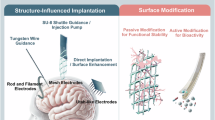
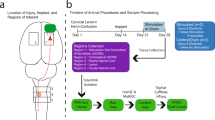
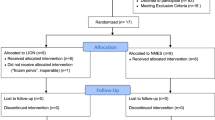
Abstract
Neural interfaces for monitoring and modulating spinal nerve activity are increasingly being designed to be flexible and stretchable to enhance their biomechanical compatibility and integration. However, excessive flexibility introduces practical limitations such as difficulty in insertion into narrow spinal spaces and long-term electrical instability, hindering real-world applications. In this study, we developed a spinal nerve interface by incorporating a liquid-metal conductor and dynamic stiffness-based variable-compliance structure, which can address the challenges of current flexible neural interface technologies. During insertion, the dynamic stiffness enhancer minimizes unintended buckling and ensures minimally invasive implantation into the intended target. The proximity of the proposed device to the spinal cord increases as it flexes automatically and rapidly in a humid environment. The liquid-metal conductor maintained stable electrical properties in freely moving rats, ensuring reliable and sustained functionality. This study lays the foundation for practical, fully implantable spinal bioelectronics designed with a focus on ease of implantation and long-term functionality.
Similar content being viewed by others
Data availability
All data are available in the main text or the supplementary materials.
References
Gupta, A., Vardalakis, N. & Wagner, F. B. Neuroprosthetics: from sensorimotor to cognitive disorders. Commun. Biol. 6, 14 (2023).
Krauss, J. K. et al. Technology of deep brain stimulation: current status and future directions. Nat. Rev. Neurol. 17, 75–87 (2021).
Isagulyan, E. et al. Spinal cord stimulation in chronic pain: technical advances. Korean J. Pain 33, 99–107 (2020).
Remacle, T. Y., Bonhomme, V. L., Renwart, H. J. P. & Remacle, J. M. Effect of multicolumn lead spinal cord stimulation on low back pain in failed back surgery patients: a three-year follow-up. Neuromodulation 20, 668–674 (2017).
Thielen, B. & Meng, E. A comparison of insertion methods for surgical placement of penetrating neural interfaces. J. Neural Eng. 18, 041003 (2021).
Kwon, Y. M. Posterior cord syndrome after spinal cord stimulation electrode lead insertion: a case report. Korean J. Neurotrauma 18, 393–398 (2022).
Minev, I. R. et al. Electronic dura mater for long-term multimodal neural interfaces. Science 347, 159–163 (2015).
Karimi, A., Shojaei, A. & Tehrani, P. Mechanical properties of the human spinal cord under the compressive loading. J. Chem. Neuroanat. 86, 15–18 (2017).
Hayek, S. M., Veizi, E. & Hanes, M. Treatment-limiting complications of percutaneous spinal cord stimulator implants: a review of eight years of experience from an academic center database. Neuromodulation 18, 603–608 (2015).
Mekhail, N. A. et al. Retrospective review of 707 cases of spinal cord stimulation: indications and complications. Pain Pract. 11, 148–153 (2011).
Greiner, N. et al. Recruitment of upper-limb motoneurons with epidural electrical stimulation of the cervical spinal cord. Nat. Commun. 12, 435 (2021).
Taccola, G., Barber, S., Horner, P. J., Bazo, H. A. C. & Sayenko, D. Complications of epidural spinal stimulation: lessons from the past and alternatives for the future. Spinal Cord 58, 1049–1059 (2020).
Rowald, A. et al. Activity-dependent spinal cord neuromodulation rapidly restores trunk and leg motor functions after complete paralysis. Nat. Med. 28, 260–271 (2022).
Nanivadekar, A. C. et al. Restoration of sensory feedback from the foot and reduction of phantom limb pain via closed-loop spinal cord stimulation. Nat. Biomed. Eng. 8, 992–1003 (2023).
Powell, M. P. et al. Epidural stimulation of the cervical spinal cord for post-stroke upper-limb paresis. Nat. Med. 29, 689–699 (2023).
Squair, J. W. et al. Neuroprosthetic baroreflex controls haemodynamics after spinal cord injury. Nature 590, 308–314 (2021).
Beliaeva, N. N. et al. Transcutaneous spinal cord stimulation attenuates blood pressure drops in orthostasis. Life 13, 26 (2023).
Petersen, E. A. Spinal cord stimulation in painful diabetic neuropathy: an overview. Diabetes Res. Clin. Pract. 206, 110760 (2023).
