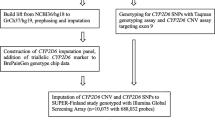
Abstract
Tamoxifen’s pharmacokinetics are strongly influenced by the highly polymorphic CYP2D6, while the influence of other genetic variants has been inconclusive. To further delineate this genotypic-phenotypic impact, we conducted a multi-ancestry genome-wide association study in 636 hormone-receptor-positive (HR+) breast cancer (BC) patients treated with 20 mg tamoxifen daily for ≥8 weeks and validated these genetic determinants in another 869 patients. Association with clinical outcomes was examined in 1326 non-metastatic HR+ patients receiving adjuvant tamoxifen. A genome-wide significant association with Z-endoxifen levels was observed at the CYP2D6 locus on chromosome 22 and its downstream region of TCF20 rs932376 A > G. Both CYP2D6 metabolizer status and TCF20 rs932376 A > G were independent predictors of endoxifen levels in multivariable analysis. CYP2D6 metabolizer status accounted for greater variability of mean endoxifen levels compared to TCF20 rs932376 A > G (91.2% vs 48.8%). These findings were replicated in validation cohorts. Neither TCF20 rs932376 nor CYP2D6 metabolizer status was significantly associated with BC outcomes after adjustment for known prognostic factors. Our study confirmed that CYP2D6 metabolizer status remains as the prime predictor of steady-state Z-endoxifen levels, while TCF20 rs932376 A > G has a smaller, independent effect. Both genetic factors were not associated with BC clinical outcomes.
Similar content being viewed by others
Data Availability
Data supporting this study have been de-identified and only known to the respective research teams involved in patient recruitment. The data is not publicly available due to confidentiality of the research participants and is governed by the data sharing policies of the respective research institutions. Please contact the corresponding author (BC) for any request.
References
Lim, J. S. L. et al. Impact of CYP2D6, CYP3A5, CYP2C9 and CYP2C19 polymorphisms on tamoxifen pharmacokinetics in Asian breast cancer patients. Br. J. Clin. Pharmacol. 71, 737–750 (2011).
Lim, J. S. L. et al. Association of CYP2C19*2 and associated haplotypes with lower norendoxifen concentrations in tamoxifen-treated Asian breast cancer patients. Br. J. Clin. Pharmacol. 81, 1142–1152 (2016).
Sutiman, N. et al. Pharmacogenetics of UGT1A4, UGT2B7 and UGT2B15 and Their Influence on Tamoxifen Disposition in Asian Breast Cancer Patients. Clin. Pharmacokinet. 55, 1239–1250 (2016).
Lim, Y. C., Desta, Z., Flockhart, D. A. & Skaar, T. C. Endoxifen (4-hydroxy-N-desmethyl-tamoxifen) has anti-estrogenic effects in breast cancer cells with potency similar to 4-hydroxy-tamoxifen. Cancer Chemother. Pharmacol. 55, 471–478 (2005).
Mürdter, T. E. et al. Activity levels of tamoxifen metabolites at the estrogen receptor and the impact of genetic polymorphisms of phase I and II enzymes on their concentration levels in plasma. Clin. Pharmacol. Ther. 89, 708–717 (2011).
Madlensky, L. et al. Tamoxifen metabolite concentrations, CYP2D6 genotype, and breast cancer outcomes. Clin. Pharmacol. Ther. 89, 718–725 (2011).
Gong, I. Y. et al. Determination of clinically therapeutic endoxifen concentrations based on efficacy from human MCF7 breast cancer xenografts. Breast Cancer Res. Treat. 139, 61–69 (2013).
Teft, W. A. et al. CYP3A4 and seasonal variation in vitamin D status in addition to CYP2D6 contribute to therapeutic endoxifen level during tamoxifen therapy. Breast Cancer Res. Treat. 139, 95–105 (2013).
Saladores, P. et al. Tamoxifen metabolism predicts drug concentrations and outcome in premenopausal patients with early breast cancer. Pharmacogenomics J. 15, 84–94 (2015).
Schroth, W. et al. Improved Prediction of Endoxifen Metabolism by CYP2D6 Genotype in Breast Cancer Patients Treated with Tamoxifen. Front. Pharmacol. 8, 582 (2017).
