Abstract
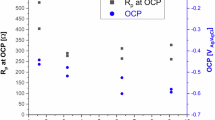
The corrosion products of steel overpacks for high-level radioactive waste disposal significantly affect their cathodic depolarization behavior during corrosion and may even alter the depolarization mechanism. In this study, the anodic polarization products of NiCu low alloy steel were prepared by potentiostatic polarization in compacted bentonite with 20% and 40% water contents. Combined with the chemical composition, structure of corrosion products, and corrosion kinetic parameters, the influence mechanism of different anodic polarization products on the corrosion behavior of steel at open circuit potential was revealed. The results showed that the products formed under weak anodic polarization potentials were mainly Fe6(OH)12CO3 and Fe6(OH)12SO4. With rising polarization potential, Fe3O4, α-FeOOH, and Fe2O3·H2O emerged with increasing relative contents. Meanwhile, the residual Fe3C also accumulated in products. At open circuit potential, α-FeOOH, Fe2O3·H2O and Fe3C from strong polarization showed stronger cathodic depolarization and micro-galvanic effects than weak polarization-derived green rust, inducing a sharp drop in the cathodic and anodic polarization resistances of steel. Additionally, products formed in the 20% water content system at weak polarization resulted in lower polarization resistances than the 40% system, whereas the opposite trend occurred under strong polarization. The former was due to more abundant and easily diffused oxygen in unsaturated bentonite’s capillary pores, which oxidized green rust to α-FeOOH/Fe2O3·H2O and enhanced cathodic depolarization capacity. The latter was attributed to the loose and porous corrosion products formed under strong polarization potential when the water was sufficient, which decreased the inhibition on steel anodic dissolution.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article, and any additional data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kaufhold, S., Hassel, A. W., Sanders, D. & Dohrmann, R. Corrosion of high-level radioactive waste iron-canisters in contact with bentonite. J. Hazard. Mater. 285, 464–473 (2015).
King, F. Container materials for the storage and disposal of nuclear waste. Corrosion 69, 986–1011 (2013).
Smart, N. R., Reddy, B., Rance, A. P., Nixon, D. J. & Diomidis, N. The anaerobic corrosion of carbon steel in saturated compacted bentonite in the Swiss repository concept. Corros. Eng. Sci. Technol. 52, 113–126 (2017).
Wang, J. High-level radioactive waste disposal in China: update 2010. J. Rock. Mech. Geotech. Eng. 2, 1–11 (2010).
Japan Nuclear Cycle Development Institute. Repository Design and Engineering Technology. Japan Nuclear Cycle Development Institute, (2000).
Martin, F. A., Bataillon, C. & Schlegel, M. L. Corrosion of iron and low alloyed steel within a water saturated brick of clay under anaerobic deep geological disposal conditions: An integrated experiment. J. Nucl. Mater. 379, 80–90 (2008).
Féron, D., Crusset, D. & Gras, J. M. Corrosion issues in the french high-level nuclear waste program. Corrosion 65, 213–223 (2009).
Liu, L. et al. Effect of moisture on corrosion behavior of Q235 steel in bentonite clay. Int. J. Electrochem. Sci. 18, 100164 (2023).
Kale, R. C. & Ravi, K. Impact of iron corrosion on swell pressure of compacted barmer bentonite induced with thermal history. Prog. Nucl. Energy 162, 104787 (2023).
Kim, G. Y., Jang, J., Lee, M., Kong, M. & Yoon, S. Corrosion behaviors of SS316L, Ti-Gr.2, Alloy 22 and Cu in KURT groundwater solutions for geological deep disposal. Nucl. Eng. Technol. 54, 4474–4480 (2022).
Sun, Y. et al. Understanding the role of alloyed Ni and Cu on improving corrosion resistance of low alloy steel in the simulated Beishan groundwater. J. Mater. Sci. Technol. 130, 124–135 (2022).
Lu, Y., Yang, J., Dong, J. & Ke, W. The corrosion behaviour of NiCu low-alloy steel in a deaerated bicarbonate solution containing Cl- ions. Acta Metall. Sin. 51, 440–448 (2015).
