS Disord. 22, 19m02578 (2020).
Ho, Y.-T., Tsai, Y.-C., Kuo, T. B. J. & Yang, C. C. H. Effects of Lactobacillus plantarum PS128 on depressive symptoms and sleep quality in self-reported insomniacs: a randomized, double-blind, placebo-controlled pilot trial. Nutrients 13, 2820 (2021).
Godzien, J. et al. Probiotic Lactobacillus plantarum 299v supplementation in patients with major depression in a double-blind, randomized, placebo-controlled trial: a metabolomics study. J. Affect. Disord. 368, 180–190 (2025).
Rudzki, L. et al. Probiotic Lactobacillus plantarum 299v decreases kynurenine concentration and improves cognitive functions in patients with major depression: a double-blind, randomized, placebo controlled study. Psychoneuroendocrinology 100, 213–222 (2019).
Milajerdi, A. et al. The effect of probiotics on inflammatory biomarkers: a meta-analysis of randomized clinical trials. Eur. J. Nutr. 59, 633–649 (2020).
Cristofori, F. et al. Anti-inflammatory and immunomodulatory effects of probiotics in gut inflammation: a door to the body. Front. Immunol. 12, 578386 (2021).
Abdi, A., Oroojzadeh, P., Valivand, N., Sambrani, R. & Lotfi, H. Immunological aspects of probiotics for improving skin diseases: influence on the gut–brain–skin axis. Biochem. Biophys. Res. Commun. 702, 149632 (2024).
Zeng, L. et al. Safety and efficacy of probiotic supplementation in 8 types of inflammatory arthritis: a systematic review and meta-analysis of 34 randomized controlled trials. Front. Immunol. 13, 961325 (2022).
Pavlidou, E., Fasoulas, A., Mantzorou, M. & Giaginis, C. Clinical evidence on the potential beneficial effects of probiotics and prebiotics in cardiovascular disease. Int. J. Mol. Sci. 23, 15898 (2022).
Chatsirisakul, O. et al. Strain-specific therapeutic potential of Lactiplantibacillus plantarum: a systematic scoping review. Nutrients 17, 1165 (2025).
Han, K. J., Lee, J.-E., Lee, N.-K. & Paik, H.-D. Antioxidant and anti-inflammatory effect of probiotic Lactobacillus plantarum KU15149 derived from Korean homemade diced-radish Kimchi. J. Microbiol. Biotechnol. 30, 591–598 (2020).
Luan, X., Feng, M. & Sun, J. Effect of Lactobacillus plantarum on antioxidant activity in fermented sausage. Food Res. Int. 144, 110351 (2021).
DÜz, M., DoĞan, Y. N. & DoĞan İ. Antioxidant activitiy of Lactobacillus plantarum, Lactobacillus sake and Lactobacillus curvatus strains isolated from fermented Turkish Sucuk. An. Acad. Bras. Cienc. 92, e20200105 (2020).
Wang, J. et al. Lactobacillus plantarum exhibits antioxidant and cytoprotective activities in porcine intestinal epithelial cells exposed to hydrogen peroxide. Oxidative Med. Cell. Longev. 2021, 8936907 (2021).
Ge, Q. et al. Antioxidant activity of Lactobacillus plantarum NJAU-01 in an animal model of aging. BMC Microbiol. 21, 182 (2021).
Lazou-Ahrén, I. et al. Probiotic-reduced inflammaging in older adults: a randomized, double-blind, placebo-controlled trial. Probiotics Antimicrob. Proteins https://doi.org/10.1007/s12602-024-10310-7 (2024).
Dzięgielewska-Gęsiak, S., Wysocka, E., Fatyga, E. & Muc-Wierzgoń, M. Relationship of SOD-1 activity in metabolic syndrome and/or frailty in elderly individuals. Metabolites 14, 514 (2024).
Silva, L. A. D., Malfatti, C. R. M., Soares, K. C. N., Silva, M. C. D. & Brasil, M. R. Unraveling the interplay of oxidative stress, aging, and skeletal muscle: insights and interventions for optimal muscle function in the elderly. J. Adv. Med. Med. Res. 35, 21–27 (2023).
Zelko, I. N., Mariani, T. J. & Folz, R. J. Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic. Biol. Med. 33, 337–349 (2002).
