Abstract
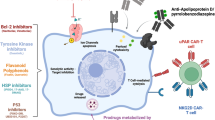
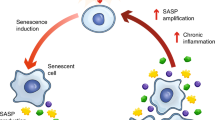
Cellular senescence, originally described as a finite proliferative arrest in cultured somatic cells, has since been recognized as a central mechanism underlying aging and the development of age-associated disorders. The progressive accumulation of senescent cells (SnCs) promotes chronic inflammation through the senescence-associated secretory phenotype (SASP) and circumvents immune-mediated clearance by upregulating pro-survival and immune checkpoint pathways. Early “first-generation” senolytics, including navitoclax (ABT-263) and the dasatinib–quercetin (D + Q) combination, provided proof-of-concept that selective removal of SnCs can alleviate certain fibrotic, metabolic, and cardiovascular pathologies in preclinical studies. However, these agents exhibited notable drawbacks, such as dose-dependent thrombocytopenia, variable therapeutic efficacy, and the emergence of resistance mechanisms. Consequently, current research has shifted toward precision senotherapy, though significant translational challenges remain. This review synthesizes three next-generation strategies developed to address limitations of early senolytic agents. (1) Immune-based senolysis: This approach applies immuno-oncology principles to counter immune evasion of SnCs. Strategies include blocking immunosuppressive ligands such as GD3 ganglioside, engineering chimeric antigen receptor (CAR) T cells to target senescence-specific surface markers like urokinase-type plasminogen activator receptor (uPAR), and exploiting metabolic vulnerabilities (e.g., glutaminolysis and ferroptosis) to sensitize SnCs to immune-mediated clearance. (2) Tissue-precision proteolysis-targeting chimeras (PROTACs): These agents recruit organ- or tissue-specific E3 ligases (e.g., von Hippel-Lindau (VHL)) to selectively degrade anti-apoptotic proteins such as BCL-xL. Localized activity may reduce systemic toxicity and mitigate dose-limiting effects observed with traditional inhibitors. (3) Microbiome–epigenetic interplay: This strategy modulates the gut–liver axis to enhance senolytic efficacy. Short-chain fatty acids (SCFAs), such as butyrate, epigenetically regulate drug transporter expression and suppress the SASP, while dietary interventions may create a microenvironment favorable to senolysis. These approaches offer potentially more targeted and personalized therapeutic options but face significant challenges, including immunopathology, manufacturing complexity, off-target effects, and long-term safety concerns. The ongoing shift from broad inhibition to precision reprogramming represents a promising but preliminary step in the treatment of age-related diseases.
Similar content being viewed by others
Data availability
No datasets were generated or analyzed during the current study.
References
Hayflick, L. & Moorhead, P. S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 25, 585–621 (1961).
Gorgoulis, V. et al. Cellular senescence: defining a path forward. Cell 179, 813–827 (2019).
Miwa, S., Kashyap, S., Chini, E. & von Zglinicki, T. Mitochondrial dysfunction in cell senescence and aging. J. Clin. Investig. 132, e158447 (2022).
Frey, Y., Haj, M., Ziv, Y., Elkon, R. & Shiloh, Y. Broad repression of DNA repair genes in senescent cells identified by integration of transcriptomic data. Nucleic Acids Res. 53, gkae1257 (2025).
Correia-Melo, C. et al. Mitochondria are required for pro-ageing features of the senescent phenotype. EMBO J. 35, 724–742 (2016).
Huang, W., Hickson, L. J., Eirin, A., Kirkland, J. L. & Lerman, L. O. Cellular senescence: the good, the bad and the unknown. Nat. Rev. Nephrol. 18, 611–627, https://doi.org/10.1038/s41581-022-00601-z (2022).
Yosef, R. et al. Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL. Nat. Commun. 7, 11190 (2016).
Zhu, Y. et al. The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging cell 14, 644–658 (2015).
