Abstract
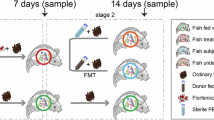
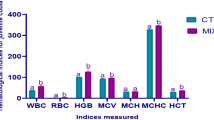
Dietary faba bean enhances fish muscle quality but concurrently reduces growth performance. The gut microbiota critically modulates muscle growth and quality. However, the specific microbial taxa, metabolites, and regulatory mechanisms responsible remain to be elucidated. This study established a differential gut microbiota model in faba-bean-fed Yellow River carp (Cyprinus carpio), used whole-intestinal microbiota transplantation (WIMT) to directly test its effect on muscle quality, and supplemented the key bacterium and its metabolite to confirm their contribution. After a 6-week faba bean diet, growth performance declined, whereas muscle texture improved (P < 0.05). This improvement was concomitant with a higher abundance of the genera Aeromonas and Cetobacterium in the gut. Following 8 weeks of daily WIMT from faba-bean-fed donors, Yellow River carp maintained normal growth performance (P > 0.05) and simultaneously showed improved muscle texture, characterized by more small-diameter fibers, lower fat content, and higher collagen levels (P < 0.05), recapitulating the donor’s key muscle phenotype. Meanwhile, WIMT reshaped the gut microbiome composition and its metabolic profile, and the marker species Cetobacterium somerae and its metabolite acetic acid showed associations with improvements in muscle quality. Further in vivo validation indicated that C. somerae reduced fat deposition and improved muscle texture, an effect possibly linked to activation of the AMPK-PGC-1α-FoxO pathway, and its metabolite acetic acid mirrored these changes. This study reveals the direct impact of gut microbiota on muscle quality through WIMT in Yellow River carp, provides novel evidence of the fish gut-muscle axis, and offers a scientific basis for improving muscle quality.

Similar content being viewed by others
Data availability
The “Gut microbiome sequencing data (fed with faba beans for 6 weeks)” are available in the NCBI SRA repository with the accession ID https://www.ncbi.nlm.nih.gov/search/all/?term=PRJNA1311008, reference number PRJNA1311008. The “Gut microbiome sequencing data (antibiotic treatment)” can be accessed in the NCBI SRA repository with the accession ID https://www.ncbi.nlm.nih.gov/search/all/?term=PRJNA1310288, reference number PRJNA1310288. The “Gut microbiome sequencing data (WIMT)” are deposited in the NCBI SRA repository and can be found using the accession ID https://www.ncbi.nlm.nih.gov/search/all/?term=PRJNA1310847, reference number PRJNA1310847. The “Muscle tissue transcriptome sequencing data” are stored in the NCBI SRA repository and are accessible via the accession ID https://www.ncbi.nlm.nih.gov/sra/PRJNA1344608, reference number PRJNA1344608. The “SCFAs data from intestinal contents” are available in the NGDC OMIX repository with the accession ID https://ngdc.cncb.ac.cn/omix/release/OMIX012256, reference number OMIX012256. The “SCFAs data from CS-CS” can be accessed in the NGDC OMIX repository using the accession ID https://ngdc.cncb.ac.cn/omix/release/OMIX012255, reference number OMIX012255. The “Non-targeted metabolomics data of intestinal contents” are deposited in the MetaboLights repository and can be found using the accession ID https://www.ebi.ac.uk/metabolights/MTBLS13128, reference number MTBLS13128.
References
Wang, A. R., Ran, C., Ringø, E. & Zhou, Z. G. Progress in fish gastrointestinal microbiota research. Rev. Aquac. 10, 626–640 (2018).
Feng, L. T., Chen, Z. N. & Bian, H. J. Skeletal muscle: Molecular structure, myogenesis, biological functions, and diseases. Med. Commun. 5, e649 (2024).
Hawley, J. A. Microbiota and muscle highway-two way traffic. Nat. Rev. Endocrinol. 16, 71–72 (2020).
Chew, W. X. et al. Gut-muscle crosstalk. A perspective on influence of microbes on muscle function. Front. Med. (Lausanne) 9, 1065365 (2023).
He, Y. W. et al. Gut microbes-muscle axis in muscle function and meat quality. Sci. China-Life Sci. https://doi.org/10.1007/s11427-024-2885-4 (2025).
Lahiri, S. et al. The gut microbiota influences skeletal muscle mass and function in mice. Sci. Transl. Med. 11, eaan5662 (2019).
Liu, C. R. et al. Understanding the gut microbiota and sarcopenia: a systematic review. J. Cachexia, Sarcopenia Muscle 12, 1393–1407 (2021).