Landis, G. N. & Tower, J. Superoxide dismutase evolution and life span regulation. Mech. Ageing Dev. 126, 365–379 (2005).
Conti, G. et al. Association of higher advanced oxidation protein products (AOPPs) levels in patients with diabetic and hypertensive nephropathy. Medicina (Kaunas) 55, 675 (2019).
Villalpando-Sánchez, D. C. et al. Advanced oxidative protein products had a diagnostic accuracy for identifying chronic Kidney Disease in adult population. Metabolites 14, 37 (2024).
Koike, A. et al. Thiol groups as a biomarker for the diagnosis and prognosis of prostate cancer. Sci. Rep. 10, 9093 (2020).
Napiórkowska-Mastalerz, M. et al. A preliminary evaluation of advanced oxidation protein products (AOPPs) as a potential approach to evaluating prognosis in early-stage breast cancer patients and its implication in tumour angiogenesis: a 7-year single-centre study. Cancers (Basel) 16, 1068 (2024).
Skvarilová, M., Bulava, A., Stejskal, D., Adamovská, S. & Bartek, J. Increased level of advanced oxidation products (AOPP) as a marker of oxidative stress in patients with acute coronary syndrome. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 149, 83–87 (2005).
Gonzalez, E. et al. An increase of plasma advanced oxidation protein products levels is associated with cardiovascular risk in incident peritoneal dialysis patients: a pilot study. Oxidative Med. Cell. Longev. 2015, 1–6 (2015).
Lou, A. et al. Advanced oxidation protein products induce inflammatory responses and invasive behaviour in fibroblast-like synoviocytes via the RAGE–NF-κB pathway. Bone Jt. Res. 10, 259–268 (2021).
Maciejczyk, M., Nesterowicz, M., Szulimowska, J. & Zalewska, A. Oxidation, glycation, and carbamylation of salivary biomolecules in healthy children, adults, and the elderly: can saliva be used in the assessment of aging? J. Inflamm. Res. 15, 2051–2073 (2022).
Baraibar, M. A., Liu, L., Ahmed, E. K. & Friguet, B. Protein oxidative damage at the crossroads of cellular senescence, aging, and age-related diseases. Oxid. Med. Cell. Longev. 2012, 919832 (2012).
Demirbilek, M. E., Kilic, N., Komurcu, H. F. & Akin, K. O. Advanced oxidation protein products in aged with dementia. Am. J. Immunol. 3, 52–55 (2007).
Saez, I. Cologne Excellence Cluster for Cellular Stress Responses in Aging-Associated Diseases (CECAD), University of Cologne, Joseph Stelzmann Strasse 26, 50931 Cologne, Germany & Vilchez, D. Protein clearance mechanisms and their demise in age-related neurodegenerative diseases. AIMS Mol. Sci. 2, 1–21 (2015).
Lin, K.-J. et al. The overcrowded crossroads: mitochondria, alpha-synuclein, and the endo-lysosomal system interaction in Parkinson’s disease. Int. J. Mol. Sci. 20, 5312 (2019).
Bougea, A. et al. Plasma alpha-synuclein levels in patients with Parkinson’s disease: a systematic review and meta-analysis. Neurol. Sci. 40, 929–938 (2019).
Noori, M. et al. The effect of probiotic-fortified kefir on depression, appetite, oxidative stress, and inflammatory parameters in Iranian overweight and obese elderly: a randomized, double-blind, placebo-controlled clinical trial. J. Health Popul. Nutr. 44, 30 (2025).
Hsu, Y.-C. et al. Efficacy of probiotic supplements on brain-derived neurotrophic factor, inflammatory biomarkers, oxidative stress and cognitive function in patients with Alzheimer’s dementia: a 12-week randomized, double-blind active-controlled study. Nutrients 16, 16 (2023).
Ma, T. et al. Targeting gut microbiota and metabolism as the major probiotic mechanism—an evidence-based review. Trends Food Sci. Technol. 138, 178–198 (2023).
Nourizadeh, R. et al. (Eds.). Impact of probiotics in modulation of gut microbiome. In Microbiome–Gut–Brain Axis 401–409 (Springer Nature, 2022).
