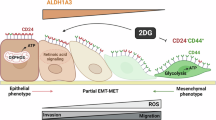
Abstract
Breast cancer stem cells (BCSCs) have been proposed as the cause of resistance to conventional treatments and of breast cancer recurrence and metastasis. This study provides compelling evidence for the role of the transcription factor POU1F1 in the increase of BCSC-like. Using POU1F1-overexpressing and knock-down breast cancer cell lines, as well as immunodeficient mouse models, our data demonstrate that POU1F1 induces a BCSC-like phenotype in breast tumor cells by deregulating markers such as CD24, CD44, CD133, and ALDH. These phenotypic modifications correlate with functional changes, i.e., increased clonogenicity, mammosphere formation, and glycolysis. In addition, we found that a subpopulation of MCF-7 cells with overexpression of POU1F1 and elevated ALDH expression exhibits both a high tumor-initiating capacity and increased resistance to chemotherapy and radiotherapy treatments. Mechanistically, these features are mediated by POU1F1 activation of the IL-6/JAK2/STAT3 pathway and up-regulation of ALDH. Janus kinase inhibitors and monoclonal anti-IL6 receptor antibodies significantly decrease ALDH expression, colony, and mammosphere formation, suggesting possible use of pharmacological inhibitors of the IL-6/JAK2/STAT3 pathway in breast tumors with elevated POU1F1 levels.
Similar content being viewed by others
Data availability
Raw and processed data for sequencing studies are available from NCBI’s Gene Expression Omnibus and are accessible through GEO Series accession numbers GSE287812 (MCF-7 cells), GSE287732 (HMEC), with and without POU1F1 overexpression (POU1F1) (RNA-seq). All other data will be available from the authors upon request.
References
Yuan, S., Norgard, R. J. & Stanger, B. Z. Cellular plasticity in cancer. Cancer Discov. 9, 837–851 (2019).
Shenoy, S. Cell plasticity in cancer: a complex interplay of genetic, epigenetic mechanisms and tumor micro-environment. Surg. Oncol. 34, 154–162 (2020).
Pérez-González, A., Bévant, K. & Blanpain, C. Cancer cell plasticity during tumor progression, metastasis and response to therapy. Nat. Cancer 4, 1063–1082 (2023).
Batlle, E. et al. The transcription factor Snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat. Cell Biol. 2, 84–89 (2000).
Cano, A. et al. The transcription factor Snail controls epithelial–mesenchymal transitions by repressing E-cadherin expression. Nat. Cell Biol. 2, 76–83 (2000).
Dongre, A. & Weinberg, R. A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 20, 69–84 (2018).
Mani, S. A. et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133, 704–715 (2008).
Meacham, C. E. & Morrison, S. J. Tumour heterogeneity and cancer cell plasticity. Nature 501, 328–337 (2013).
Saw, P. E., Liu, Q., Wong, P. P. & Song, E. Cancer stem cell mimicry for immune evasion and therapeutic resistance. Cell Stem Cell 31, 1101–1112 (2024).
Massagué, J. & Ganesh, K. Metastasis-initiating cells and ecosystems. Cancer Discov. 11, 971–994 (2021).
Oskarsson, T., Batlle, E. & Massagué, J. Metastatic stem cells: sources, niches, and vital pathways. Cell Stem Cell 14, 306–321 (2014).
Batlle, E. & Clevers, H. Cancer stem cells revisited. Nat. Med. 23, 1124–1134 (2017).
Lytle, N. K., Barber, A. G. & Reya, T. Stem cell fate in cancer growth, progression and therapy resistance. Nat. Rev. Cancer 18, 669–680 (2018).
Torborg, S. R., Li, Z., Chan, J. E. & Tammela, T. Cellular and molecular mechanisms of plasticity in cancer. Trends Cancer 8, 735–746 (2022).
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J. & Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proc. Nat. Acad. Sci. USA 100, 3983–3988 (2003).
Ginestier, C. et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 1, 555–567 (2007).
Andersen, B. & Rosenfeld, M. G. POU domain factors in the neuroendocrine system: lessons from developmental biology provide insights into human disease. Endocr. Rev. 22, 2–35 (2001).
Lefevre, C. et al. Tissue-specific expression of the human growth hormone gene is conferred in part by the binding of a specific trans-acting factor. EMBO J. 6, 971–981 (1987).
