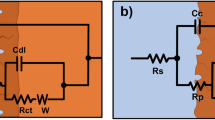
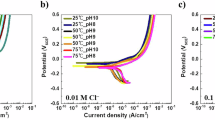
Abstract
Hydrogen atom generation and permeation on nuclear waste storage canister surfaces threaten disposal safety. This study investigated the corrosion behavior of hydrogen-charged and non-charged copper in an anoxic, sulfide-containing groundwater environment simulating nuclear waste disposal conditions in Beishan, China. Chloride ions induced pitting corrosion in non-charged copper. The mass transfer rate of \(\text{Cu}{\text{Cl}}_{2}^{-}\) species within pitting pores was higher compared to that of Cu⁺ through the Cu2S layer, resulting in excessive thickening of corrosion products at pitting sites. By contrast, porous corrosion products on hydrogen-charged copper formed non-passivating films, leading to uniform substrate corrosion. The corrosion product thickness exhibited sub-parabolic growth over time. This predictable sub-parabolic growth pattern is crucial for reliable service life assessment of deep geological repositories rather than unpredictable pitting corrosion.
Similar content being viewed by others
Data availability
The authors declare that the data related to the findings of this study are available within the paper and any other supporting data could be provided by the corresponding author on reasonable request.
References
NWMO. Postclosure Safety Assessment of a Used Fuel Repository in Crystalline Rock, NWMO-TR-2017-02 (Nuclear Waste Management Organization, Toronto, Canada, 2017).
King, F. Copper Corrosion Under Expected Conditions in a Deep Geologic Repository, Report TR-01-23, (SKB, Stockholm, Sweden, 2001).
Hall, D. S. & Keech, P. G. An overview of the Canadian corrosion program for the long-term management of nuclear waste. Corros. Eng. Sci. Techn. 52, 2–5 (2017).
Chen, J., Qin, Z. & Shoesmith, D. W. Long-term corrosion of copper in a dilute anaerobic sulfide solution. Electrochim. Acta 56, 7854–7861 (2011).
Damien, F. K. & Druyts, B.F. (eds.) Sulphur-Assisted Corrosion in Nuclear Disposal Systems (CRC Press, 2020. https://doi.org/10.1201/9781003059448.
Chen, J., Qin, Z. & Shoesmith, D. W. Key parameters determining structure and properties of sulphide films formed on copper corroding in anoxic sulphide solutions. Corros. Eng. Sci. Techn. 49, 415–419 (2014).
Guo, M. et al. The susceptibility of copper to pitting corrosion in borate-buffered aqueous solutions containing chloride and sulfide. J. Electrochem. Soc. 166, C550 (2019).
Chen, J., Qin, Z., Martino, T. & Shoesmith, D. W. Non-uniform film growth and micro/macro-galvanic corrosion of copper in aqueous sulphide solutions containing chloride. Corros. Sci. 114, 72–78 (2017).
Chen, J., Qin, Z. & Shoesmith, D. W. Kinetics of corrosion film growth on copper in neutral chloride solutions containing small concentrations of sulfide. J. Electrochem. Soc. 157, C338 (2010).
Chen, J., Qin, Z. & Shoesmith, D. W. Copper corrosion in aqueous sulfide solutions under nuclear waste repository conditions. MRS Proc 1475, 465–470 (2012).
Martino, T., Partovi-Nia, R., Chen, J., Qin, Z. & Shoesmith, D. W. Mechanisms of film growth on copper in aqueous solutions containing sulphide and chloride under voltammetric conditions. Electrochim. Acta 127, 439–447 (2014).
Mao, F., Dong, C., Sharifi-Asl, S., Lu, P. & Macdonald, D. D. Passivity breakdown on copper: influence of chloride Ion. Electrochim. Acta 144, 391–399 (2014).
Dong, C. et al. Passivity breakdown on copper: influence of temperature. J. Electrochem. Soc. 163, C707 (2016).
