Abstract
Biological mechanoreceptors convert tissue strain into distinct spike trains. In contrast, their soft electronic counterparts still rely on discrete components for sensing, preprocessing, and neuronal firing. Here, we integrate these functional components into a single and scalable device by combining mechano-electric transduction and volatile threshold switching within an Ag/freestanding epitaxial SrTiO3/Pt membrane laminated onto a flexible polyethylene naphthalate substrate. Tensile strain (0–2.6%) lowers Ag⁺ migration energy and reduces the switching voltage from 1.04 to 0.24 V. Under constant bias, the spike frequency increases by more than two orders of magnitude, enabling tunable, self-oscillating ‘neurons’ operating below 100 pJ per spike, comparable to biological mechanoreceptors and ~25× more efficient than current flexible sensors. The device maintains its full functionality after over 400 bending cycles, demonstrating its potential as a mechanically programmable, ultralow-power building block for next-generation electronic skins, soft robotics, and bio-integrated prosthetics.
Similar content being viewed by others
Introduction
Modern electronics are increasingly inspired by biomimicry, the emulation of sophisticated biological processes, enabling advanced sensing and signaling functionalities in areas such as neuromorphic computing, electronic skins (e-skins), bio-inspired robotics, and wearable healthcare devices1,2,3,4,5,6,7,8. A particularly striking feature of biological systems is their capacity to generate distinctly different responses of neurons to identical external stimulus under varying mechanical conditions, such as bending or stretching of skin and skeletal structures9,10.
In humans, mechanical deformation of the skin can result in completely different neural signaling patterns compared to the undeformed state, even under the same stimulus conditions (Fig.1a)11,12. Emulating such mechanosensory neuron behavior in a single, deformable solid-state device would enable the integration of sensing, pre-processing, and neuronal signaling into a unified platform, significantly reducing wiring complexity and energy consumption13,14,15. However, replicating this multi-functional behavior within a single artificial element remains a considerable challenge.
a Schematic illustration comparing action potential patterns generated by biological neurons under mechanical deformation, such as bending or stretching, with those observed in the normal, unstretched state of human skin. b Conventional flexible mechanosensory electronics, wherein strain sensing, signal conversion, and neuronic processing are separated into distinct modules connected by peripheral circuitry, leading to increased system size and power consumption. c Schematic of the device architecture and operational mechanism of the strain-modulated TS device, which integrates strain sensing, charge accumulation, and neuron-like spiking behavior within a single, flexible platform.
Over the past decade, flexible electronics have achieved substantial progress, enabling devices to preserve their functional integrity under significant mechanical stress16,17,18,19. These advancements have enhanced our understanding of the interplay between mechanical strain and electronic performance, largely through mechanical endurance testing or the incorporation of external strain sensors16,17,18,19. However, relatively few approaches leverage mechanical deformation itself as an active input to directly modulate device operation. Examples include piezotronic ZnO tunneling sensors gated by tensile strain20, strain-programmable MoS₂ transistors that exhibit a twofold increase in mobility under 0.7% bending strain21, and diffusive memristors coupled with external capacitors, wherein the spiking frequency can be tuned by mechanical perturbation22. However, none of these systems achieves the integration of sensing, charge accumulation, and neuron-like spiking within a single, flexible element. Most existing artificial mechanosensory systems rely on separate sensing and processing components, necessitating complex electronic interfaces for signal transduction and communication (Fig. 1b)23. This architectural separation increases system complexity and energy demands, and ultimately limits the ability to mimic the dynamic and integrated responses of biological systems.
Here, we demonstrate that mechanical strain can directly modulate neuron-like spiking behavior in a single flexible threshold switching (TS) device, thereby collapsing the entire sensor–neuron architecture into one functional element. (Fig. 1c). The device operates via a volatile TS mechanism governed by silver ion (Ag+) migration, where bending-induced tensile strain directly modulates ion transport. Under identical electrical biases, the application of tensile strain reduces the threshold voltage and increases the spiking frequency. This behavior closely resembles the biological mechanism whereby mechanical stretching of the skin reconfigures ion channel activity, resulting in altered neuronal responses under constant external stimuli. We anticipate that this mechanically driven switching paradigm will be especially valuable for next-generation neuromorphic sensors, soft robotics, wearable electronics, and biomedical interfaces, where the real-time and dynamic translation of mechanical cues into electrical responses is imperative24,25,26,27. Our findings establish a foundational strategy for exploiting mechanical deformation as an active and tunable input modality in flexible electronic systems.