Iltis, C. et al. A ganglioside-based immune checkpoint enables senescent cells to evade immunosurveillance during aging. Nat. Aging 5, 219–236 (2025).
Wang, B. et al. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat. Rev. Mol. Cell Biol. 25, 958–978 (2024).
Basisty, N. et al. A proteomic atlas of senescence-associated secretomes for aging biomarker development. PLoS Biol. 18, e3000599 (2020).
Ogrodnik, M. et al. Guidelines for minimal information on cellular senescence experimentation in vivo. Cell 187, 4150–4175 (2024).
Di Micco, R., Krizhanovsky, V., Baker, D. & d’Adda di Fagagna, F. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 22, 75–95 (2021).
Schafer, M. J. et al. Cellular senescence mediates fibrotic pulmonary disease. Nat. Commun. 8, 14532 (2017).
Jeon, O. H. et al. Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nat. Med. 23, 775–781 (2017).
Bussian, T. J. et al. Clearance of senescent glial cells prevents tau-dependent pathology and cognitive decline. Nature 562, 578–582 (2018).
Kuźniar, J. et al. Connections between cellular senescence and alzheimer’s disease—a narrative review. Int. J. Mol. Sci. 26, 8638 (2025).
Xu, C. et al. The role of cellular senescence in cardiovascular disease. Cell Death Discov. 11, 431 (2025).
Childs, B. G. et al. Senescent intimal foam cells are deleterious at all stages of atherosclerosis. Science 354, 472–477 (2016).
Hickson, L. J. et al. Senolytics decrease senescent cells in humans: preliminary report from a clinical trial of dasatinib plus quercetin in individuals with diabetic kidney disease. EBioMedicine 47, 446–456 (2019).
McHugh, D., Durán, I. & Gil, J. Senescence as a therapeutic target in cancer and age-related diseases. Nat. Rev. Drug Discov. 24, 57–71 (2025).
Hu, L. et al. Why senescent cells are resistant to apoptosis: an insight for senolytic development. Front. Cell Dev. Biol. 10, 822816 (2022).
Zhang, L. et al. Cellular senescence: a key therapeutic target in aging and diseases. J. Clin. Investig. 132, e158450 (2022).
Ge, M. et al. Senolytic targets and new strategies for clearing senescent cells. Mech. Ageing Dev. 195, 111468 (2021).
Calabrò, A. et al. Senotherapeutics to counteract senescent cells are prominent topics in the context of anti-ageing strategies. Int. J. Mol. Sci. 25, 1792 (2024).
Wang, Y. et al. Implication of cellular senescence in the progression of chronic kidney disease and the treatment potencies. Biomed. Pharmacother. 135, 111191 (2021).
Chaib, S., Tchkonia, T. & Kirkland, J. L. Cellular senescence and senolytics: the path to the clinic. Nat. Med. 28, 1556–1568 (2022).
Ota, H. & Kodama, A. Dasatinib plus quercetin attenuates some frailty characteristics in SAMP10 mice. Sci. Rep. 12, 2425 (2022).
Chang, J. et al. Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nat. Med. 22, 78–83 (2016).
Zhu, Y. et al. Identification of a novel senolytic agent, navitoclax, targeting the Bcl-2 family of anti-apoptotic factors. Aging Cell 15, 428–435 (2016).
Mylonas, K. J. et al. Cellular senescence inhibits renal regeneration after injury in mice, with senolytic treatment promoting repair. Sci. Transl. Med. 13, eabb0203 (2021).
González-Gualda, E. et al. Galacto-conjugation of navitoclax as an efficient strategy to increase senolytic specificity and reduce platelet toxicity. Aging Cell 19, e13142 (2020).
Wilson, W. H. et al. Navitoclax, a targeted high-affinity inhibitor of BCL-2, in lymphoid malignancies: a phase 1 dose-escalation study of safety, pharmacokinetics, pharmacodynamics, and antitumour activity. Lancet Oncol. 11, 1149–1159 (2010).