Zhang, T. et al. Effects of the addition of Cu and Ni on the Corrosion Behavior Of Weathering Steels In Corrosive Industrial Environments. J. Mater. Eng. Perform. 29, 2531–2541 (2020).
Chen, X., Dong, J., Han, E. & Ke, W. The influence of Ni alloying on corrosion behaviour of low alloy steels under wet/dry cyclic conditions. Can. Metall. Q. 46, 195–206 (2007).
Refait, P., Drissi, H. & Marie, Y. The substitution of Fe2+ ions by Ni2+ ions in green rust one compounds. Chem. Struct. Bonding 90, 389–394 (1994).
Wu, W., Dai, Z., Liu, Z., Liu, C. & Li, X. Synergy of Cu and Sb to enhance the resistance of 3%Ni weathering steel to marine atmospheric corrosion. Corros. Sci. 183, 109353 (2021).
Sun, Y. et al. Effects of iron corrosion products on the degradation of bentonite structure and properties. npj Mater. Degrad. 7, 1–11 (2023).
Refait, P. et al. Electrochemical formation of carbonated corrosion products on carbon steel in deaerated solutions. Electrochim. Acta 79, 210–217 (2012).
Lee, C. T., Qin, Z., Odziemkowski, M. & Shoesmith, D. W. The influence of groundwater anions on the impedance behaviour of carbon steel corroding under anoxic conditions. Electrochim. Acta 51, 1558–1568 (2006).
Lu, C., Samper, J., Fritz, B., Clement, A. & Montenegro, L. Interactions of corrosion products and bentonite: An extended multicomponent reactive transport model. Phys. Chem. Earth A/B/C. 36, 1661–1668 (2011).
Refait, P., Benali, O., Abdelmoula, M. & Génin, J. M. R. Formation of ‘ferric green rust’ and/or ferrihydrite by fast oxidation of iron(II-III) hydroxychloride green rust. Corros. Sci. 45, 2435–2449 (2003).
Xue, F. et al. Effect of residual dissolved oxygen on the corrosion behavior of low carbon steel in 0.1M NaHCO3 solution. J. Mater. Sci. Technol. 34, 1349–1358 (2018).
Wei, X. et al. Study on corrosion behavior of low carbon steel under different water conditions in bentonite of China-Mock-Up. Appl. Clay Sci. 167, 23–32 (2019).
Wei, X. et al. Influence of Deteriorated Bentonite Sediments on the Corrosion Behavior of NiCu Low Alloy Steel. Acta Metall. Sin. (Engl. Lett.) 35, 1011–1022 (2022).
Honda, A. et al. Effect of compacted bentonite on the corrosion behavior of Carbon steel as geological isolation overpack material. MRS Proc. 212, 287 (1990).
Eliyan, F. F. & Alfantazi, A. Mechanisms of corrosion and electrochemical significance of metallurgy and environment with corrosion of iron and steel in bicarbonate and carbonate solutions - A review. Corrosion. 70, 880–898 (2014).
Dhakal, M. et al. Effects of water content on the corrosion behavior of NiCu low alloy steel embedded in compacted GMZ bentonite. J. Mater. Sci. Technol. 222, 94–110 (2025).
Wei, X. et al. Effects of bentonite content on the corrosion evolution of low carbon steel in simulated geological disposal environment. J. Mater. Sci. Technol. 66, 46–56 (2021).
Kiczka, M. et al. Applied Geochemistry Reactive transport modelling of iron bentonite interaction in the FEBEX in situ experiment. Appl. Geochem. 170, 106057 (2024).
Smart, N. R. The corrosion behavior of carbon steel radioactive waste packages: a summary review Of Swedish And U.K. Research. In CORROSION; NACE-P3013 (2008).
Shibata, T., Watanabe, M., Taniguchi, N. & Shimizu, A. Modelling of carbon steel corrosion under oxygen depleted environment. Corros. Eng. Sci. Technol. 49, 435–441 (2014).
He, X. & Ahn, T. Carbon Steel Corrosion in Anoxic Alkaline Solution. In CORROSION; NACE-2015-5571 (2015).
Lu, Y., Dong, J. & Ke, W. Corrosion evolution of low alloy steel in deaerated bicarbonate solutions. J. Mater. Sci. Technol. 31, 1047–1058 (2015).