Results
Freestanding epitaxial membrane on flexible substrates
Freestanding single-crystalline membranes were fabricated through the sequence illustrated in Fig. 2a–d. First, a sacrificial Sr₃Al₂O₆ (SAO) layer and the desired functional oxide layer were sequentially deposited on atomically flat (001)-oriented SrTiO₃ (STO) substrates using pulsed laser deposition systems28,29,30,31(Fig. 2a; Supplementary Fig. S1; see Methods). In this study, we used STO as the target freestanding membrane, whereas a Pt-coated polyethylene naphthalate (PEN) film served as the flexible substrate. The resulting heterostructure was then spin-coated with photoresist and attached to thermal-release tape to provide mechanical support. When the stack was floated on deionized water, the SAO layer selectively dissolved, releasing a freestanding membrane with photoresist/tape mechanical support (Fig. 2b; see Methods). After complete removal of SAO, the membrane was transferred onto the flexible substrate (Fig. 2c). Finally, the photoresist was removed with acetone, yielding a strain-free membrane robustly integrated on a flexible substrate (Fig. 2d). The thicknesses of STO and SAO were determined by AFM. STO thickness (~100 nm) was measured from a transferred STO membrane on a PEN substrate, whereas SAO thickness (~20 nm) was measured from a masking-based calibration method (Supplementary Fig. S2).
a–d Schematic illustration of the water-assisted transfer process of the freestanding STO membrane onto a flexible PEN substrate. e High-resolution θ–2θ X-ray diffraction (XRD) profiles centered on the STO (002) reflection before (black) and after (blue) transfer. f AFM images (1 × 1 µm²) of the membrane surface in the as-transferred state and after 400 bending cycles between tensile strain ε = 2.61% and 0%. g Cross-sectional schematic of the STO/Pt/PEN (top), along with optical images of the fabricated heterostructure (bottom left) and the device mounted on a mechanical bending stage for strain application (bottom right).
To verify the epitaxial quality before and after transfer onto the flexible PEN substrate, high-resolution X-ray diffraction (XRD) measurements were performed in θ-2θ scan mode for both STO/SAO/(001)-oriented STO and STO/Pt/PEN. The XRD θ–2θ profile reveals that the (002) reflection of the freestanding STO film remains at the same 2θ position (2θ = 46.65° ± 0.005°) before and after transfer onto the Pt-coated PEN substrate (Fig. 2e). This result highlights the structural robustness of the freestanding membrane and the non-destructive nature of the water-assisted delamination and transfer technique. The double-peak feature in the STO (002) reflection arises from the lattice-strain contrast between the unstrained substrate and the strained epitaxial film32. The STO film grown on the SAO sacrificial layer experiences in-plane tensile strain due to the larger effective lattice constant of SAO (3.961 Å), leading to a reduced out-of-plane lattice constant (3.892 Å) relative to bulk STO (3.905 Å).
The lateral topography of the freestanding membranes was further characterized using atomic force microscopy (AFM) and optical microscopy. These analyses confirmed that the millimeter-scale films were free of cracks in their unstrained state (Supplementary Fig. S3). Since strain relaxation typically occurs in the vicinity of cracks33, the absence of visible cracks suggests that the STO membranes maintain homogeneous in-plane strain, thereby providing optimal conditions for strain-dependent measurements. AFM imaging over 1 × 1 µm² areas reveals that the as-transferred STO films on Pt-coated PEN substrates are atomically smooth, exhibiting a root mean square roughness (Rrms) of 0.6 nm (Fig. 2f). After 400 bending cycles at a maximum tensile strain of ε = 2.61%, Rrms increases slightly to 0.8 nm, representing an approximate 33% rise but still remaining within the sub-nanometer regime.
Figure 2g shows schematic cross-sectional and optical images of the flexible device structure, consisting of a freestanding-STO/Pt/PEN with a top Ag electrode (see Methods). The bending radius was precisely controlled using a vernier caliper, enabling reproducible cyclic bending tests with well-defined and consistent tensile strain levels.
Strain-modulated TS device
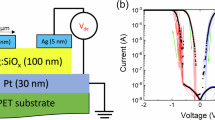
In devices incorporating a Ag top electrode and a STO dielectric layer, the TS switching behavior is governed by the dynamic migration of Ag+ ions, following the electrochemical reaction Ag → Ag+ + e−. Under an applied electric field, Ag+ ions migrate toward the bottom electrode, initiating the switching process34,35 (Fig. 3a). TS characteristics of the Ag/freestanding-STO/Pt/PEN devices was evaluated by sweeping the external voltage from 0 V to 1.3 V in 0.01 V increments, with a compliance current of 1 µA to prevent permanent breakdown (Fig. 3b). When the applied voltage exceeds the threshold voltage (Vth), Ag+ ions from the top electrode infiltrate the STO layer and form localized conductive filaments, resulting in a pronounced drop in resistance and transition to the low-resistance (ON) state. This ON state is maintained by a hold voltage (Vhold), which sustains the integrity of the filament. When the applied voltage drops below Vhold, the conductive filaments dissolve as Ag+ ions return to the top electrode, restoring the device to its high-resistance (OFF) state.
a Schematic illustration of the TS mechanism. b Representative current–voltage (I-V) characteristic showing abrupt switching behavior at the threshold voltage (Vth) and spontaneous return to the high-resistance state when the applied voltage falls below the hold voltage. c Crystal-chemistry origin of strain sensitivity: tensile strain modulates the local lattice environment. d Energy barrier of Ag+ ion, depending on tensile strain.