Frampton, J., Murphy, K. G., Frost, G. & Chambers, E. S. Short-chain fatty acids as potential regulators of skeletal muscle metabolism and function. Nat. Metab. 2, 840–848 (2020).
Lei, J. Q. et al. Intestinal Microbiota Regulate Certain Meat Quality Parameters in Chicken. Front. Nutr. 25, 747705 (2022).
Luo, Z. B. et al. Fecal transplant from myostatin deletion pigs positively impacts the gut-muscle axis. Elife 12, e81858 (2023).
Chapagain, P., Walker, D., Leeds, T., Cleveland, B. M. & Salem, M. Distinct microbial assemblages associated with genetic selection for high- and low- muscle yield in rainbow trout. BMC Genomics 21, 820 (2020).
Su, W. F. et al. Recirculating aquaculture-fasting strategy (RASF) to modulate gut microbiota and enhance the fish meat quality of bighead carp (Aristichthys nobilis). Food Chem. 481, 143946 (2025).
Wang, E. L. et al. Rice flowering improves the muscle nutrient, intestinal microbiota diversity, and liver metabolism profiles of tilapia (Oreochromis niloticus) in rice-fish symbiosis. Microbiome 10, 231 (2022).
He, Y. L. et al. Decoding the gut-microbiota-muscle nexus: Multi-omics integration reveals mTOR driven flesh modulation in rice-fish co-cultured common carp (Cyprinus carpio). Aquaculture 612, 743090 (2026).
Wu, H. X. et al. Oligosaccharides improve the flesh quality and nutrition value of Nile tilapia fed with high carbohydrate diet. Food Chem. Mol. Sci. 3, 100040 (2021).
Wu, H. X. et al. Microbiota derived butyrate affected the muscle texture of Nile tilapia (Oreochromis niloticus) fed with different protein sources. Food Chem. 393, 133392 (2022).
Zhang, Y., Liang, X., Zhan, W., Chen, L. & Lou, B. Short-chain fatty acids affect skeletal muscle composition, fiber recruitment, and metabolism of the small yellow croaker (Larimichthys polyactis). Aquac. Rep. 45, 103169 (2025).
Li, L. K. et al. Valerate enhances flesh hardness by inhibiting muscle proliferation and differentiation in Nile tilapia (Oreochromis niloticus): An in vivo and in vitro study. Aquaculture 600, 742222 (2025).
Khalili, T. S. & Sampels, S. Nutritional Value of Fish: Lipids, Proteins, Vitamins, and Minerals. Rev. Fish. Sci. Aquac. 26, 243–253 (2018).
Song, D. Y. et al. Effects of faba bean on growth performance and fillet texture of Yellow River carp, Cyprinus carpio haematopterus. Aquac. Rep. 17, 100379 (2020).
Yu, E. M. et al. Proteomic and metabolomic basis for improved textural quality in crisp grass carp (Ctenopharyngodon idellus C.et V) fed with a natural dietary prooxidant. Food Chem. 325, 126906 (2020).
Chen, L. J. et al. Reactive oxygen species (ROS)-mediated regulation of muscle texture in grass carp fed with dietary oxidants. Aquaculture 8, 737150 (2021).
Li, Z. F. et al. Diet influences the accumulation of short-chain fatty acids associated with the gut microbiota in the grass carp (Ctenopharyngodon idellus) hindgut. Appl. Ecol. Environ. Res. 17, 13435–13453 (2019).
Zhou, L. et al. Intestinal Microbiota of Grass Carp Fed Faba Beans: A Comparative Study. Microorganisms 7, 465 (2019).
Yu, K. et al. Exploring the muscle-hardening mechanisms via the muscle-gut axis in tilapia (Oreochromis niloticus) fed with faba bean (Vicia faba L.) supplementary diets. Aquac. Rep. 37, 102268 (2024).
Li, N. et al. Spatial heterogeneity of bacterial colonization across different gut segments following inter-species microbiota transplantation. Microbiome 8, 161 (2020).
Yang, Y. P. et al. Whole intestinal microbiota transplantation is more effective than fecal microbiota transplantation in reducing the susceptibility of DSS-induced germ-free mice colitis. Front. Immunol. 14, 1143526 (2023).
Listrat, A. et al. How muscle structure and composition influence meat and flesh quality. Sci. World J 2016, 3182746 (2016).
Wang, Z. et al. The flesh texture of teleost fish: Characteristics and interventional strategies. Rev. Aquac. 16, 508–535 (2024).