Klener, P. et al. BH3 mimetics in hematologic malignancies. Int. J. Mol. Sci. 22, 10157 (2021).
Xu, M. et al. Senolytics improve physical function and increase lifespan in old age. Nat. Med. 24, 1246–1256 (2018).
Justice, J. N. et al. Senolytics in idiopathic pulmonary fibrosis: results from a first-in-human, open-label, pilot study. EBioMedicine 40, 554–563 (2019).
Farr, J. N. et al. Effects of intermittent senolytic therapy on bone metabolism in postmenopausal women: a phase 2 randomized controlled trial. Nat. Med. 30, 2605–2612 (2024).
Lelarge, V. et al. Senolytics: from pharmacological inhibitors to immunotherapies, a promising future for patients’ treatment. NPJ Aging 10, 12 (2024).
Battaglia-Vieni, A. et al. Dasatinib and quercetin combination increased kidney damage in acute folic acid-induced experimental nephropathy. Pharmaceuticals 18, 822–822 (2025).
Yousefzadeh, M. J. et al. Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine 36, 18–28 (2018).
Zhu, Y. et al. New agents that target senescent cells: the flavone, fisetin, and the BCL-XL inhibitors, A1331852 and A1155463. Aging 9, 955–963 (2017).
Harrison, D. E. et al. Astaxanthin and meclizine extend lifespan in UM-HET3 male mice; fisetin, SG1002 (hydrogen sulfide donor), dimethyl fumarate, mycophenolic acid, and 4-phenylbutyrate do not significantly affect lifespan in either sex at the doses and schedules used. Geroscience 46, 795–816 (2024).
Krishnakumar, I. M. et al. Enhanced bioavailability and pharmacokinetics of a novel hybrid-hydrogel formulation of fisetin orally administered in healthy individuals: a randomised double-blinded comparative crossover study. J. Nutr. Sci. 11, e74 (2022).
Huang, Y., He, Y., Makarcyzk, M. J. & Lin, H. Senolytic peptide FOXO4-DRI selectively removes senescent cells from in vitro expanded human chondrocytes. Front. Bioeng. Biotechnol. 9, 677576 (2021).
Bourgeois, B. et al. The disordered p53 transactivation domain is the target of FOXO4 and the senolytic compound FOXO4-DRI. Nat. Commun. 16, 5672 (2025).
Baar, M. P. et al. Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 169, 132–147.e116 (2017).
Wang, Y. et al. Discovery of piperlongumine as a potential novel lead for the development of senolytic agents. Aging 8, 2915–2926 (2016).
Guerrero, A. et al. Cardiac glycosides are broad-spectrum senolytics. Nat. Metab. 1, 1074–1088 (2019).
Triana-Martínez, F. et al. Identification and characterization of cardiac glycosides as senolytic compounds. Nat. Commun. 10, 4731 (2019).
Beltzig, L., Christmann, M. & Kaina, B. Abrogation of cellular senescence induced by temozolomide in glioblastoma cells: search for senolytics. Cells 11, 2588–2588 (2022).
L’Hôte, V. et al. Ouabain and chloroquine trigger senolysis of BRAF-V600E-induced senescent cells by targeting autophagy. Aging Cell 20, e13447 (2021).
Carpenter, V. J., Saleh, T. & Gewirtz, D. A. Senolytics for cancer therapy: is all that glitters really gold?. Cancers 13, 723 (2021).
Wang, L., Lankhorst, L. & Bernards, R. Exploiting senescence for the treatment of cancer. Nat. Rev. Cancer 22, 340–355 (2022).
Tantawy, S. I. et al. Targeting MCL-1 protein to treat cancer: opportunities and challenges. Front. Oncol. 13, 1226289 (2023).
Troiani, M. et al. Single-cell transcriptomics identifies Mcl-1 as a target for senolytic therapy in cancer. Nat. Commun. 14, 4386 (2023).
Saliev, T. & Singh, P. B. Targeting senescence: a review of senolytics and senomorphics in anti-ageing interventions. Biomolecules 15, 860 (2025).