Brookes, M. J., Whitehead, S., Gaya, D. R. & Hawthorne, A. B. Practical guidance on the use of faecal calprotectin. Frontline Gastroenterol. 9, 87–91 (2018).
Joshi, S., Lewis, S. J., Creanor, S. & Ayling, R. M. Age-related faecal calprotectin, lactoferrin and tumour M2-PK concentrations in healthy volunteers. Ann. Clin. Biochem. 47, 259–263 (2010).
Mulak, A., Koszewicz, M., Panek-Jeziorna, M., Koziorowska-Gawron, E. & Budrewicz, S. Fecal calprotectin as a marker of the gut immune system activation is elevated in Parkinson’s disease. Front. Neurosci. 13, 992 (2019).
Dumitrescu, L. et al. Serum and fecal markers of intestinal inflammation and intestinal barrier permeability are elevated in Parkinson’s disease. Front. Neurosci. 15, 689723 (2021).
Gallo, A. et al. Diagnostic accuracy of fecal calprotectin in discriminating organic-inflammatory gastrointestinal diseases and functional gastrointestinal disorders in older patients. J. Pers. Med. 14, 227 (2024).
Heinzel, S. et al. Elevated fecal calprotectin is associated with gut microbial dysbiosis, altered serum markers and clinical outcomes in older individuals. Sci. Rep. 14, 13513 (2024).
Seethaler, B. et al. Biomarkers for assessment of intestinal permeability in clinical practice. Am. J. Physiol. Gastrointest. Liver Physiol. 321, G11–G17 (2021).
Aquilani, R. et al. Effects of a metabolic mixture on gut inflammation and permeability in elderly patients with Chronic Kidney Disease: a proof-of-concept study. Metabolites 12, 987 (2022).
Malíčková, K. et al. Fecal zonulin is elevated in Crohn’s disease and in cigarette smokers. Pract. Lab. Med. 9, 39–44 (2017).
Qi, Y. et al. Intestinal permeability biomarker zonulin is elevated in healthy aging. J. Am. Med. Dir. Assoc. 18, 810.e1–810.e4 (2017).
Mörkl, S. et al. Gut microbiota, dietary intakes and intestinal permeability reflected by serum zonulin in women. Eur. J. Nutr. 57, 2985–2997 (2018).
Heidt, C. et al. Assessment of intestinal permeability and inflammation bio-markers in patients with rheumatoid arthritis. Nutrients 15, 2386 (2023).
Moreno-Navarrete, J. M., Sabater, M., Ortega, F., Ricart, W. & Fernández-Real, J. M. Circulating zonulin, a marker of intestinal permeability, is increased in association with obesity-associated insulin resistance. PLoS ONE 7, e37160 (2012).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 105, 16731–16736 (2008).
Machiels, K. et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut 63, 1275–1283 (2014).
Salvi, P. S. & Cowles, R. A. Butyrate and the intestinal epithelium: modulation of proliferation and inflammation in homeostasis and disease. Cells 10, 1775 (2021).
Canani, R. B. et al. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 17, 1519–1528 (2011).
Hodgkinson, K. et al. Butyrate’s role in human health and the current progress towards its clinical application to treat gastrointestinal disease. Clin. Nutr. 42, 61–75 (2023).
Formiga, R. & Sokol, H. Faecalibacterium prausnitzii: one species with multiple potential implications in cancer research. Gut 74, 1038–1039 (2025).
Navab-Moghadam, F. et al. The association of type II diabetes with gut microbiota composition. Microb. Pathog. 110, 630–636 (2017).
Zhang, Y. et al. Faecalibacterium prausnitzii prevents age-related heart failure by suppressing ferroptosis in cardiomyocytes through butyrate-mediated LCN2 regulation. Gut Microbes 17, 2505119 (2025).
Yin, Q. et al. Ecological dynamics of Enterobacteriaceae in the human gut microbiome across global populations. Nat. Microbiol. 10, 541–553 (2025).
Biagi, E. et al. Through ageing, and beyond: gut microbiota and inflammatory status in seniors and centenarians. PLoS ONE 5, e10667 (2010).
Ma, T. et al. Probiotic consumption relieved human stress and anxiety symptoms possibly via modulating the neuroactive potential of the gut microbiota. Neurobiol. Stress 14, 100294 (2021).