To utilize the interplay between ion migration and the epitaxial crystallinity of STO, tensile strain was applied to the Ag/freestanding-STO/Pt/PEN device. STO has a perovskite lattice structure in which corner-sharing TiO₆ octahedra form straight and parallel Ti–O–Ti chains36 (Fig. 3c). Under tensile stress, these chains rotate about in-plane axes, resulting in the bending the Ti–O–Ti bonds and an increase in the in-plane lattice parameter d37. This structural distortion increases the cation–vacancy separation (r), thereby weakening the Madelung potential term38.
As a result, the Coulombic potential well that confines oxygen vacancies (VO+) becomes shallower, effectively lowering the energy barrier for vacancy migration. Density functional theory (DFT) calculations using the nudged elastic band (NEB) method predict that applying 2–4% tensile strain can reduce the VO+ migration barrier by up to 25%39. Although these calculations were reported in earlier work, the trend supports our interpretation that tensile strain facilitates Ag⁺ migration by widening the oxygen sublattice channels. Our device operates via Ag+ ion migration rather than oxygen vacancy transport, strain-induced lattice expansion is expected to similarly enhance Ag+ dynamics, particularly when Ag⁺ diffuses along the oxygen sublattice. While quantitative NEB-based analysis of Ag⁺ migration remains limited, existing evidence suggests that mechanical strain facilitates ion transport by widening lattice channels and reducing local energy barriers (Fig. 3d). This effect is attributed to the expansion of lattice pathways and the alleviation of structural hindrance for mobile cations.
To investigate the strain-dependent electrical behavior of the Ag/freestanding-STO/Pt/PEN device, current-voltage (I-V) measurements were conducted under 6 different tensile strain (ε) conditions (Fig. 4a; Supplementary Fig. S4). TS curves were obtained by repeatedly sweeping the voltage from 0 V to 2 V in 0.01 V steps over 30 consecutive cycles (Supplementary Fig. S5). The results show that increasing strain systematically lowers the 30-cycle averaged Vth. As ε increases from 0 to 2.61%, the mean Vth decreases from 1.04 ± 0.05 V to 0.24 ± 0.05 V with a slope of –0.28 ± 0.02 V per percent strain (Fig. 4b; Supplementary Table 1). To further evaluate the reproducibility of volatile switching, we measured I–V characteristics from multiple Ag/freestanding-STO/Pt/PEN devices under identical electrical conditions and varying mechanical strain states (Supplementary Fig. S6). The device-to-device variation in Vth, quantified across 20 devices, showed a normalized standard deviation (standard deviation/mean) of 0.09 at 0% strain and 0.36 at 2.61% strain, indicating increased variability under high mechanical deformation.
a Representative I-V characteristics measured under six tensile strain levels (ε = 0, 0.35, 0.87, 1.37, 1.67, and 2.61%). Measurements were conducted using a voltage sweep from 0 V to 2 V with a step of 10 mV and a current compliance of 1 µA. b Strain-dependent variation of the Vth, averaged over n = 30 switching cycles for each strain condition. c, Activation energy (Ea) for Ag+ ion migration, which is extracted via Arrhenius analysis of the filament formation kinetics (see Supplementary Fig. S7).
Furthermore, we observe that the activation energy (Ea) in the Ag/freestanding-STO/Pt/PEN device decreases as the applied strain increases (Fig. 4c; Supplementary Fig. S7). Since ion diffusion coefficient follows:
the reduced Ea exponentially accelerates ion transport, enabling easier and faster filament formation. These findings confirm that mechanical elongation lowers the energy barrier governing ion migration and filament nucleation. Memristive neuron circuits generally operate under an integrate-and-fire mechanism, in which a spike is generated once the accumulated membrane potential exceeds Vth. In this context, a reduced Vth enables neuron-like firing at lower voltages, while a decreased Ea accelerates charge integration. Together, these effects shorten the inter-spike interval and increase the firing rate under a given stimulus. Thus, strain engineering offers a powerful strategy to tune both the energy efficiency and dynamic behavior of memristive neuromorphic devices.