You, L. & Wu, Q. Cellular senescence in tumor immune escape: mechanisms, implications, and therapeutic potential. Crit. Rev. Oncol. Hematol. 208, 104628 (2025).
Song, P., An, J. & Zou, M. H. Immune clearance of senescent cells to combat ageing and chronic diseases. Cells 9, https://doi.org/10.3390/cells9030671 (2020).
Khandwala, P., Desai, D. & Lau, A. Prevalence of autoimmune diseases in patients treated with immune checkpoint inhibitors: an epidemiological study using a global network of health care organizations. ACR Open Rheumatol. 7, e11787 (2025).
Qin, S. et al. Novel immune checkpoint targets: moving beyond PD-1 and CTLA-4. Mol. Cancer 18, 155 (2019).
Pereira, B. I. et al. Senescent cells evade immune clearance via HLA-E-mediated NK and CD8+ T cell inhibition. Nat. Commun. 10, 2387 (2019).
Wang, T. W. et al. Blocking PD-L1–PD-1 improves senescence surveillance and ageing phenotypes. Nature 611, 358–364 (2022).
Amor, C. et al. Senolytic CAR T cells reverse senescence-associated pathologies. Nature 583, 127–132 (2020).
Deng, Y. et al. Targeting senescent cells with NKG2D-CAR T cells. Cell Death Discov. 10, 217 (2024).
Amor, C. et al. Prophylactic and long-lasting efficacy of senolytic CAR T cells against age-related metabolic dysfunction. Nat. Aging 4, 336–349 (2024).
Huang, Y. & Liu, T. Step further towards targeted senolytic therapy: therapeutic potential of uPAR-CAR T cells for senescence-related diseases. Signal Transduct. Target. Ther. 5, 155 (2020).
Lim, W. A. The emerging era of cell engineering: harnessing the modularity of cells to program complex biological function. Science 378, 848–852 (2022).
Abdo, L., Batista-Silva, L. R. & Bonamino, M. H. Cost-effective strategies for CAR-T cell therapy manufacturing. Mol. Ther. Oncol. 33, 200980 (2025).
Cao, L. Y. et al. CAR-T cell therapy clinical trials: global progress, challenges, and future directions from ClinicalTrials.gov insights. Front. Immunol. 16, 1583116 (2025).
Yang, D. et al. NKG2D-CAR T cells eliminate senescent cells in aged mice and nonhuman primates. Sci. Transl. Med. 15, eadd1951 (2023).
Fan, G., Wang, Y. & Wang, R. Recharging aged-CAR with NAD+ to boost immunotherapy. Trends Cancer 11, 711–712 (2025).
Kim, J. et al. Inhibition of glutaminase elicits senolysis in therapy-induced senescent melanoma cells. Cell Death Dis. 15, 902 (2024).
Johmura, Y. et al. Senolysis by glutaminolysis inhibition ameliorates various age-associated disorders. Science 371, 265–270 (2021).
Kureel, S. K. & Rasmussen, B. B. Targeting ferroptosis to eliminate senescent cells: mechanisms and therapeutic potential. Aging Dis. 16, 1–18 (2025).
Loo, T. M. et al. Senescence-associated lysosomal dysfunction impairs cystine deprivation-induced lipid peroxidation and ferroptosis. Nat. Commun. 16, 6617 (2025).
Feng, Y. et al. Iron retardation in lysosomes protects senescent cells from ferroptosis. Aging 16, 7683–7703 (2024).
Beatty, A. et al. Ferroptotic cell death triggered by conjugated linolenic acids is mediated by ACSL1. Nat. Commun. 12, 2244 (2021).
Xiang, W. et al. Senescent macrophages induce ferroptosis in skeletal muscle and accelerate osteoarthritis-related muscle atrophy. Nat. Aging 5, 1295–1316 (2025).
