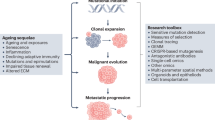
Abstract
Given global population aging and the absence of aging-reversal therapies, elucidating the aging-related cancer risk association and developing cancer prevention strategies are imperative. This population-based cohort study analyzed data from the UK Biobank. Aging was assessed through four validated markers, including Klemera-Doubal method (KDM), PhenoAge, leukocyte telomere length (TL) and chronological age. Over a median follow-up of 13.5 years, significant associations between all aging measures and elevated overall cancer risk were observed. False discovery rate (FDR)-corrected analyses revealed a significant association in a biological aging group for seven site-specific cancers, including esophageal, colorectal, pancreatic, skin, kidney, urinary tract cancers, and lymphoma, as evaluated by the four aging markers. A significant interaction (FDR-corrected p < 0.05) between Life's Essential 8 (LE8) and PhenoAge was observed for lung cancer risk. Furthermore, joint analyses revealed that elevated LE8 scores modify this risk for overall cancer among individuals with biological aging, with consistent risk reductions observed for esophageal, gastric, breast, and uterine cancers across all aging markers. These findings suggest that a higher LE8 score is associated with lower cancer risk in the context of biological aging.
Similar content being viewed by others
Data availability
Due to the privacy and sensitive information contained in the data, the data used for this study will not be made publicly available. The raw and derived data from UK Biobank are available to approved investigators (https://www.ukbiobank.ac.uk/). All relevant data will be shared on reasonable request to the corresponding authors.
Code availability
Codes used for this study are available upon reasonable request from the authors.
References
Partridge, L., Deelen, J. & Slagboom, P. E. Facing up to the global challenges of ageing. Nature 561, 45–56 (2018).
Dogra, S. et al. Active aging and public health: evidence, implications, and opportunities. Annu. Rev. Public Health 43, 439–459 (2022).
Guo, J. et al. Aging and aging-related diseases: from molecular mechanisms to interventions and treatments. Signal Transduct. Target. Ther. 7, 391 (2022).
Klemera, P. & Doubal, S. A new approach to the concept and computation of biological age. Mech. Ageing Dev. 127, 240–248 (2006).
Levine, M. E. et al. An epigenetic biomarker of aging for lifespan and healthspan. Aging 10, 573–591 (2018).
Rossiello, F., Jurk, D., Passos, J. F. & d’Adda di Fagagna, F. Telomere dysfunction in ageing and age-related diseases. Nat. Cell Biol. 24, 135–147 (2022).
Morales Berstein, F. et al. Assessing the causal role of epigenetic clocks in the development of multiple cancers: a Mendelian randomization study. eLife 11, e75374 (2022).
Yang, Z., Shen, Y., Zhang, T., Tang, X. & Mao, R. Associations of biological age accelerations and genetic risk with incident endometrial cancer: a prospective analysis in UK Biobank. Int. J. Surg. Lond. Engl. 111, 512–519 (2024).
Bian, L. et al. Associations of combined phenotypic aging and genetic risk with incident cancer: a prospective cohort study. eLife 13, RP91101 (2024).
Mak, J. K. L. et al. Clinical biomarker-based biological aging and risk of cancer in the UK Biobank. Br. J. Cancer 129, 94–103 (2023).
López-Otín, C., Pietrocola, F., Roiz-Valle, D., Galluzzi, L. & Kroemer, G. Meta-hallmarks of aging and cancer. Cell Metab. 35, 12–35 (2023).
Park, M. D. et al. Hematopoietic aging promotes cancer by fueling IL-1α-driven emergency myelopoiesis. Science 386, eadn0327 (2024).
Li, X. et al. Accelerated aging mediates the associations of unhealthy lifestyles with cardiovascular disease, cancer, and mortality. J. Am. Geriatr. Soc. 72, 181–193 (2024).
Schmitt, C. A., Wang, B. & Demaria, M. Senescence and cancer - role and therapeutic opportunities. Nat. Rev. Clin. Oncol. 19, 619–636 (2022).
Schneider, C. V. et al. Association of telomere length with risk of disease and mortality. JAMA Intern. Med. 182, 291 (2022).
Lloyd-Jones, D. M. et al. Life’s essential 8: updating and enhancing the American Heart Association’s construct of cardiovascular health: a presidential advisory from the American Heart Association. Circulation 146, e18–e43 (2022).
Zhang, J. et al. Associations of Life’s Essential 8 and fine particulate matter pollution with the incidence of atrial fibrillation. J. Hazard. Mater. 459, 132114 (2023).
Wu, Z. et al. Life’s essential 8, genetic susceptibility, and risk of incident pancreatic cancer: a prospective cohort study. https://doi.org/10.1002/ijc.35184.
Zhao, Y., Song, Y., Li, X. & Guo, A. Association of life’s essential 8 cardiovascular health with breast cancer incidence and mortality according to genetic susceptibility of breast cancer: a prospective cohort study. Breast Cancer Res. 26, 121 (2024).
Cardiovascular health and cancer mortality: evidence from US NHANES and UK Biobank cohort studies. BMC Med. https://doi.org/10.1186/s12916-024-03553-2 (2024).
Hou, Y. et al. Association between cardiovascular health, cancer and its prognosis: a prospective cohort study. Public Health 242, 1–6 (2025).
Yang, C. et al. Life’s essential 8 and specific cancer risk and mortality in men and women: a population-based cohort analysis of 332,417 United Kingdom participants. BMC Cancer 25, 632 (2025).