Goetz, M. P. & Ingle, J. N. CYP2D6 genotype and tamoxifen: considerations for proper nonprospective studies. Clin. Pharmacol. Ther. 96, 141–144 (2014).
Rae, J. M., Regan, M., Leyland-Jones, B., Hayes, D. F. & Dowsett, M. CYP2D6 genotype should not be used for deciding about tamoxifen therapy in postmenopausal breast cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 31, 2753–2755 (2013).
Brauch, H. & Schwab, M. Prediction of tamoxifen outcome by genetic variation of CYP2D6 in post-menopausal women with early breast cancer. Br. J. Clin. Pharmacol. 77, 695–703 (2014).
Sanchez-Spitman, A. B. et al. A Genome-Wide Association Study of Endoxifen Serum Concentrations and Adjuvant Tamoxifen Efficacy in Early-Stage Breast Cancer Patients. Clin. Pharmacol. Ther. 116, 155–164 (2024).
Hennig, E. E. et al. Non-CYP2D6 Variants Selected by a GWAS Improve the Prediction of Impaired Tamoxifen Metabolism in Patients with Breast Cancer. J. Clin. Med. 8, 1087 (2019).
Khor, C. C. et al. Cross-Ancestry Genome-Wide Association Study Defines the Extended CYP2D6 Locus as the Principal Genetic Determinant of Endoxifen Plasma Concentrations. Clin. Pharmacol. Ther. 113, 712–723 (2023).
Binkhorst, L., Mathijssen, R. H. J., Jager, A. & van Gelder, T. Individualization of tamoxifen therapy: much more than just CYP2D6 genotyping. Cancer Treat. Rev. 41, 289–299 (2015).
Rekdal, C., Sjøttem, E. & Johansen, T. The nuclear factor SPBP contains different functional domains and stimulates the activity of various transcriptional activators. J. Biol. Chem. 275, 40288–40300 (2000).
Sanz, L., Moscat, J. & Diaz-Meco, M. T. Molecular characterization of a novel transcription factor that controls stromelysin expression. Mol. Cell. Biol. 15, 3164–3170 (1995).
Lyngsø, C. et al. Interaction between the transcription factor SPBP and the positive cofactor RNF4. An interplay between protein binding zinc fingers. J. Biol. Chem. 275, 26144–26149 (2000).
Elvenes, J. et al. Pax6 represses androgen receptor-mediated transactivation by inhibiting recruitment of the coactivator SPBP. PloS One 6, e24659 (2011).
Gburcik, V., Bot, N., Maggiolini, M. & Picard, D. SPBP is a phosphoserine-specific repressor of estrogen receptor alpha. Mol. Cell. Biol. 25, 3421–3430 (2005).
Pan, X., Ning, M. & Jeong, H. Transcriptional Regulation of CYP2D6 Expression. Drug Metab. Dispos. Biol. Fate Chem. 45, 42–48 (2017).
Yang, X. et al. Systematic genetic and genomic analysis of cytochrome P450 enzyme activities in human liver. Genome Res. 20, 1020–1036 (2010).
MacLehose, R. F., Ahern, T. P., Collin, L. J., Li, A. & Lash, T. L. CYP2D6 Phenotype and Breast Cancer Outcomes: A Bias Analysis and Meta-Analysis. Cancer Epidemiol. Biomarkers Prev. 34, 224–233 (2025).
Goetz, M. P. et al. First-in-Human Phase I Study of the Tamoxifen Metabolite Z-Endoxifen in Women With Endocrine-Refractory Metastatic Breast Cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 35, 3391–3400 (2017).
Mürdter, T. E. et al. Supplementation of Tamoxifen with low-dose Endoxifen in breast cancer patients with impaired tamoxifen metabolism (TAMENDOX): a randomized controlled phase 1/2 trial. Clin. Cancer Res. 31, 4903–4911 (2025).
Awada, Z. et al. Pharmacogenomics variation in drug metabolizing enzymes and transporters in relation to docetaxel toxicity in Lebanese breast cancer patients: paving the way for OMICs in low and middle income countries. Omics J. Integr. Biol. 17, 353–367 (2013).
Eccles, D. et al. Prospective study of Outcomes in Sporadic versus Hereditary breast cancer (POSH): study protocol. BMC Cancer 7, 160 (2007).