Cruickshank-Taylor, A. B. et al. Proteolysis targeting chimeras as senolytics: an emerging senotherapy for combating aging. J. Pharmacol. Exp. Ther. 392, 103752 (2025).
Vogler, M. et al. The BCL2 family: from apoptosis mechanisms to new advances in targeted therapy. Signal Transduct. Target. Ther. 10, 91 (2025).
Mu, X. et al. Nanotechnology advances proteolysis targeting chimeras (PROTACs): transition from basic research to clinical application. Int. J. Nanomed. 20, 12177–12198 (2025).
Eladl, O. Molecular glues and PROTACs in targeted protein degradation: mechanisms, advances, and therapeutic potential. Biochem. Pharmacol. 242, 117297 (2025).
Liu, Y. et al. Proteolysis-targeting chimera (PROTAC) nanomedicines toward cancer treatment: from synthesis to therapeutic delivery. Biomaterials 325, 123621 (2026).
Madhukar, G. et al. E3 ubiquitin ligases and their therapeutic potential in disease management. Biochem. Pharmacol. 236, 116875 (2025).
Bondeson, D. P. et al. Catalytic in vivo protein knockdown by small-molecule PROTACs. Nat. Chem. Biol. 11, 611–617 (2015).
Burslem, G. M. et al. The advantages of targeted protein degradation over inhibition: an RTK case study. Cell Chem. Biol. 25, 67–77.e63 (2018).
Khan, S. et al. A selective BCL-XL PROTAC degrader achieves safe and potent antitumor activity. Nat. Med. 25, 1938–1947 (2019).
Yang, Y. et al. A BCL-xL/BCL-2 PROTAC effectively clears senescent cells in the liver and reduces MASH-driven hepatocellular carcinoma in mice. Nat. Aging 5, 386–400 (2025).
He, Y. et al. Using proteolysis-targeting chimera technology to reduce navitoclax platelet toxicity and improve its senolytic activity. Nat. Commun. 11, 1996 (2020).
Khan, S. et al. PROTAC-mediated dual degradation of BCL-xL and BCL-2 is a highly effective therapeutic strategy in small-cell lung cancer. Cells 13, 528 (2024).
Chang, L. C. et al. A first-in-class dual degrader of Bcl-2/Bcl-xL reverses HIV latency and minimizes ex vivo reservoirs from patients. Int. J. Mol. Sci. 26, 2772 (2025).
Chen, C. et al. Liver-targeting chimeras as a potential modality for the treatment of liver diseases. J. Control. Release 374, 627–638 (2024).
Yuan, S. et al. Liver-targeted degradation of BRD4 reverses hepatic fibrosis and enhances metabolism in murine models. Theranostics 15, 7270–7290 (2025).
Peng, Y. et al. Development of cathepsin B-responsive GalNAc-PROTACs for hepatocyte-targeting protein degradation. J. Med. Chem. 69, 517–532 (2026).
Iyer, A. K., Khaled, G., Fang, J. & Maeda, H. Exploiting the enhanced permeability and retention effect for tumor targeting. Drug Discov. Today 11, 812–818 (2006).
Syahputra, E. W. et al. PROTAC delivery strategies for overcoming physicochemical properties and physiological barriers in targeted protein degradation. Pharmaceutics 17, 501 (2025).
Boyajian, J. L. et al. Microbiome and human aging: probiotic and prebiotic potentials in longevity, skin health and cellular senescence. Nutrients 13, 4550 (2021).
Biţă, A. et al. Nicotinamide riboside, a promising vitamin B3 derivative for healthy aging and longevity: current research and perspectives. Molecules 28, 6078 (2023).
Jing, M. & Jiang, Y. Microbiome-mediated crosstalk between T2DM and MASLD: a translational review focused on function. Front. Endocrinol. 16, 1677175 (2025).
Rees, N. P., et al. Defining microbiota-derived metabolite butyrate as a senomorphic: therapeutic potential in the age-related T cell senescence. Aging cell 24, https://doi.org/10.1111/acel.70257 (2025).