Strain-modulated neuron-like properties
To investigate the impact of mechanical deformation on neuron-like behavior, we systematically varied the bending strain of Ag/freestanding-STO/Pt/PEN TS device22. The high relative permittivity (εr ≈ 300) and geometric thickness (d ≈ 100 nm) of the STO layer yield a parasitic capacitance on the order of tens of picofarads (Cpar ≈ ε0εrA/d)40,41, furnishing an internal charge-integration node that eliminates the need for an external membrane capacitor42,43. Combined with the volatile TS characteristics, this intrinsic capacitance enables a self-contained relaxation-oscillator loop without any external capacitance components.
Figure 5 shows the electromechanical modulation of our neuron-like TS device under incremental tensile strain (ε = 0–2.61%). This study aims to elucidate the relationship between mechanical strain and neural emulation performance, thereby advancing the development of robust and flexible biomimetic systems. For each strain level, the ‘Output Voltage’ in Fig. 5 corresponds to the transient voltage across the external load resistor (50 MΩ), which directly reflects the current through the device during strain-dependent spiking (Supplementary Fig. S8). As illustrated in Fig. 5a, the neuron-like spiking characteristics are strongly modulated by applied strain. Figure 5b quantifies this effect: the minimum bias required to initiate sustained oscillations decreases monotonically with strain from 2.0 V at ε = 0% to 0.7 V at ε = 2.61% (intermediate values of 1.85 V at 0.35%, 1.40 V at 0.87%, 1.20 V at 1.37%, and 0.85 V at 1.67%) (Supplementary Fig. S9). This ~65% reduction in excitation threshold demonstrates that tensile deformation concurrently lowers the intrinsic switching barrier and the circuit-level drive voltage required for autonomous spiking.
a Output voltage traces measured across a 50 MΩ load resistor under a constant DC input bias (Vin = 1–2.2 V) at six different tensile strain levels (ε = 0, 0.35, 0.87, 1.37, 1.67, and 2.61%). b Minimum input voltage (Vin) required to sustain continuous spiking activity as a function of ε. c Firing frequency as a function of strain at different Vin values. d Energy consumption per spike plotted against ε. e Spike trains recorded while toggling ε between 0% (flat state) and 2.61% (bent state) at constant Vin of 2 V. f Stability of the firing rate evaluated after 400 mechanical bending–release cycles. g Energy–complexity map comparing 9 state-of-the-art sensory-neuron circuits with the present strain-programmable system (indicated by red star). An asterisk (*) following a number indicates that the value is ≥ that number. h, Conceptual schematic of a large-area array composed of strain-programmable TS neurons, highlighting the potential for scalable integration in neuromorphic sensing systems.
Strain also markedly increases the spiking frequency at fixed input voltages (Fig. 5c). The firing rate increases from 0 Hz to 41.17 Hz at an input bias of 1 V; from 0 Hz to 55.88 Hz at 1.5 V; from 5.88 Hz to 82.35 Hz at 2 V; and from 14.77 Hz to 223.5 Hz at 2.2 V. This demonstrates that mechanical strain acts as a robust, voltage-independent tuning parameter, enabling precise modulation of spiking throughput in flexible TS neurons. The firing frequency increases nearly linearly with strain, yielding sensitivities of 14.8–78.5 Hz %⁻¹ across \({V}_{{in}}=1.0{\mbox{-}}2.2\) V and up to 115.8 Hz %⁻¹ in the high-strain region. The corresponding minimum detectable strain, derived from the frequency detection limit (1 Hz), is approximately 0.013% at 2.2 V. These results confirm that the device operates as both a sensitive strain transducer and a neuromorphic oscillator without sacrificing sensing performance (Supplementary Table 2).
Figure 5d quantifies the total energy consumption of the circuit under varying strain conditions. The Ag/freestanding-STO/Pt/PEN device co-locates mechano-electric transduction and TS behavior within a single 100 nm-thick membrane. The high-κ (εr ≈ 300) single-crystalline STO layer helps to concentrate the local electric field near the Ag/STO interface, thereby facilitating Ag⁺ injection and switching at relatively low voltages (~1 V). However, the absolute switching voltage is determined by a combination of interfacial barrier characteristics and strain-modulated ion migration dynamics, rather than by the dielectric constant alone. Owing to its wide bandgap (3.2 eV) and low intrinsic defect density, the switching current remains below 50 µA. Additionally, the transient and diffusive nature of the Ag nanofilaments promotes self-rupture, further limiting charge flow. The freestanding STO membrane possesses an intrinsic capacitance of several tens of picofarads (Supplementary Fig. S10), which supplies the charge storage necessary for self-sustained oscillation without requiring an external capacitor. The low energy consumption (96–300 pJ per spike under Vin = 2 V and Rload = 50 MΩ) arises primarily from the low operation voltage, large load resistance, and short spike width. As a result, the total energy consumed per spiking event, calculated by E =\(\int \left({V}_{{in}}\times I\right)dt\) under an input bias of Vin = 2 V and a local resistance of Rload = 50 MΩ, remains within the range of 96 to 300 pJ across the full strain range (ε = 0–2.61%). This energy consumption falls within the range typical for biological neurons (1–100 pJ) and is up to two orders of magnitude lower than that of state-of-the-art multi-chip flexible sensory-neuron systems18,44,45,46. This consistent system-level comparison highlights that the present single-element device achieves lower total energy consumption by co-locating sensing and spiking functionalities The minimum spiking energy, recorded at 96 pJ, also lies squarely within the estimated metabolic cost of a cortical action potential (50–100 pJ)47 and is over 40 times lower than the most energy-efficient flexible neuromorphic neurons reported to date (3.9–50 nJ per spike)48. Given the capacitive energy \(E=\frac{1}{2}C{V}^{2}\), and the fact that capacitance (\(C\)) scales with electrode area, reducing the top electrode size from 50 × 50 µm² to 10 × 10 µm² would decrease the intrinsic capacitance by a factor of 25. This downscaling would theoretically lower the energy cost per spike to approximately 4 pJ, well below the metabolic noise floor of biological tissue.