Kong, D. et al. Size matching effect between anion vacancies and halide ions in passive film breakdown on copper. Electrochim. Acta 292, 817–827 (2018).
Sandström, R. The role of hydrogen in copper. SKB. https://skb.se/wp-content/uploads/2015/05/1420051-PM-The-role-of-hydrogen-in-copper.pdf (2014).
Ruiz-Fresneda, M. A. et al. Impact of microbial processes on the safety of deep geological repositories for radioactive waste. Front. Microbiol. 14, 1134078 (2023).
Hall, D.S., Behazin, M., Jeffrey Binns, W. & Keech, P. G. An evaluation of corrosion processes affecting copper-coated nuclear waste containers in a deep geological repository. Prog. Mater Sci. 118, 100766 (2021).
Beaton, D., Pelletier, P. & Goulet, R. R. Microbial degradation of cellulosic material and gas generation: implications for the management of low- and intermediate-level radioactive waste. Front. Microbiol. 10, 204 (2019).
NWMO. Postclosure Safety Assessment of a Used Fuel Repository in Crystalline Rock; NWMO-TR-2018-08 (Nuclear Waste Management Organization, Toronto, Canada, 2018).
Svensk Kärnbränslehantering AB. Supplementary information on Canister Integrity Issues, SKB TR-19-15 (SKB, 2019).
Liu, X. et al. The influence of hydrogen permeation on the protection performance of the Cu coating of nuclear waste containers. Corros. Sci. 221, 111314 (2023).
Chen, J., Qin, Z., Wu, L., Noël, J. J. & Shoesmith, D. W. The influence of sulphide transport on the growth and properties of copper sulphide films on copper. Corros. Sci. 87, 233–238 (2014).
Rickard, D. & Luther, G. W. I. Chemistry of iron sulfides. ChemInform 38, https://doi.org/10.1002/chin.200719200 (2007).
Meyer, B., Ward, K., Koshlap, K. & Peter, L. Second dissociation constant of hydrogen sulfide. Inorg. Chem. 22, 2345–2346 (1983).
Kamyshny, A., Goifman, A., Gun, J., Rizkov, D. & Lev, O. Equilibrium distribution of polysulfide ions in aqueous solutions at 25 degrees C: a new approach for the study of polysulfides’ equilibria. Environ. Sci. Technol. 38, 6633–6644 (2004).
Cobble, J. W. & Stephens, H. P. Thermodynamic properties of the aqueous sulfide and bisulfide ions and the second ionization constant of hydrogen sulfide over extended temperatures. Inorg. Chem. 10, 619–625 (1971).
Gennero De Chialvo, M. R. & Arvia, A. J. The electrochemical behaviour of copper in alkaline solutions containing sodium sulphide. J. Appl. Electrochem. 15, 685–696 (1985).
Licht, S. & Manassen, J. The second dissociation constant of H2S. J. Electrochem. Soc. 134, 918 (1987).
Biesinger, M. C. Advanced analysis of copper X-ray photoelectron spectra. Surf. Interface Anal. 49, 1325–1334 (2017).
Trotochaud, L. et al. Water adsorption and dissociation on polycrystalline copper oxides: Effects of environmental contamination and experimental protocol. J. Phys. Chem. B 122, 1000–1008 (2018).
Deng, X., Herranz, T., Weis, C., Bluhm, H. & Salmeron, M. Adsorption of water on Cu2O and Al2O3 thin Films. J. Phys. Chem. C 112, 9668–9672 (2008).
Yao, H. B., Li, Y. & Wee, A. T. S. An XPS investigation of the oxidation/corrosion of melt-spun Mg. Appl. Surf. Sci. 158, 112–119 (2000).
Santamaria, M., Di Quarto, F., Zanna, S. & Marcus, P. Initial surface film on magnesium metal: A characterization by X-ray photoelectron spectroscopy (XPS) and photocurrent spectroscopy (PCS). Electrochim. Acta 53, 1314–1324 (2007).