Saeedi Saravi, S. S. et al. Gut microbiota-dependent increase in phenylacetic acid induces endothelial cell senescence during aging. Nat. Aging 5, 1025–1045 (2025).
Calder, P. C. Omega-3 fatty acids and inflammatory processes: from molecules to man. Biochem. Soc. Trans. 45, 1105–1115 (2017).
Gutiérrez, S., Svahn, S. L. & Johansson, M. E. Effects of omega-3 fatty acids on immune cells. Int. J. Mol. Sci. 20, 5028 (2019).
Shvedova, M. et al. Topical ABT-263 treatment reduces aged skin senescence and improves subsequent wound healing. Aging 17, 16–32 (2024).
Fuchs, C. D. & Trauner, M. Role of bile acids and their receptors in gastrointestinal and hepatic pathophysiology. Nat. Rev. Gastroenterol. Hepatol. 19, 432–450 (2022).
Jang, D. H. et al. The connection between aging, cellular senescence and gut microbiome alterations: a comprehensive review. Aging Cell 23, e14315 (2024).
Yang, H. et al. Gut microbial-derived phenylacetylglutamine accelerates host cellular senescence. Nat. Aging 5, 401–418 (2025).
Wilmanski, T. et al. Gut microbiome pattern reflects healthy ageing and predicts survival in humans. Nat. Metab. 3, 274–286 (2021).
Boehme, M. et al. Microbiota from young mice counteracts selective age-associated behavioral deficits. Nat. Aging 1, 666–676 (2021).
Sangfuang, N. et al. Investigating the bidirectional interactions between senotherapeutic agents and human gut microbiota. Eur. J. Pharm. Sci. 209, 107098 (2025).
Tang, W. H., Kitai, T. & Hazen, S. L. Gut microbiota in cardiovascular health and disease. Circ. Res. 120, 1183–1196 (2017).
Xiang, X. et al. Novel PET imaging probe for quantitative detection of senescence in vivo. J. Med. Chem. 67, 5924–5934 (2024).
Alum, E. U. et al. Targeting cellular senescence for healthy aging: advances in senolytics and senomorphics. Drug Des. Dev. Ther. 19, 8489–8522 (2025).
Lindner, T. et al. Targeting of activated fibroblasts for imaging and therapy. EJNMMI Radiopharm. Chem. 4, 16 (2019).
Teo, Y. V. et al. Cell-free DNA as a biomarker of aging. Aging Cell 18, e12890 (2019).
Moser, T. et al. Bridging biological cfDNA features and machine learning approaches. Trends Genet 39, 285–307 (2023).
Mansfield, L. et al. Emerging insights in senescence: pathways from preclinical models to therapeutic innovations. NPJ Aging 10, 53 (2024).
Wang, X. et al. Rejuvenation strategy for inducing and enhancing autoimmune response to eliminate senescent cells. Aging Dis. 16, 2273–2292 (2024).
Khan, S. et al. Immune dysregulation in cancer patients developing immune-related adverse events. Br. J. Cancer 120, 63–68 (2019).
Justice, J. N. et al. Development of clinical trials to extend healthy lifespan. Cardiovasc. Endocrinol. Metab. 7, 80–83 (2018).
Rani, A. et al. Failure of senolytic treatment to prevent cognitive decline in a female rodent model of aging. Front. Aging Neurosci. 16, 1384554 (2024).
Guida, J. L. et al. Strategies to prevent or remediate cancer and treatment-related aging. J. Natl. Cancer Inst. 113, 112–122 (2021).
Kirkland, J. L. & Tchkonia, T. Senolytic drugs: from discovery to translation. J. Intern. Med. 288, 518–536 (2020).
Kulkarni, A. S., Gubbi, S. & Barzilai, N. Benefits of metformin in attenuating the hallmarks of aging. Cell Metab. 32, 15–30 (2020).
Wang, L. et al. Targeting p21Cip1 highly expressing cells in adipose tissue alleviates insulin resistance in obesity. Cell Metab. 34, 75–89.e78 (2022).