Ji, Y. S., Zhan, G., Tan, Z., Hu, Y. & Gao, F. Process control of reinforcement corrosion in concrete. Part 1: Effect of corrosion products. Constr. Build. Mater. 79, 214–222 (2015).
Legrand, L. et al. Electroanalytical and Kinetic Investigations on the Carbonate Green Rust-Fe(III) Redox System. J. Electrochem. Soc. 150, B45 (2003).
Olowe, A. A. & Génin, J. M. R. The influence of concentration on the oxidation of ferrous hydroxide in acidic sulphated aqueous medium: Particle size analysis of lepidocrocite. Corros. Sci. 32, 1021–1028 (1991).
Refait, P., Abdelmoula, M. & Genin, J.-M. R. Mechanisms of formation and structure of green rust one in aqueous corrosion of Iron in the presence of chloride ions. Corros. Sci. 40, 436–449 (1998).
Dong, J., Nishimura, T. & Kodama, T. Corrosion Behavior of Carbon Steel in Bicarbonate (HCO 3) Solutions. MRS Proc. 713, JJ11.8 (2002).
De Windt, L., Marsal, F., Corvisier, J. Ô & Pellegrini, D. Modeling of oxygen gas diffusion and consumption during the oxic transient in a disposal cell of radioactive waste. Appl. Geochem. 41, 115–127 (2014).
Smart, N. R., Rance, A. P. & Werme, L. O. Anaerobic Corrosion of Steel in Bentonite. MRS Proc. 807, 441 (2003).
Bonnel, A. et al. Corrosion Study of a Carbon Steel in Neutral Chloride Solutions by Impedance Techniques. J. Electrochem. Soc. 130, 753–761 (1983).
Génin, J. M. R., Refait, P. H. & Abdelmoula, M. Green rusts and their relationship to iron corrosion; a key role in microbially influenced corrosion. Hyperfine Interact. 139–140, 119–131 (2002).
Refait, P. & Génin, J. M. R. The transformation of chloride-containing green rust one into sulphated green rust two by oxidation in mixed Cl- and SO42- aqueous media. Corros. Sci. 36, 55–65 (1994).
Refait, P. et al. Electrochemical formation of green rusts in deaerated seawater-like solutions. Electrochim. Acta 56, 6481–6488 (2011).
Hao, X., Dong, J., Etim, I. I. N., Wei, J. & Ke, W. Sustained effect of remaining cementite on the corrosion behavior of ferrite-pearlite steel under the simulated bottom plate environment of cargo oil tank. Corros. Sci. 110, 296–304 (2016).
Videm, K., Kvarekval, J., Perez, T. E. & Fitzsimons, G. Surface Effects on the Electrochemistry of Iron and Carbon Steel Electrodes in Aqueous CO2 Solutions. In CORROSION 96; NACE-96001 (1996).
Liu, H. et al. The synergy between cementite spheroidization and Cu alloying on the corrosion resistance of ferrite-pearlite steel in acidic chloride solution. J. Mater. Sci. Technol. 84, 65–75 (2021).
Leupin, O. X. et al. Anaerobic corrosion of carbon steel in bentonite: An evolving interface. Corros. Sci. 187, 109523 (2021).
Wersin, P. et al. Unravelling the corrosion processes at steel/bentonite interfaces in in situ tests. Mater. Corros. 74, 1716–1727 (2023).
Hendi, R. et al. Effect of black clay soil moisture on the electrochemical behavior of API X70 pipeline steel. Mater. Res. Express 5, (2018).
Zhang, Q. et al. Hydrogen permeation behavior of carbon steel during corrosion in highly pressed saturated bentonite. Front. Mater. 9, 1–11 (2022).
King, F. Nuclear Waste Canister Materials: Corrosion Behavior and Long-Term Performance in Geological Repository Systems. In Geological Repository Systems for Safe Disposal of Spent Nuclear Fuels and Radioactive Waste (Woodhead Publishing, 2017).
Villar, M. V. & Lloret, A. Influence of temperature on the hydro-mechanical behaviour of a compacted bentonite. 26, 337–350 (2004).
Bradbury, M. H. & Baeyens, B. Porewater chemistry in compacted re-saturated MX-80 bentonite. 61, 329–338 (2003).