Dong, M. et al. Dietary protein levels changed the hardness of muscle by acting on muscle fiber growth and the metabolism of collagen in sub-adult grass carp (Ctenopharyngodon idella). J. Anim. Sci. Biotechnol. 13, 109 (2022).
Shi, Y. et al. Dietary sanguinarine supplementation recovers the decrease in muscle quality and nutrient composition induced by high-fat diets of grass carp (Ctenopharyngodon idella). Anim. Nutr. 17, 208–219 (2024).
Zhang, X. Y. et al. Mechanisms of hepatic dysfunction in Nile tilapia (Oreochromis niloticus) fed a high-fava bean (Vicia faba L.) diet. Anim. Nutr. 22, 61–71 (2025).
Xu, G. L. et al. Impact of Faba Bean (Vicia faba L.) Diet and Subsequent Withdrawal on GIFT Tilapia (Oreochromis niloticus) Muscle Quality. Fishes 10, 170 (2025).
Gallet, A. et al. Disruption of fish gut microbiota composition and holobiont’s metabolome during a simulated Microcystis aeruginosa (Cyanobacteria) bloom. Microbiome 11, 108 (2023).
Qi, X. Z. et al. Vitamin B12 produced by Cetobacterium somerae improves host resistance against pathogen infection through strengthening the interactions within gut microbiota. Microbiome 11, 135 (2023).
Wang, A. R. et al. Intestinal Cetobacterium and acetate modify glucose homeostasis via parasympathetic activation in zebrafish. Gut Microbes 13, 1–15 (2021).
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2021).
Novelle, M. G., Naranjo-Martínez, B., Lόpez-Cάnovas, J. L. & Díaz-Ruiz, A. Fecal microbiota transplantation, a tool to transfer healthy longevity. Ageing Res. Rev. 103, 102585 (2025).
Mo, X. X. et al. Faecal microbiota transplantation from young rats attenuates age-related sarcopenia revealed by multiomics analysis. J. Cachexia, Sarcopenia Muscle 14, 2168–2183 (2023).
Yin, J. et al. Obese Ningxiang pig-derived microbiota rewires carnitine metabolism to promote muscle fatty acid deposition in lean DLY pigs. Innov. (Camb.) 4, 100486 (2023).
Zhou, H. et al. Gut microbiota absence and transplantation affect growth and intestinal functions: An investigation in a germ-free pig model. Anim. Nutr. 7, 295–304 (2021).
Wu, D. Y. et al. Effects of fecal microbiota transplantation and fecal virome transplantation on LPS-induced intestinal injury in broilers. Poult. Sci. 103, 103316 (2024).
Liu, Q. Y. et al. Early fecal microbiota transplantation continuously improves chicken growth performance by inhibiting age-related Lactobacillus decline in jejunum. Microbiome 13, 49 (2025).
Han, Q., Huang, X. G., Yan, F. Y., Yin, J. & Xiao, Y. P. The role of gut microbiota in the skeletal muscle development and fat deposition in pigs. Antibiotics (Basel) 11, 793 (2022).
Meng, Z. et al. Nutritive value of faba bean (Vicia faba L.) as a feedstuff resource in livestock nutrition: A review. Food Sci. Nutr. 9, 5244–5262 (2021).
Hintze, K. J. et al. Broad scope method for creating humanized animal models for animal health and disease research through antibiotic treatment and human fecal transfer. Gut Microbes 5, 183–191 (2014).
Ruiz, A., Gisbert, E. & Andree, K. B. Impact of the diet in the gut microbiota after an inter-species microbial transplantation in fish. Sci. Rep. 14, 4007 (2024).
Han, Z. R. et al. Fecal microbiota transplantation accelerates restoration of florfenicol-disturbed intestinal microbiota in a fish model. Commun. Biol. 7, 1006 (2024).
Garlock, T. M. et al. Environmental, economic, and social sustainability in aquaculture: the aquaculture performance indicators. Nat. Commun. 15, 5274 (2024).
Zhang, Z. M. et al. Potential Functions of the Gut Microbiome and Modulation Strategies for Improving Aquatic Animal Growth. Rev. Aquac. 17, e12959 (2025).
Zhang, J. M. et al. Effects of fecal bacteria on growth, digestive capacity, antioxidant capacity, intestinal health of large yellow croaker (Larimichthys crocea) larvae. Aquaculture 562, 738796 (2023).
Raymo, G., Ali, A., Ahmed, R. O. & Salem, M. Early-Life Fecal Transplantation from High Muscle Yield Rainbow Trout to Low Muscle Yield Recipients Accelerates Somatic Growth through Respiratory and Mitochondrial Efficiency Modulation. Microorganisms 12, 261 (2024).