Kamil, R. Z., Murdiati, A., Juffrie, M. & Rahayu, E. S. Gut microbiota modulation of moderate undernutrition in infants through gummy Lactobacillus plantarum Dad-13 consumption: a randomized double-blind controlled trial. Nutrients 14, 1049 (2022).
Baxter, N. T. et al. Dynamics of human gut microbiota and short-chain fatty acids in response to dietary interventions with three fermentable fibers. MBio 10, e02566–18 (2019).
Soto-Martin, E. C. et al. Vitamin biosynthesis by human gut butyrate-producing bacteria and cross-feeding in synthetic microbial communities. MBio 11, e00886-20 (2020).
Park, J. et al. Dietary vitamin B1 intake influences gut microbial community and the consequent production of short-chain fatty acids. Nutrients 14, 2078 (2022).
Pham, V. T., Dold, S., Rehman, A., Bird, J. K. & Steinert, R. E. Vitamins, the gut microbiome and gastrointestinal health in humans. Nutr. Res. 95, 35–53 (2021).
Tarracchini, C. et al. Exploring the vitamin biosynthesis landscape of the human gut microbiota. mSystems 9, e0092924 (2024).
Culp, E. J. & Goodman, A. L. Cross-feeding in the gut microbiome: ecology and mechanisms. Cell Host Microbe 31, 485–499 (2023).
Marcelino, V. R. et al. Disease-specific loss of microbial cross-feeding interactions in the human gut. Nat. Commun. 14, 6546 (2023).
Acknowledgements
This research was funded by the National Centre for Research and Development (NCBiR, Poland), under the grant number POIR.01.01.01-00-0985/17.
Author information
Authors and Affiliations
Contributions
R.J.: writing—review and editing, writing—original draft, software, data curation, visualization, formal analysis, conceptualization, and funding acquisition; A.M.: methodology, investigation, and data curation; E.K.M.: methodology, investigation, and data curation; A.G.: methodology, investigation, and data curation; B.K.: resources, methodology, and investigation; K.K.: methodology and investigation; J.W.-K.: methodology and investigation; D.W.: methodology, investigation, and data curation; K.M. methodology, investigation, and data curation; M.N.-Ch.: methodology, investigation, and data curation; D.L.: methodology, investigation, and data curation; K.G.: methodology, investigation, and data curation; M.G.: methodology, investigation, and data curation; N.P.: methodology, investigation, and data curation; G.P.: methodology, investigation, and data curation; G.K.: methodology, investigation, and data curation; J.M.: supervision, writing—review and editing, resources, project administration, and conceptualization. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
This study was sponsored by Olimp Laboratories Sp. z o.o., which received a public research grant from the National Centre for Research and Development (NCBiR, Poland). The funding agency had no role in the study design, data analysis, interpretation, or the decision to submit the article for publication. The study was conceptually designed by investigators from the Academy of Physical Education, Katowice, Poland, and Medical University of Silesia, Katowice, Poland, in cooperation with the R&D department of Olimp Laboratories Sp. z o.o., and subsequently approved by the sponsor. The investigational product was developed and manufactured in-house at Olimp Laboratories Sp. z o.o. by company employees who are also co-authors of this paper. The sponsor had no role in data analysis, interpretation of results, manuscript writing, or the decision to submit for publication. These details are now explicitly stated in the revised manuscript to ensure full transparency. All aspects of patient recruitment, data collection, data management, and preliminary statistical analysis were performed independently by the Academy of Physical Education, Katowice, Poland and Medical University of Silesia, Katowice, Poland research teams. The final statistical analyses were conducted by the first author and independently cross-checked by the Academy of Physical Education, Katowice, Poland and Medical University of Silesia, Katowice, Poland co-authors to ensure accuracy. JM, RJ, BK, KK, JW-K, DW, and KM are employees of Olimp Laboratories Sp. z o.o. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jastrząb, R., Małecki, A., Kmiecik-Małecka, E. et al. Probiotic Lactiplantibacillus plantarum OL3246 supports healthy aging by enhancing quality of life, reducing inflammation, and modulating gut microbiota: a pilot study. npj Aging 12, 37 (2026). https://doi.org/10.1038/s41514-026-00338-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41514-026-00338-0