Rossi, M. & Abdelmohsen, K. The emergence of senescent surface biomarkers as senotherapeutic targets. Cells 10, 1740 (2021).
Wong, F. et al. Discovering small-molecule senolytics with deep neural networks. Nat. Aging 3, 734–750 (2023).
Smer-Barreto, V. et al. Discovery of senolytics using machine learning. Nat. Commun. 14, 3445 (2023).
Xu, Z. et al. A generative AI-discovered TNIK inhibitor for idiopathic pulmonary fibrosis: a randomized phase 2a trial. Nat. Med. 31, 2602–2610 (2025).
Tang, Q. et al. AI-driven robotics laboratory identifies pharmacological TNIK inhibition as a potent senomorphic agent. Aging Dis. 16, 345–360 (2024).
Finch, C. E. Senolytics and cell senescence: historical and evolutionary perspectives. Evol. Med. Public Health 12, 82–85 (2024).
Inci, N. et al. Translation of cellular senescence to novel therapeutics: insights from alternative tools and models. Front. Aging 3, 828058 (2022).
Ocampo, A. et al. In vivo amelioration of age-associated hallmarks by partial reprogramming. Cell 167, 1719–1733.e1712 (2016).
Krizhanovsky, V. et al. Senescence of activated stellate cells limits liver fibrosis. Cell 134, 657–667 (2008).
Demaria, M. et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev. Cell 31, 722–733 (2014).
Lavarti, R. et al. The context-dependent effect of cellular senescence: from embryogenesis and wound healing to aging. Ageing Res. Rev. 109, 102760 (2025).
Peng, Y. et al. Acting on ethics and governance of aging research. Trends Mol. Med. 29, 419–421 (2023).
Saliev, T. & Singh, P. B. Age reprogramming: innovations and ethical considerations for prolonged longevity. Biomed. Rep. 22, 96 (2025).
Riessland, M. et al. Therapeutic targeting of senescent cells in the CNS. Nat. Rev. Drug Discov. 23, 817–837 (2024).
Barthet, V. J. A. & Lowe, S. W. Killing wisely: precision senolytics in the age of frailty. Genes Dev. 39, 910–913 (2025).
Kim, Y. et al. Metabolic remodeling in cancer and senescence and its therapeutic implications. Trends Endocrinol. Metab. 35, 732–744 (2024).
Wu, J. et al. Advancing T-cell immunotherapy for cellular senescence and disease: mechanisms, challenges, and clinical prospects. Ageing Res. Rev. 109, 102783 (2025).
Yang, J. H. et al. Loss of epigenetic information as a cause of mammalian aging. Cell 186, 305–326.e327 (2023).
Sarkar, T. J. et al. Transient non-integrative expression of nuclear reprogramming factors promotes multifaceted amelioration of aging in human cells. Nat. Commun. 11, 1545 (2020).
Lu, J. Y. et al. Prevalent mesenchymal drift in aging and disease is reversed by partial reprogramming. Cell 188, 5895–5911.e5817 (2025).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 82172210) and the Xijing Hospital Advancement Program (Grant No. XJZT24CY38). The funders had no role in the study design, data collection, analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Weidong Zhang: Conceptualization, Writing—Original Draft, Visualization, Literature Curation. Shijie Song: Writing—Review and Editing, Visualization. Yue Zhang: Writing—Review and Editing, Literature Curation. Yong Pan: Writing—Review and Editing, Validation. Dahai Hu: Conceptualization, Supervision, Funding Acquisition, Writing—Review and Editing. Yunchuan Wang: Conceptualization, Supervision, Funding Acquisition, Writing—Review and Editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, W., Song, S., Zhang, Y. et al. Emerging strategies in senotherapeutics: from broad-spectrum senolysis to precision reprogramming. npj Aging (2026). https://doi.org/10.1038/s41514-026-00355-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41514-026-00355-z