Song, D. Y. et al. Effects of faba bean (Vicia faba L.) on fillet quality of Yellow River carp (Cyprinus carpio) via the oxidative stress response. Food Chem 388, 132953 (2022).
Ghosh, S. S., Wang, J., Yannie, P. J. & Ghosh, S. Intestinal barrier dysfunction, LPS translocation, and disease development. J. Endocr. Soc. 4, bvz039 (2020).
Kong, M. et al. A well-established gut microbiota enhances the efficiency of nutrient metabolism and improves the growth performance of trachinotus ovatus. Int. J. Mol. Sci. 25, 5525 (2024).
Lan, X. et al. Alterations of the gut microbiota and metabolomics associated with the different growth performances of macrobrachium rosenbergii Families. Anim. (Basel) 13, 1539 (2023).
Chapman, M. A., Mukund, K., Subramaniam, S., Brenner, D. & Lieber, R. L. Three distinct cell populations express extracellular matrix proteins and increase in number during skeletal muscle fibrosis. Am. J. Physiol. -Cell Physiol. 312, C131–C143 (2017).
Chen, W. J., Lin, I. H., Lee, C. W. & Chen, Y. F. Aged skeletal muscle retains the ability to remodel extracellular matrix for degradation of collagen deposition after muscle injury. Int. J. Mol. Sci. 22, 2123 (2021).
Hellerschmied, D. et al. Molecular features of the UNC-45 chaperone critical for binding and folding muscle myosin. Nat. Commun. 10, 4781 (2019).
Hadjisavva, R., Anastasiou, O., Ioannou, P. S., Zheltkova, M. & Skourides, P. A. Adherens junctions stimulate and spatially guide integrin activation and extracellular matrix deposition. Sci. Rep. 40, 111091 (2022).
Li, Y. B. et al. Microbial metabolite sodium butyrate enhances the anti-tumor efficacy of 5-fluorouracil against colorectal cancer by modulating PINK1/Parkin signaling and intestinal flora. Sci. Rep. 14, 13063 (2024).
Liu, L., Fu, C. Y. & Li, F. C. Acetate affects the process of lipid metabolism in rabbit liver, skeletal muscle and adipose tissue. Anim. (Basel) 9, 799 (2019).
Qian, L. et al. Peroxisome proliferator-activated receptor gamma coactivator-1 (PGC-1) family in physiological and pathophysiological process and diseases. Signal Transduct. Target. Ther. 9, 50 (2024).
Fasano, C., Disciglio, V., Bertora, S., Signorile, M. L. & Simone, C. FOXO3a from the Nucleus to the Mitochondria: A Round Trip in Cellular Stress Response. Cells 8, 1110 (2019).
Stecher, B. The roles of inflammation, nutrient availability and the commensal microbiota in enteric pathogen infection. Microbiol. Spectrum 3, 3 (2015).
He, X. G. et al. Effects of broad bean diet on the growth performance, muscle characteristics, antioxidant capacity, and intestinal health of Nile Tilapia (Oreochromis niloticus). Anim. (Basel) 13, 3705 (2023).
Li, L. K. et al. Intestinal microbiota contributes to the heterogeneity of fat deposition by promoting mitochondrial fatty acid β-oxidation. Gut Microbes 17, 2593076 (2025).
Mann, E. R., Lam, Y. K. & Uhlig, H. H. Short-chain fatty acids: linking diet, the microbiome and immunity. Nat. Rev. Immunol. 24, 577–595 (2024).
Liao, X. M. et al. Vitamin D influences gut microbiota and acetate production in zebrafish (Danio rerio) to promote intestinal immunity against invading pathogens. Gut microbes 15, 2187575 (2023).
Chen, Y. X. et al. Probiotic efficacy of Cetobacterium somerae (CGMCC No. 28843): promoting intestinal digestion, absorption, and structural integrity in juvenile grass carp (Ctenopharyngodon idella). J. Anim. Sci. Biotechnol. 16, 103 (2025).
Wang, S. D., Li, X., Zhang, M. Z. & Li, M. Cetobacterium somerae-derived argininosuccinic acid promotes intestinal and liver ureagenesis to alleviate ammonia intoxication. Microbiome 13, 163 (2025).
Cao, X. L. et al. Determining the potential roles of branched-chain amino acids in the regulation of muscle growth in common carp (Cyprinus carpio) based on transcriptome and MicroRNA sequencing. Aquac. Nutr. 2023, 7965735 (2023).