Petersen, E. A. et al. Long-term efficacy of high-frequency (10 kHz) spinal cord stimulation for the treatment of painful diabetic neuropathy: 24-Month results of a randomized controlled trial. Diabetes Res. Clin. Pract. 203, 110865 (2023).
Kozai, T. D. Y. et al. Mechanical failure modes of chronically implanted planar silicon-based neural probes for laminar recording. Biomaterials 37, 25–39 (2015).
Rosenauer, P., Kratzer, C., Larisegger, S. & Radl, S. Extraction of mechanical parameters via molecular dynamics simulation: application to polyimides. Polymers 16, 813 (2024).
Sanzari, I. et al. Parylene C topographic micropattern as a template for patterning PDMS and Polyacrylamide hydrogel. Sci. Rep. 7, 5764 (2017).
Cattani, M., Salvadori, M. C., Melo, L. L., Vaz, A. R. & Brown, I. G. Measurement of the elastic modulus of nanostructured gold and platinum thin films. Phys. Rev. B Condens. Matter Mater. Phys. 67, 153404 (2003).
Oldroyd, P., Gurke, J. & Malliaras, G. G. Stability of thin film neuromodulation electrodes under accelerated aging conditions. Adv. Funct. Mater. 33, 2208881 (2023).
Dong, R. et al. Printed stretchable liquid metal electrode arrays for in vivo neural recording. Small 17, 2006612 (2021).
Hong, S., Rachim, V. P., Baek, J. H. & Park, S. M. Real-time multiaxial strain mapping using computer vision integrated optical sensors. npj Flex. Electron. 7, 30 (2023).
Tavakoli, M. et al. EGaIn-assisted room-temperature sintering of silver nanoparticles for stretchable, inkjet-printed, thin-film electronics. Adv. Mater. 30, e1801852 (2018).
Zhang, X. et al. Liquid metal-based electrode array for neural signal recording. Bioengineering 10, 578 (2023).
Lee, W. et al. Universal assembly of liquid metal particles in polymers enables elastic printed circuit board. Science 378, 637–641 (2022).
Oh, N. G., Hwang, S. Y. & Na, Y. H. Fabrication of a PVA-based hydrogel microneedle patch. ACS Omega 7, 25179–25185 (2022).
Huang, S. M., Liu, S. M., Tseng, H. Y. & Chen, W. C. Effect of citric acid on swelling resistance and physicochemical properties of post-crosslinked electrospun polyvinyl alcohol fibrous membrane. Polymers 15, 1738 (2023).
Baca Ramos, K., Olivas Armendáriz, I., Chapa González, C., Álvarez, V. A. & García-Casillas, P. E. Cytotoxic effect of polyvinyl alcohol-magnetite composite. SN Appl. Sci. 5, 254 (2023).
Roh, Y. et al. Transient shuttle for a widespread neural probe with minimal perturbation. npj Flex. Electron. 8, 40 (2024).
Ma, X. et al. Capillary-force-assisted clean-stamp transfer of two-dimensional materials. Nano Lett. 17, 6961–6967 (2017).
Mariello, M. et al. Novel flexible triboelectric nanogenerator based on metallized porous PDMS and Parylene C. Energies 13, 1625 (2020).
Kim, W. et al. Highly stretchable and conductive kirigami-like double-layer electrodes for motion-insensitive wearable electronics. Compos. B Eng. 283, 111655 (2024).
Deng, J. et al. Electrical bioadhesive interface for bioelectronics. Nat. Mater. 20, 229–236 (2021).
Kroeker, S. G. & Ching, R. P. Coupling between the spinal cord and cervical vertebral column under tensile loading. J. Biomech. 46, 773–779 (2013).
Rose, T. L. & Robblee, L. S. Electrical stimulation with Pt electrodes. VIII. Electrochemically safe charge injection limits with 0.2 ms pulses (neuronal application). IEEE Trans. Biomed. Eng. 37, 1118–1120 (2002).
Kim, J. H., Kim, S., So, J. H., Kim, K. & Koo, H. J. Cytotoxicity of gallium-indium liquid metal in an aqueous environment. ACS Appl. Mater. Interfaces 10, 17448–17454 (2018).
Woodington, B. J. et al. Electronics with shape actuation for minimally invasive spinal cord stimulation. Sci. Adv. 7, 7833–7858 (2021).
Hazelgrove, B. et al. Detection of spinal action potentials with subdural electrodes in freely moving rodents. Sci. Rep. 15, 30635 (2025).