Endurance was evaluated through 400 bending cycles oscillating between 0% and 2.61% strain under a fixed bias of 2 V. In the pristine flat state, the junction exhibits sparse spiking at 30 Hz (Fig. 5e). Intermediate endurance measurements up to 300 bending cycles show that the spiking frequency remains constant within ± 10%, demonstrating stable strain-modulated threshold switching and excellent mechanical reliability (Supplementary Fig. S11). Under maximum strain (ε ≈ 2.6%), the spike rate surges to 380 Hz, a 12.7-fold increase that reflects the strain-induced lowering of the Ag⁺ injection barrier. After 400 cycles, the flat-state firing rate modestly increases to 32 Hz (≈ + 6.7%), whereas the strained-state rate is fully recovered to 380 Hz. This negligible drift falls within cycle-to-cycle variation (Fig. 5f), confirming the endurance of mechano-electric transduction and TS behavior.
To assess how effectively the Ag/freestanding-STO/Pt/PEN neuron compresses both power and hardware overhead, we benchmarked ten state-of-the-art sensory-neuron devices on a two-dimensional landscape of circuit complexity versus energy per spike for the complete circuit (Fig. 5g)18,44,45,46,48,49,50,51,52. For comparison, the energy values in Fig. 5g were recalculated using the operating parameters (V, R, Δt) reported in refs. 18,44,45, and 46 to represent total circuit-level energies including contributions from sensors and load networks. The recalculated results yield ~2–6 nJ (VO₂ neuron), ~300 pJ (NbOₓ mott memristor), ~300–900 pJ (NbOₓ mechnoreceptor), and ~0.2–0.4 nJ (TaOₓ sensory system). These values confirm that our single-element mechanosensory neuron achieves low energy consumption of ~96 pJ per spike at the true system level by co-locating sensing and spiking functionalities. Previous systems typically occupy the upper-right quadrant, consuming 2.5 nJ–4.5 µJ per spike and requiring 2–5 external components (e.g., strain sensor, capacitor, resistor). In contrast, our strain-programmable TS neuron resides in the lower-left extreme, achieving 96 pJ per spike (minimum measured value) with only one external resistor. This represents a ∼25-fold energy reduction relative to the next-best flexible system and a 2–5× decrease in component count compared with prior mechanically responsive neurons. These results highlight the advantage of co-locating mechano-electric sensing, charge integration, and TS behavior within a single Ag filament, establishing a new benchmark for ultralow-power, minimal-footprint neuromorphic elements compatible with soft and wearable form factors.
Wafer-scale implementation of this platform, at densities exceeding 1.6 × 10⁴ neurons per cm², could enable direct mechanical-to-spike conversion in soft robots, prosthetics, and epidermal patches, delivering 96 pJ spikes without the need for discrete strain sensors or external capacitors. At a representative spiking rate of 41.2 Hz, the total power dissipation is estimated to be less than 0.1 mW cm⁻², based on the relation P = \(E\times f\times {N}_{{area}}\,\)= 96 pJ × 41.2 Hz × (1.56 ×104) cm-2. This ultra-low power density significantly alleviates thermal and energy constraints in high-resolution mechanosensing applications. Notably, the demonstrated neuron density surpasses the densest mechanoreceptor innervation found in human fingertips (≈2.4 × 10² cm⁻²)53 by two orders of magnitude and exceeds that of the most advanced soft-electronics tactile arrays (455 pixels cm⁻²)54 by approximately 35-fold. Furthermore, a straightforward lithographic reduction of the electrode pad size to 10 × 10 µm2 would theoretically increase the neuron density beyond 10⁶ cm⁻² (Supplementary Fig. S12), a regime previously accessible only in rigid, nanofabricated crossbar memory arrays, while preserving the integrated sensor-neuron functionality within a single-element architecture. Although these advantages remain to be demonstrated in a fully integrated system, our findings suggest that strain-programmable neuromorphic sheets hold strong potential as foundational building blocks for future biomimetic interfaces, including high-density tactile sensing arrays and adaptive musculoskeletal prosthetics.