Nelson, C. J., Albert, V. R., Elsholtz, H. P., Lu, L. I. & Rosenfeld, M. G. Activation of cell-specific expression of rat growth hormone and prolactin genes by a common transcription factor. Science 239, 1400–1405 (1988).
Dollé, P. et al. Expression of GHF-1 protein in mouse pituitaries correlates both temporally and spatially with the onset of growth hormone gene activity. Cell 60, 809–820 (1990).
Gil-Puig, C., Blanco, M., García-Caballero, T., Segura, C. & Pérez-Fernández, R. Pit-1/GHF-1 and GH expression in the MCF-7 human breast adenocarcinoma cell line. J. Endocrinol. 173, 161–167 (2002).
Gil-Puig, C. et al. Pit-1 is expressed in normal and tumorous human breast and regulates GH secretion and cell proliferation. Eur. J. Endocrinol. 153, 335–344 (2005).
Ben-Batalla, I. et al. Deregulation of the Pit-1 transcription factor in human breast cancer cells promotes tumor growth and metastasis. J. Clin. Invest. 120, 4289–4302 (2010).
Seoane, S. et al. POU1F1 transcription factor promotes breast cancer metastasis via recruitment and polarization of macrophages. J. Pathol. 49, 381–394 (2019).
Martínez-Ordoñez, A. et al. POU1F1 transcription factor induces metabolic reprogramming and breast cancer progression via LDHA regulation. Oncogene 40, 2725–2740 (2021).
Tang, C., Lei, X., Xiong, L., Hu, Z. & Tang, B. HMGA1B/2 transcriptionally activated-POU1F1 facilitates gastric carcinoma metastasis via CXCL12/CXCR4 axis-mediated macrophage polarization. Cell Death Dis. 12, 422 (2021).
Liu, S. et al. Breast cancer stem cells are regulated by mesenchymal stem cells through cytokine networks. Cancer Res. 71, 614–624 (2011).
Korkaya, H., Liu, S. & Wicha, M. S. Regulation of cancer stem cells by cytokine networks: attacking cancer’s inflammatory roots. Clin. Cancer Res. 17, 6125–6129 (2011).
Korkaya, H. et al. Activation of an IL6 inflammatory loop mediates trastuzumab resistance in HER2+ breast cancer by expanding the cancer stem cell population. Mol. Cell 47, 570–584 (2012).
Kang, S., Narazaki, M., Metwally, H. & Kishimoto, T. Historical overview of the interleukin-6 family cytokine. J. Exp. Med. 217, e20190347 (2020).
Li, W. et al. Unraveling the roles of CD44/CD24 and ALDH1 as cancer stem cell markers in tumorigenesis and metastasis. Sci. Rep. 7, 13856 (2017).
Shan, N. L., Shin, Y., Yang, G., Furmanski, P. & Suh, N. Breast cancer stem cells: a review of their characteristics and the agents that affect them. Mol. Carcinog. 60, 73–100 (2021).
Tang, C., Zhang, H., Deng, W.-S., Xiong, L.-Q. & Zkou, L.-Q. Role of POU1F1 promoting the properties of stemness of gastric carcinoma through ENO1-mediatedglycolysis reprogramming. Kaohsiung J. Med. Sci. 39, 904–915 (2023).
DeBerardinis, R. J., Lum, J. J., Hatzivassiliou, G. & Thompson, C. B. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 7, 11–20 (2008).
Liao, J. et al. Ovarian cancer spheroid cells with stem cell-like properties contribute to tumor generation, metastasis and chemotherapy resistance through hypoxia-resistant metabolism. PLoS ONE 9, e84941 (2014).
Luo, M. et al. ZMYND8 protects breast cancer stem cells against oxidative stress and ferroptosis through activation of NRF2. J. Clin. Invest. 134, e171166 (2024).
Rosenbluth, J. M. et al. Organoid cultures from normal and cancer-prone human breast tissues preserve complex epithelial lineages. Nat. Commun. 11, 1711 (2020).
Ye, F. et al. The Presence of EpCAM-/CD49f+ cells in breast cancer is associated with a poor clinical outcome. J. Breast Cancer 18, 242–242 (2015).