Karlstaedt, A., Moslehi, J. & de Boer, R. A. Cardio-onco-metabolism: metabolic remodelling in cardiovascular disease and cancer. Nat. Rev. Cardiol. 19, 414–425 (2022).
Hibler, E. A. & Lloyd-Jones, D. M. Addressing the ‘common soil’ of risk factors for cardiovascular disease and cancer. JACC CardioOncology 3, 59–61 (2021).
Li, X. et al. Longitudinal trajectories, correlations and mortality associations of nine biological ages across 20-years follow-up. eLife 9, e51507 (2020).
Belsky, D. W. et al. Eleven telomere, epigenetic clock, and biomarker-composite quantifications of biological aging: do they measure the same thing? Am. J. Epidemiol. 187, 1220–1230 (2018).
Wang, X. et al. Joint association of biological aging and lifestyle with risks of cancer incidence and mortality: A cohort study in the UK Biobank. Prev. Med. 182, 107928 (2024).
Peng, Y. Cardiovascular health, polygenic risk score, and cancer risk: a prospective cohort study. Am. J. Clin. Nutr. 120, 785–793 (2024).
Zhang, J. et al. Relation of Life’s essential 8 to the genetic predisposition for cardiovascular outcomes and all-cause mortality: results from a national prospective cohort. Eur. J. Prev. Cardiol. 30, 1676–1685 (2023).
Wu, D. et al. Better Life’s Essential 8 contributes to slowing the biological aging process: a cross-sectional study based on NHANES 2007-2010 data. Front. Public Health 12, 1295477 (2024).
Chen, H. et al. Adherence to Life’s essential 8 is associated with delayed biological aging: a population-based cross-sectional study. Rev. Esp. Cardiol. Engl. Ed. 78, 37–46 (2025).
Zhang, R. et al. Association between life’s essential 8 and biological ageing among US adults. J. Transl. Med. 21, 622 (2023).
Van Sloten, T. et al. Association of midlife cardiovascular health and subsequent change in cardiovascular health with incident cancer. JACC CardioOncol. 5, 39–52 (2023).
Liu, Y. et al. Dysregulation of immunity by cigarette smoking promotes inflammation and cancer: a review. Environ. Pollut. Barking Essex 1987 339, 122730 (2023).
Garg, S. K., Maurer, H., Reed, K. & Selagamsetty, R. Diabetes and cancer: two diseases with obesity as a common risk factor. Diab. Obes. Metab. 16, 97–110 (2014).
Lane, J. M. et al. Genetics of circadian rhythms and sleep in human health and disease. Nat. Rev. Genet. 24, 4–20 (2023).
Chaput, J.-P. et al. The role of insufficient sleep and circadian misalignment in obesity. Nat. Rev. Endocrinol. 19, 82–97 (2023).
Pathak, S. K. et al. Oxidative stress and cyclooxygenase activity in prostate carcinogenesis: targets for chemopreventive strategies. Eur. J. Cancer Oxf. Engl. 41, 61–70 (2005).
Liu, W. et al. Dysregulated cholesterol homeostasis results in resistance to ferroptosis increasing tumorigenicity and metastasis in cancer. Nat. Commun. 12, 5103 (2021).
Kidoguchi, S. et al. New concept of onco-hypertension and future perspectives. Hypertens. Dallas Tex. 1979 77, 16–27 (2021).
Stensrud, M. J. & Hernán, M. A. Why test for proportional hazards? JAMA 323, 1401–1402 (2020).
Levine, M. E. Modeling the rate of senescence: can estimated biological age predict mortality more accurately than chronological age? J. Gerontol. A Biol. Sci. Med. Sci. 68, 667–674 (2013).
Liu, Z. et al. A new aging measure captures morbidity and mortality risk across diverse subpopulations from NHANES IV: a cohort study. PLoS Med. 15, e1002718 (2018).
Kwon, D. & Belsky, D. W. A toolkit for quantification of biological age from blood chemistry and organ function test data: BioAge. GeroScience 43, 2795–2808 (2021).
Codd, V. et al. Measurement and initial characterization of leukocyte telomere length in 474,074 participants in UK Biobank. Nat. Aging 2, 170–179 (2022).
Ye, Q. et al. Telomere length and chronological age across the human lifespan: a systematic review and meta-analysis of 414 study samples including 743,019 individuals. Ageing Res. Rev. 90, 102031 (2023).
Yang, H. et al. Life’s essential 8, genetic susceptibility, and risk of inflammatory bowel diseases: a population-based cohort study. Int. J. Behav. Nutr. Phys. Act. 21, 66 (2024).
Blane, D., Townsend, P., Phillimore, P. & Beattie, A. Health and deprivation: inequality and the north. Br. J. Sociol. 40, 344 (1987).
Acknowledgements
We are grateful to the participants and study staff of UK Biobank. This research received no external funding.
Author information
Authors and Affiliations
Contributions
Y.L., Z.X., X.G., and H.F. contributed to the conception and design of this study. Z.X., W.Z., X.G., Y.Z., S.Y., Z.J., Y.H., and J.Z. were involved in the acquisition of data. Z.X., H.F., and J.L. performed analysis. Z.X., H.F., and J.L. were involved in data interpretation. Y.Z.., H.F., W.Z., and J.L. wrote the first draft of the manuscript. All the authors discussed the results, revised the manuscript, and agreed to its publication. H.F. and Y.L. supervised the study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, J., Zhang, Y., Zhang, W. et al. Aging and increased cancer risk: exploring the potential of LE8 score to mitigate risk. npj Aging (2026). https://doi.org/10.1038/s41514-026-00352-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41514-026-00352-2