Xue, F., Wei, X., Dong, J., Wang, C. & Ke, W. Effect of chloride ion on corrosion behavior of low carbon steel in 0.1 M NaHCO3 solution with different dissolved oxygen concentrations. J. Mater. Sci. Technol. 35, 596–603 (2019).
Shinagawa, T., Kanemoto, Y. & Ohtaka, A. Oriented α-Fe2O3 and Fe3O4 nanoporous films obtained by topotactic-like pseudomorphic transformation of γ- and δ-FeOOH films. J. Mater. Chem. C. 12, 9957–9967 (2024).
Lair, V., Antony, H., Legrand, L. & Chaussé, A. Electrochemical reduction of ferric corrosion products and evaluation of galvanic coupling with iron. Corros. Sci. 48, 2050–2063 (2006).
Kaufhold, S., Dohrmann, R., Ufer, K. & Meyer, F. M. Comparison of methods for the quantification of montmorillonite in bentonites. Appl. Clay Sci. 22, 145–151 (2002).
Rossi, E. et al. Analysis of naturally-generated corrosion products due to chlorides in 20-year old reinforced concrete: An elastic modulus-mineralogy characterization. Corros. Sci. 184, (2021).
Cambier, S. M., Verreault, D. & Frankel, G. S. Raman investigation of anodic undermining of coated steel during environmental exposure. Corrosion 70, 1219–1229 (2014).
Refait, P., Girault, P., Jeannin, M. & Rose, J. Influence of arsenate species on the formation of Fe(III) oxyhydroxides and Fe(II-III) hydroxychloride. Colloids Surfaces A Physicochem. Eng. Asp. 332, 26–35 (2009).
Azoulay, I., Conforto, E., Refait, P. & Rémazeilles, C. Study of ferrous corrosion products on iron archaeological objects by electron backscattered diffraction (EBSD). Appl. Phys. A Mater. Sci. Process. 110, 379–388 (2013).
De Faria, D. L. A., Venâncio Silva, S. & De Oliveira, M. T. Raman microspectroscopy of some iron oxides and oxyhydroxides. J. Raman Spectrosc. 28, 873–878 (1997).
Das, S. & Hendry, M. J. Application of Raman spectroscopy to identify iron minerals commonly found in mine wastes. Chem. Geol. 290, 101–108 (2011).
Wang, Y. et al. Understanding the role of alloyed Cu and P in the initial rust composition of weathering steel formed in a simulated coastal-industrial atmosphere. Corros. Sci. 193, 109912 (2021).
Freeman, J. J., Wang, A., Kuebler, K. E., Jolliff, B. L. & Haskin, L. A. Characterization of natural feldspars by raman spectroscopy for future planetary exploration. Can. Mineral. 46, 1477–1500 (2008).
Mernagh, T. P. Use of the laser Raman microprobe for discrimination amongst feldspar minerals. J. Raman Spectrosc. 22, 453–457 (1991).
Mullet, M., Khare, V. & Ruby, C. XPS study of Fe(II)-Fe(III) (oxy)hydroxycarbonate green rust compounds. Surf. Interface Anal. 40, 323–328 (2008).
Wagner, C. D. et al. NIST standard reference database 20, Version 3.4 (Web version). Natl. Inst. Stand. Technol. Gaithersburg, MD 20899, (2003).
Biesinger, M. C., Payne, B. P., Lau, L. W. M., Gerson, A. & Smart, R. S. C. X-ray photoelectron spectroscopic chemical state Quantification of mixed nickel metal, oxide and hydroxide systems. Surf. Interface Anal. 41, 324–332 (2009).
Kosec, T., Qin, Z., Chen, J., Legat, A. & Shoesmith, D. W. Copper corrosion in bentonite/saline groundwater solution: Effects of solution and bentonite chemistry. Corros. Sci. 90, 248–258 (2015).
Wu, W., Cheng, X., Zhao, J. & Li, X. Benefit of the corrosion product film formed on a new weathering steel containing 3% nickel under marine atmosphere in Maldives. Corros. Sci. 165, (2020).
Biesinger, M. C. et al. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and. Ni. Appl. Surf. Sci. 257, 2717–2730 (2011).