Discussion
We demonstrate the integration of sensing, charge accumulation, and neuron-like spiking within a single, scalable Ag/freestanding-STO/Pt/PEN membrane, wherein the spiking behavior is directly modulated by mechanical strain. Application of tensile strain to the epitaxial perovskite lattice increases the Ti–O–Ti bond angle, thereby reducing the Ag⁺ migration barrier and monotonically lowering the threshold voltage. This results in a spike frequency enhancement exceeding two orders of magnitude while maintaining energy consumption up to 96 pJ per spike. Importantly, this energy lies within the 1–100 pJ range typical of biological mechanoreceptors55 and is at least 40 times lower than the most advanced soft-electronics neuromorphic devices, which still consume approximately 3.9–50 nJ per spiking event48. The freestanding membrane preserves atomic-scale crystallinity and stable electrical performance over more than 400 mechanical bending cycles. Its intrinsic high-κ capacitance eliminates the need for external capacitive components, enabling neuron densities exceeding 10⁴ cm⁻² under budgets below 0.1 mW cm-². These results establish tensile strain as an efficient, bio-comparable input modality for ultralow-energy neuromorphic operation and highlight the potential of this system as a mechanically compliant building block for large-area electronic skins, soft robotics, and bio-integrated prosthetic interfaces.
Methods
Thin film deposition
STO/SAO films were epitaxially grown on (001) single-crystalline STO substrates via pulsed laser deposition using a KrF excimer laser (Coherent, COMPexPro 205 F). Before deposition, the STO substrates were etched by NH4F-buffered HF solution and annealed at 1000 °C for 1 hour in air to form a TiO2-terminated step-terrace surface. The SAO and STO films were deposited using the same laser energy density of 2.0–2.2 Jcm-2 at a repetition rate of 5, 3 Hz, while maintaining the substrate temperatures of 750 °C and 700 °C, and O2 pressures of 0 mTorr and 100 mTorr, respectively.
Device fabrication
To prepare the flexible substrate, a PEN film was first coated with a 10-nm-thick Pt layer using e-beam evaporation. To transfer the freestanding oxide heterostructures onto these flexible substrates, the sample was coated with photoresist (AZ 5214-E, Merck Performance Materials), adhered to thermal release tape supported by PEN, and immersed in DI water for 3 days. Afterward, the photoresist was removed using acetone, and the sample was rinsed with isopropyl alcohol (IPA). Au/Ag top electrodes (30/25 nm thick, 50 × 50 μm2 in size) were deposited onto the STO surface using an e-beam evaporator through a metal shadow mask.
Characterizations
AFM images of the freestanding STO were obtained using a Pt/Ir-coated tip in a commercial atomic force microscope (Park Systems, XE-100). The I–V characteristics in DC mode were measured using a probe station equipped with a semiconductor characterization system (Keithley, 4200-SCS). For neuron-like spiking measurements, the Ag/freestanding-STO/Pt/PEN device was connected in series with a 50 MΩ load resistor and biased by a DC source (Vin) using a Keithley 4200 system. The output voltage (Vout) was recorded across the load resistor with a Tektronix oscilloscope (6.25 GS/s), such that Vout = I × Rload. This configuration forms a relaxation-oscillator circuit, where the intrinsic capacitance of the STO layer and the volatile threshold switching of Ag⁺ ions generate self-sustained spiking signals.
Energy calculation
The energy consumption per spike was calculated directly from experimentally measured waveforms in the relaxation-oscillator circuit composed of the Ag/freestanding-STO/Pt/PEN device and a 50 MΩ load resistor. The instantaneous device current was obtained from the measured output voltage \({V}_{{\rm{out}}}(t)\) across the load resistor as
and the energy consumed per spike (E) is obtained by integrating this power over the total spike period \([{t}_{1},{t}_{2}]\):
This analysis is based entirely on experimental data recorded by the oscilloscope and does not involve simulation.
Data availability
All data supporting the findings of this study are available within the Article and its Supplementary Information files.
References
Liu, Z. et al. A memristor-based adaptive neuromorphic decoder for brain-computer interfaces. Nat. Electron. 8, 362 (2025).
Kudithipudi, D. et al. Neuromorphic computing at scale. Nature 637, 801 (2025).
Boahen, E. K. et al. Bio-inspired neuromorphic sensory systems from intelligent perception to nervetronics. Adv. Sci. 12, 2409568 (2025).
Jiang, C. et al. Neuromorphic antennal sensory system. Nat. Commun. 15, 2109 (2024).