Harland, B. et al. Daily electric field treatment improves functional outcomes after thoracic contusion spinal cord injury in rats. Nat. Commun. 16, 5372 (2025).
Guo, X. J. et al. Epidural combined optical and electrical stimulation induces high-specificity activation of target muscles in spinal cord injured rats. Front. Neurosci. 17, 1282558 (2023).
Gad, P. et al. Development of a multi-electrode array for spinal cord epidural stimulation to facilitate stepping and standing after a complete spinal cord injury in adult rats. J. Neuroeng. Rehabil. 10, 2 (2013).
Castagnola, V. et al. Parylene-based flexible neural probes with PEDOT coated surface for brain stimulation and recording. Biosens. Bioelectron. 67, 450–457 (2015).
Woodington, B. J. et al. Flexible circumferential bioelectronics to enable 360-degree recording and stimulation of the spinal cord. Sci. Adv. 10, eadl1230 (2024).
Momin, M. et al. 3D-printed flexible neural probes for recordings at single-neuron level. Device 2, 100519 (2024).
Barshutina, M. N. et al. PDMS-CNT composite for soft bioelectronic neuronal implants. Compos. B Eng. 247, 110286 (2022).
Chen, Q. et al. Robust fabrication of ultra-soft tunable PDMS microcapsules as a biomimetic model for red blood cells. Soft Matter 19, 5249–5261 (2023).
Troughton, J. G., Ansong, Y. O., Duobaite, N. & Proctor, C. M. Finite element analysis of electric field distribution during direct current stimulation of the spinal cord: Implications for device design. APL Bioeng. 7, 046109 (2023).
Kang, W. et al. Closed-loop direct control of seizure focus in a rodent model of temporal lobe epilepsy via localized electric fields applied sequentially. Nat. Commun. 13, 7805 (2022).
Hukins, D. W. L., Mahomed, A. & Kukureka, S. N. Accelerated aging for testing polymeric biomaterials and medical devices. Med. Eng. Phys. 30, 1270–1274 (2008).
Ye, S. et al. SuperAnimal pretrained pose estimation models for behavioral analysis. Nat. Commun. 15, 5165 (2024).
Mathis, A. et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat. Neurosci. 21, 1281–1289 (2018).
Stringer, C., Wang, T., Michaelos, M. & Pachitariu, M. Cellpose: a generalist algorithm for cellular segmentation. Nat. Methods 18, 100–106 (2021).
Piccinini, F., Tesei, A., Arienti, C. & Bevilacqua, A. Cell counting and viability assessment of 2D and 3D cell cultures: expected reliability of the trypan blue assay. Biol. Proced. Online 19, 8 (2017).
Cho, M. et al. Neuroprosthetic closed-loop strategy for sustained blood pressure reduction via simultaneous stimulation and recording from the spinal cord. Neurotherapeutics 22, e00758 (2025)
Goldman, L. M. & Tustison, R. W. High durability infrared transparent coatings. Proc. SPIE Int Soc. Opt. Eng. 2286, 316–324 (1994).
Choi, E., Jeon, J. S. & Seo, J. Cyclic compressive behavior of polyurethane rubber springs for smart dampers. Smart Struct. Syst. 20, 739–757 (2017).
Acknowledgements
This research was supported by the National Research Foundation of Korea (NRF) under multiple programs: the Basic Science Research Program funded by the Ministry of Education (RS-2024-00412658, 2020R1A6A1A03047902); the Ministry of Science and ICT (MSIT) (RS-2025-00517742, RS-2025-17492968); the Pioneer Research Center Program funded by the Ministry of Science, ICT & Future Planning (2022M3C1A3081294). Figures 2d, and 6a, c, d, e were partially generated using BioRender resources.
Author information
Authors and Affiliations
Contributions
S.H. and S.M.P. conceived the study. S.H., M.C.1, M.K., S.L., H.K., M.C.2, H.J.M., J.P., Y.J.A., and W.K. developed the methodology. S.H., S.P., and H.K. performed the investigations. S.H. wrote the manuscript under the supervision of S.M.P. S.M.P. reviewed and edited the final version. All authors were involved in the discussion and finalization of the manuscript.* M. C.1: Mingeun Cho/M. C.2: Minhye Choo.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hong, S., Pak, S., Cho, M. et al. Unidirectional dynamic stiffness modulation enables easily insertable and conformally attachable spinal bioelectronic device. npj Flex Electron (2026). https://doi.org/10.1038/s41528-026-00557-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41528-026-00557-1