Kloprogge, J. T. & Wood, B. J. Handbook of Mineral Spectroscopy: Volume 1: X-Ray Photoelectron Spectra. (Elsevier, 2020).
Smith, M. et al. Reaction mechanisms and diagnostic mineralogy of intertidal steel corrosion: An X-ray photoelectron spectroscopy study. Corros. Commun. 13, 68–82 (2024).
Grosvenor, A. P., Kobe, B. A., Biesinger, M. C. & McIntyre, N. S. Investigation of multiplet splitting of Fe 2p XPS spectra and bonding in iron compounds. Surf. Interface Anal. 36, 1564–1574 (2004).
Murase, Y., Masuda, H. & Katayama, H. Crystallographic orientation and microstructure dependences of surface potential for annealed S45C Steel Using Combined Scanning Kelvin Force Probe Microscopy and Electron Backscatter Diffraction Analyses. Mater. Trans. 61, 482–489 (2020).
Wei, X., Dong, J. & Ke, W. Progress on a corrosion study of low carbon steel for HLW container in a simulated geological disposal environment in China. Corros. Commun. 1, 10–17 (2021).
Power, G. P. & Ritchie, I. M. Mixed potential measurements in the elucidation of corrosion mechanisms- 1. Introductory theory. Electrochim. Acta. 26, 1073–1078 (1981).
Wei, X. et al. Effects of Aerobic and Anoxic Conditions on the Corrosion Behavior of NiCu Low Alloy Steel in the Simulated Groundwater Solutions. Acta Metall. Sin. (Engl. Lett.) 36, 745–757 (2023).
Zheng, M. et al. Determination of representative ground-water for corrosion assessment of candidate materials used in Beishan Area preselected for high-level radioactive waste disposal repository. J. Chin. Soc. Corros. Prot. 36, 185–190 (2016).
Chen, L. et al. Investigation of the thermal-hydro-mechanical (THM) behavior of GMZ bentonite in the China-Mock-up test. Eng. Geol. 172, 57–68 (2014).
Pourbaix, M. Atlas of electrochemical equilibria in aqueous solutions. NACE Int. 312, (1974).
Pourbaix, M. Lectures on Electrochemical Corrosion. (Springer Science & Business Media, 2012).
Liu, M., Zeng, Y. & Luo, J. L. Roles of major alloying elements in steels and alloys on corrosion under biomass hydrothermal liquefaction (HTL) conversion. Corros. Sci. 218, 111148 (2023).
Wagman, D. D. The NBS tables of chemical thermodynamic properties. Selected values for inorganic and C1 and C2 organic substances in SI units. J. Phys. Chem. Ref. Data 11, (1982).
Génin, J. M. R. et al. Thermodynamic equilibria in aqueous suspensions of synthetic and natural Fe(II)-Fe(III) green rusts: Occurrences of the mineral in hydromorphic soils. Environ. Sci. Technol. 32, 1058–1068 (1998).
ASTM International Conducting Potentiodynamic Polarization Resistance Measurements. Astm G. 59 97, 1–4 (2003).
ASTM G1-03. ASTM G1 - 03 Standard practice for preparing, cleaning, and evaluating corrosion test specimens. ASTM Spec. Tech. Publ. 505–510 (2003).
Moudler, J. F., Stickle, W. F., Sobol, P. E. & Bomben, K. D. Handbook of X-ray photoelectron spectroscopy. Perkin-Elmer, Eden Prairie, MN 52, (1992).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 52173304 and U1867216), and China Scholarship Council (No. 2018SLJ014508).
Author information
Authors and Affiliations
Contributions
M.D. conducted the investigation, performed the methodology and data analysis, handled the software, wrote the original draft of the manuscript; H.B.O., N.C., and Q.R. participated in the sample preparation, investigation, software handling, and data analysis; X.G contributed in the formal analysis and software; X.W. and J.D. conceived the project, provided supervision and validation, reviewed and edited the manuscript, and provided funding acquisition; W.K. contributed in visualization. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dhakal, M., Wei, X., Oli, H.B. et al. Influence of anodic polarization products of NiCu low alloy steel in compacted bentonite on its corrosion behavior. npj Mater Degrad (2026). https://doi.org/10.1038/s41529-026-00782-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41529-026-00782-z