Cao, X. L. et al. Branched-chain amino acids target miR-203a/fosb axis to promote skeletal muscle growth in common carp (Cyprinus carpio). Aquac. Nutr. 2025, 9406490 (2025).
Chen, Q. et al. An integrated analysis of transcriptome and metabolome reveals three BCAAs relieve lipid accumulation by inhibiting lipid synthesis and promoting lipid oxidation in the liver of largemouth bass (Micropterus salmoides). Aquaculture 581, 19 (2024).
Maruta, H., Abe, R. & Yamashita, H. Effect of long-term supplementation with acetic acid on the skeletal muscle of aging sprague dawley rats. Int. J. Mol. Sci. 23, 4691 (2022).
Iwao, M. et al. Supplementation of branched-chain amino acids decreases fat accumulation in the liver through intestinal microbiota-mediated production of acetic acid. Sci. Rep. 10, 18768 (2020).
Li, L. et al. Acetic acid influences BRL-3A cell lipid metabolism via the AMPK signalling pathway. Cell. Physiol. Biochem. 45, 2021–2030 (2018).
Liu, Y. et al. Vitamin D3 mitigates lipopolysaccharide-induced oxidative stress, tight junction damage and intestinal inflammatory response in yellow catfish, Pelteobagrus fulvidraco. Comp. Biochem Physiol. C. Toxicol. Pharm. 243, 108982 (2021).
Doyle, A., Zhang, G. H., Abdel Fattah, E. A., Eissa, N. T. & Li, Y. P. Toll-like receptor 4 mediates lipopolysaccharide-induced muscle catabolism via coordinate activation of ubiquitin-proteasome and autophagy-lysosome pathways. FASEB J 25, 99–110 (2011).
Dai, Y. J. et al. Molecular cloning of adipose triglyceride lipase (ATGL) gene from blunt snout bream and its expression after LPS-induced TNF-α factor. Fish. Physiol. Biochem. 44, 1143–1157 (2018).
Feng, J. C. et al. Dietary sodium acetate (SA) improves the growth performance, intestinal health, and carbohydrate metabolism of juvenile common carp (Cyprinus carpio). Aquac. Rep. 27, 101350 (2022).
Xie, M. X. et al. Effects of Cetobacterium somerae fermentation product on gut and liver health of common carp (Cyprinus carpio) fed diet supplemented with ultra-micro ground mixed plant proteins. Aquaculture 543, 736943 (2021).
Mi, J. L. et al. Dietary (−)-Epicatechin supplementation regulates myofiber development, fillet quality, and antioxidant status of Yellow River carp (Cyprinus carpio). Aquaculture 572, 739542 (2023).
Campos, V. J. et al. Characterization of fillets and skins from two varieties of genetically improved farmed Nile tilapia (Oreochromis niloticus). PLoS ONE 20, e0314928 (2025).
Cheng, L. J. et al. Lactiplantibacillus plantarum Ameliorated Growth Performance and Intestinal Inflammation by Modulating Gut Microbial Structure and Function of Yellow River Carp (Cyprinus carpio). Probiotics Antimicrob Proteins https://doi.org/10.1007/s12602-025-10589-0 (2025).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant numbers U22A20532, 32473180); and the Special Fund for Henan Agriculture Research System (grant numbers HARS-22-16-S). The authors acknowledge the participants’ contributions to this research, and we are grateful to Professor Xiaolin Meng, Doctor Qingyang Su, and Master Yuan Liu (College of Fisheries, Henan Normal University, Xinxiang, China) for isolating and culturing the Cetobacterium somerae CFH_001 strain.
Author information
Authors and Affiliations
Contributions
All authors, L.J.C., Y.J.L., Y.J.Z., C.B.Q., L.P.Y., X.Y., and G.X.N. contributed to the conceptualizations, methodology, and experimentations of the manuscript. L.J.C., Y.J.L., and Y.J.Z.: investigation, animal husbandry, sample collection, measurement of experimental parameters, analysis, and interpretation of data. L.J.C.: visualization and writing the original draft. L.J.C., Y.J.L., and Y.J.Z.: writing, reviewing, and editing. L.J.C., Y.J.L., C.B.Q., L.P.Y., and X.Y.: validation, analysis, and interpretation of data, reviewing and editing the manuscript. G.X.N.: supervision, project administratio,n and funding acquisition. All the authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cheng, L., Li, Y., Zhang, Y. et al. Cetobacterium somerae as a microbial correlate of improved muscle quality after intestinal microbiota transplantation in Yellow River carp (Cyprinus carpio). npj Biofilms Microbiomes (2026). https://doi.org/10.1038/s41522-026-00955-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41522-026-00955-3