Taniguchi, S. et al. Tumor-initiating cells establish an IL-33–TGF-β niche signaling loop to promote cancer progression. Science 369, eaay1813 (2020).
Gilabert, M. et al. Poly (ADP-Ribose) polymerase 1 (PARP1) overexpression in human breast cancer stem cells and resistance to olaparib. PLoS ONE 9, e104302 (2014).
Liu, C.-C. et al. ALDH1A1 activity in tumor-initiating cells remodels myeloid-derived suppressor cells to promote breast cancer progression. Cancer Res. 81, 5919–5934 (2021).
Zanoni, M., Bravaccini, S., Fabbri, F. & Arienti, C. Emerging roles of aldehyde dehydrogenase isoforms in anti-cancer therapy resistance. Front. Med. 9, 795762 (2022).
Gao, Z., Xue, K., Zhang, L. & Wei, M. Over-expression of POU class 1 homeobox 1 transcription factor (Pit-1) predicts poor prognosis for breast cancer patients. Med. Sci. Monit. 22, 4121–4125 (2016).
Lee, H. et al. Cancer stem cells: landscape, challenges and emerging therapeutic innovations. Sig. Transduct. Target Ther. 10, 248 (2025).
Quan, Q. et al. Cancer stem-like cells with hybrid epithelial/mesenchymal phenotype leading the collective invasion. Cancer Sci. 111, 467–476 (2020).
Yousefnia, S., Ghaedi, K., Forootan, F. S. & Esfahani, M. H. N. Characterization of the stemness potency of mammospheres isolated from the breast cancer cell lines. Tumor Biol. 41, 1–14 (2019).
Manore, S. G., Doheny, D. L., Wong, G. L. & Lo, H.-W. IL-6/JAK/STAT3 signaling in breast cancer metastasis: biology and treatment. Front. Oncol. 12, 866014 (2022).
Wang, T. et al. JAK/STAT3-regulated fatty acid β-oxidation is critical for breast cancer stem cell self-renewal and chemoresistance. Cell Metab. 27, 136–150.e5 (2018).
Ma, Y. et al. LncRNA XIST regulates breast cancer stem cells by activating proinflammatory IL-6/STAT3 signaling. Oncogene 42, 1419–1437 (2023).
Lin, W.-H. et al. STAT3 phosphorylation at Ser727 and Tyr705 differentially regulates the EMT–MET switch and cancer metastasis. Oncogene 40, 791–805 (2020).
Yousefnia, S. et al. Mechanistic pathways of malignancy in breast cancer stem cells. Front. Oncol. 10, 452 (2020).
Elston, C. W. & Ellis, I. O. Pathological prognosis factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathol 19, 403–410 (1991).
Acknowledgements
We thank Dr. A. Martinez-Ordoñez for his valuable suggestions and critical reading of the manuscript. This study was supported by FEDER/Ministerio de Ciencia, Innovación y Universidades-Agencia Estatal de Investigación (PID2021-127394OB-100) and the EU MSCA-DC eRaDicate-101119427 to R.P.-F. L.A.C. and S.R.-G. are fellows of the Xunta de Galicia Predoctoral Scholarship 2020. M.G. is a Marie-Curie PhD student (101119427).
Author information
Authors and Affiliations
Contributions
Leandro Avila: Methodology, investigation, validation, formal analysis, data curation, and writing. Samuel Seoane: Methodology, investigation, formal analysis, and data curation. Sandra Rodriguez-Gonzalez: Methodology and formal analysis. Magda Gois: Methodology, investigation, and formal analysis. Maria Efigenia Arias: Methodology, investigation, and formal analysis. David Martinez-Delgado: Data curation. Noemi Lado: Data curation. Tomas Garcia-Caballero: Data curation, methodology, and investigation. Pablo Aguiar: Methodology and investigation. Roman Perez-Fernandez: Visualization, validation, supervision, resources, project administration, methodology, funding acquisition, formal analysis, data curation, conceptualization, and writing & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Avila, L., Seoane, S., Rodriguez-Gonzalez, S. et al. POU1F1 induces cancer stem cell-like traits in breast cancer cells by IL-6/JAK2/STAT3 activation and enrichment of ALDH. npj Breast Cancer (2026). https://doi.org/10.1038/s41523-026-00929-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-026-00929-w