Kang, K. et al. Bionic artificial skin with a fully implantable wireless tactile sensory system for wound healing and restoring skin tactile function. Nat. Commun. 15, 10 (2024).
Krauhausen, I. et al. Bio-inspired multimodal learning with organic neuromorphic electronics for behavioral conditioning in robotics. Nat. Commun. 15, 4765 (2024).
Zhang, L. et al. Skin-inspired, sensory robots for electronic implants. Nat. Commun. 15, 4777 (2024).
Xia, H. et al. Shaping high-performance wearable robots for human motor and sensory reconstruction and enhancement. Nat. Commun. 15, 1760 (2024).
Ren, X., Yuan, Y., Li, J. & Hu, B. Mechanoadaptive bioelectronics for deep tissue sensing. Chin. J. Chem. 43, 567 (2025).
Proske, U., Wise, A. K. & Gregory, J. E. The role of muscle receptors in the detection of movements. Prog. Neurobiol. 60, 85 (2000).
Edin, B. B. & Abbs, J. H. Finger movement responses of cutaneous mechanoreceptors in the dorsal skin of the human hand. J. Neurophysiol. 65, 657 (1991).
Hudson, K. et al. Effects of changing skin mechanics on the differential sensitivity to surface compliance by tactile afferents in the human finger pad. J. Neurophysiol. 114, 2249 (2015).
Liu, J. Multidimensional free shape‑morphing flexible neuromorphic devices with regulation at arbitrary points. Nat. Commun. 16, 756 (2025).
Wu, X. et al. Wearable in-sensor reservoir computing using optoelectronic polymers with through-space charge-transport characteristics for multi-task learning. Nat. Commun. 14, 468 (2023).
Dang, B. et al. Reconfigurable in-sensor processing based on a multi-phototransistor-one-memristor array. Nat. Electron. 7, 991 (2024).
Zhu, J. et al. A flexible LIF neuron based on NbOx memristors for neural interface applications. Proc. IEEE EDTM. 1 (2021).
Jiang, C. et al. A flexible artificial sensory nerve enabled by nanoparticle-assembled synaptic devices for neuromorphic tactile recognition. Adv. Sci. 9, 2106124 (2022).
Yuan, R. et al. A calibratable sensory neuron based on epitaxial VO2 for spike-based neuromorphic multisensory system. Nat. Commun. 13, 3973 (2022).
Chun, S. et al. An artificial neural tactile sensing system. Nat. Electron 4, 429 (2021).
Yu, Q. et al. Highly sensitive strain sensors based on piezotronic tunneling junction. Nat. Commun. 13, 778 (2022).
Datye, I. et al. Strain-enhanced mobility of monolayer MoS₂. Nano Lett. 22, 8052 (2022).
Pattnaik, D. et al. Stress-induced artificial neuron spiking in diffusive memristors. Commun. Eng. 3, 315 (2024).
Mishra, S., Yadav, B. & Kulkarni, G. U. A strain-sensitive neuromorphic device emulating mechanoreception for different skin sensitivities. J. Mater. Chem. C. 12, 18243 (2024).
Kim, Y. et al. A bioinspired flexible organic artificial afferent nerve. Science 360, 998 (2018).
Marchal-Crespo, L. & Reinkensmeyer, D. J. Review of control strategies for robotic movement training after neurologic injury. J. Neuroeng. Rehabil. 6, 20 (2009).
Wang, C., Wang, C., Huang, Z. & Xu, S. Materials and structures toward soft electronics. Adv. Mater. 30, 1801368 (2018).
Amjadi, M., Kyung, K.-U., Park, I. & Sitti, M. Stretchable, skin-mountable, and wearable strain sensors and their potential applications: a review. Adv. Funct. Mater. 26, 1678 (2016).
Lu, D. et al. Synthesis of freestanding single-crystal perovskite films and heterostructures by etching of sacrificial water-soluble layers. Nat. Mater. 15, 1255 (2016).
Ji, D. et al. Freestanding crystalline oxide perovskites down to the monolayer limit. Nature 570, 87 (2019).
Dong, G. et al. Super-elastic ferroelectric single-crystal membrane with continuous electric dipole rotation. Science 366, 475 (2019).
Hong, S. S. et al. Extreme tensile strain states in La0.7Ca0.3MnO3 membranes. Science 368, 71 (2020).
Dai, Y. et al. Engineering of the Curie temperature of epitaxial Sr1−xBaxTiO3 films via strain. J. Appl. Phys. 120, 114101 (2016).
Kim, S.-R. & Nairn, J. A. Fracture mechanics analysis of coating/substrate systems: Part II—experiments in bending. Eng. Fract. Mech. 65, 595 (2000).
Yan, X. B. et al. The resistive switching mechanism of Ag/SrTiO₃/Pt memory cells. Electrochem. Solid-State Lett. 13, H87 (2010).