Puszkiel, A. et al. Factors affecting tamoxifen metabolism in patients with breast cancer: preliminary results of the French PHACS study. Clin. Pharmacol. Ther. 106, 585–595 (2019).
Arellano, C., Allal, B., Goubaa, A., Roché, H. & Chatelut, E. An UPLC-MS/MS method for separation and accurate quantification of tamoxifen and its metabolites isomers. J. Pharm. Biomed. Anal. 100, 254–261 (2014).
Caudle, K. E. et al. Standardizing CYP2D6 genotype to phenotype translation: consensus recommendations from the clinical pharmacogenetics implementation consortium and Dutch pharmacogenetics working group. Clin. Transl. Sci. 13, 116–124 (2020).
Harrell, F. E. Regression Modeling Strategies: With Applications to Linear Models, Logistic Regression, and Survival Analysis. (Springer, 2001).
Tolaney, S. M. et al. Updated standardized definitions for efficacy end points (STEEP) in adjuvant breast cancer clinical trials: STEEP version 2.0. J. Clin. Oncol. 39, 2720–2731 (2021).
Little, J. et al. STrengthening the reporting of genetic association studies (STREGA)-an extension of the STROBE statement. Genet. Epidemiol. 33, 581–598 (2009).
Acknowledgements
This study was supported by grants from the National Medical Research Council Singapore (NMRC/1159/2008, NMRCB1011, NRFCG1516, NMRCG13163 and NMRC/CIRG/1423/2015, MOH-000377), the National Research Foundation of Singapore grant (NRF-NRFI2018-01); the Robert Bosch Foundation, Stuttgart, Germany and German Federal Ministry of Education and Research (BMBF; 01ZP0502, 01EK1509A); the Deutsche Foschungsgemeinschaft (DFG, German Research Foundation) under Germany's Excellence Strategy - EXC 2180–390900677 and DFG grants SCHR 1323/2-1 and MU 1727/2-1; the German Cancer Consortium (DKTK), Partner Site Tübingen, 72074 Tübingen and the Interfaculty Center for Pharmacogenomics and Drug Research (ICEPHA), University of Tuebingen (no grant number). The funding sources had no involvement in the study design, collection, analysis and interpretation of data, writing of the manuscript and decision for publication. Additional funding for this study was provided by the Biomedical Research Council, Agency for Science, Technology and Research, Singapore (no grant number). The funders had no role in the study design, collection, analysis and interpretation of data as well as drafting and submission of this manuscript.
Author information
Authors and Affiliations
Contributions
BC conceived and designed the study. EHL, EC, SC, TEM, JEA, NS, JSLL, SSL, CA, ZL, KSS, DME, WJT, TM, NKZ, SW, BG, LD, QG, CC, HME, LH, JD, RCHN, YSY, MW, FYW, NSW, PACS, RD, PK, UL, TL, AT, ES, ME, UH, PAF, MWB, FD, MWK, HBB, WS, WR, MS were involved in patient collection, conduct of study and data collection. CCK, WSO, TEM, HBB, MS, FT and BC contributed to data analysis and interpretation. CCK, WSO and BC drafted the manuscript. All authors participated in manuscript editing, review and approval of final revised manuscript.
Corresponding author
Ethics declarations
Competing interests
Carlos Caldas is a member of the AstraZeneca External Science Panel and has received research grants (administered by the University of Cambridge) from AstraZeneca, Genentech, Roche and Servier. Helena M. Earl has received research grants from Roche and Sanofi-Aventis (administered by Cambridge University Hospital Trust), honoraria and travel expenses from Daiichi-Sankyo, Astra Zeneca, Pfizer, Amgen and Prime Oncology all outside the submitted work. Jean E. Abraham has received research grants from AstraZeneca (administered by the Cambridge University Hospital Trust and the University of Cambridge, Dept of Oncology), honoraria and travel expenses from Astra Zeneca and Pfizer all outside the submitted work. All other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Khor, C.C., Ong, W.S., Lim, E.H. et al. A multi-ancestry genome-wide study of tamoxifen metabolism and breast cancer recurrence. npj Breast Cancer (2026). https://doi.org/10.1038/s41523-026-00931-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-026-00931-2