Hu, H. et al. Enhanced resistance switching in ultrathin Ag/SrTiO₃/(La, Sr)MnO₃ memristors and their long-term plasticity for neuromorphic computing. Appl. Phys. Lett. 119, 023502 (2021).
Xu, R. et al. Strain-induced room-temperature ferroelectricity in SrTiO₃ membranes. Nat. Commun. 11, 3141 (2020).
Li, M., Li, J., Chen, L.-Q., Gu, B.-L. & Duan, W. Effets of strain and oxygen vacancies on the ferroelectric and antiferrodistortive distortions in PbTiO₃/SrTiO₃ superlattice. Phys. Rev. B 92, 115435 (2015).
Zhou, Z., Chu, D. & Cazorla, C. Ab initio description of oxygen vacancies in epitaxially strained SrTiO₃ at finite temperatures. Sci. Rep. 11, 11499 (2021).
Al-Hamadany, R. et al. Impact of tensile strain on the oxygen vacancy migration in SrTiO₃: density functional theory calculations. J. Appl. Phys. 113, 224108 (2013).
van der Berg, R. A., Blom, P. W. M., Cillessen, J. F. M. & Wolf, R. M. Field-dependent permittivity in metal-semiconducting SrTiO₃ Schottky diodes. Appl. Phys. Lett. 66, 697 (1995).
Pontes, F. M., Lee, E. J. H., Leite, E. R., Longo, E. & Varela, J. A. High dielectric constant of SrTiO₃ thin films prepared by chemical process. J. Mater. Sci. 35, 4783 (2000).
Wells, R. B. & Barnes, B. Capacitor-free leaky integrator for biomimic artificial neurons. Electron. Lett. 38, 974 (2002).
Pei, Y. et al. Ultra robust negative differential resistance memristor for hardware neuron circuit implementation. Nat. Commun. 16, 48 (2025).
Wang, T. et al. A bio-inspired neuromorphic sensory system. Adv. Intell. Syst. 4, 2200047 (2022).
Li, F. et al. A skin-inspired artificial mechanoreceptor for tactile enhancement and integration. ACS Nano 15, 16422 (2021).
Zhang, X. et al. An artificial spiking afferent nerve based on Mott memristors for neurorobotics. Nat. Commun. 11, 51 (2020).
Lee, Y., Park, H.-L., Kim, Y. & Lee, T.-W. Organic electronic synapses with low energy consumption. Joule 5, 794 (2021).
Li, Z. et al. Crossmodal sensory neurons based on high-performance flexible memristors for human-machine in-sensor computing system. Nat. Commun. 15, 7275 (2024).
Fu, T. et al. Self-sustained green neuromorphic interfaces. Nat. Commun. 12, 3351 (2021).
Nath, S. K. et al. Optically tunable electrical oscillations in oxide-based memristors for neuromorphic computing. Adv. Mater. 36, 2400904 (2024).
Chen, C. et al. A photoelectric spiking neuron for visual depth perception. Adv. Mater. 34, 2201895 (2022).
Sarkar, T. et al. An organic artificial spiking neuron for in situ neuromorphic sensing and biointerfacing. Nat. Electron. 5, 774 (2022).
Johansson, R. S. & Vallbo, A. B. Tactile sensibility in the human hand. relative and absolute densities of four types of mechanoreceptive units in glabrous skin. J. Physiol. 286, 283 (1979).
Hu, X. D. et al. High-resolution, high-speed and low-cost flexible tactile sensor array system. Measurement 241, 115630 (2025).
Wang, T. et al. Reconfigurable neuromorphic memristor network for ultralow-power smart textile electronics. Nat. Commun. 13, 7432 (2022).
Acknowledgements
This work was supported by the National R&D Programs (Nos. 2022R1A2C3004135 and RS-2025-02243032) and Nano·Material Technology Development Program (No. 2021M3H4A1A03054864) through the National Research Foundation of Korea (NRF) funded by Ministry of Science and ICT, and Korea Basic Science Institute (National Research Facilities and Equipment Center) grant funded by the Ministry of Education (No. 2022R1A6C101A754).
Author information
Authors and Affiliations
Contributions
S.K. and C.Y. planned the projects and designed the experiments. S.K. performed the electrical experiments. S.K. and W.R. fabricated the sample. S.K. and J.J. performed atomic force microscopy measurements and XRD measurements, and data analysis. G.O. assisted with the analysis of electrical data. S.K. and B.H.P. supervised the research and interpreted the results, participated in discussions, and wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, S., Yoon, C., Jeon, J. et al. Mechanosensory neuron implemented by a single freestanding epitaxial SrTiO3 capacitor. npj Flex Electron 10, 18 (2026). https://doi.org/10.1038/s41528-025-00520-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41528-025-00520-6