Saikova, S. et al. X-ray photoelectron, Cu L3MM Auger and X-ray absorption spectroscopic studies of Cu nanoparticles produced in aqueous solutions: the effect of sample preparation techniques. Appl. Surf. Sci. 258, 8214–8221 (2012).
Déroubaix, G. & Marcus, P. X-ray photoelectron spectroscopy analysis of copper and zinc oxides and sulphides. Surf. Interface Anal. 18, 39–46 (1992).
Poulston, S., Parlett, P. M., Stone, P. & Bowker, M. Surface oxidation and reduction of CuO and Cu2O Studied using XPS and XAES. Surf. Interface Anal. 24, 811–820 (1996).
Sharifi-Asl, S. Corrosion Issues in High-level Nuclear Waste Containers, Ph. D, The Pennsylvania State University, https://etda.libraries.psu.edu/catalog/18973 (2013).
Bae, S.-E., Stewart, K. L. & Gewirth, A. A. Nitrate Adsorption and Reduction on Cu(100) in Acidic Solution. J. Am. Chem. Soc. 129, 10171–10180 (2007).
Prince, N. P. et al. A SEXAFS and X-ray standing wave study of the Cu(111)(√7 × √7)R19°-S surface: Adsorbate-substrate and adsorbate-adsorbate registry. Surf. Sci. 230, 13–26 (1990).
Suggs, D. W. & Bard, A. J. Scanning Tunneling Microscopic Study with Atomic Resolution of the Dissolution of Cu(111) in Aqueous Chloride Solutions. J. Am. Chem. Soc. 116, 10725–10733 (1994).
Chen, J., Qin, Z. & Shoesmith, D. W. Rate controlling reactions for copper corrosion in anaerobic aqueous sulphide solutions. Corros. Eng. Sci. Techn. 46, 138–141 (2011).
Smith, J.M. The Corrosion and Electrochemistry of Copper in Aqueous, Anoxic Sulphide Solutions, Ph. D, (The University of Western Ontario (Canada), 2008).
Mountain, B. W. & Seward, T. M. The hydrosulphide/sulphide complexes of copper(I): experimental determination of stoichiometry and stability at 22°C and reassessment of high temperature data. Geochim. Cosmochim. Acta 63, 11–29 (1999).
Chen, J., Qin, Z. & Shoesmith, D. W. The mechanism of sulphide film growth on copper in anaerobic sulphide solutions under natural corrosion conditions. Innov. Corrosion Mater. Sci. 8, 108–112 (2018).
Tinter, U. & Wiemhöfer, H.-D. Chemical diffusion coefficients of the low temperature phases of CuxSe and CuxS - investigations with point electrodes. Solid State Ionics 9–10, 1213–1220 (1983).
Sanchez, S., Cassaignon, S., Vedel, J. & Meier, H. G. Copper diffusion in solid copper sulfide electrode. Electrochim. Acta 41, 1331–1339 (1996).
Cassaignon, S., Pauporté, T. H., Guillemoles, J.-F. & Vedel, J. Copper diffusion in copper sulfide: a systematic study. Ionics 4, 364–371 (1998).
Małyszko, J. & Scendo, M. Electrode kinetics of the Cu(II)/Cu(I) system at platinum in water + dimethylsulphoxide (DMSO) mixtures. J. Electroanal. Chem. 250, 61–72 (1988).
Khairy, E. M. & Darwish, N. A. Studies on copper-semiconducting layer-electrolyte systems—I. Electrode potentials of copper and electrodeposited copper sulphide in S2− and Cu2+ media. Corros. Sci. 13, 141–147 (1973).
Brug, G. J., van den Eeden, A. L. G., Sluyters-Rehbach, M. & Sluyters, J. H. The analysis of electrode impedances complicated by the presence of a constant phase element. J. Electroanal. Chem. Interfacial Electrochem. 176, 275–295 (1984).
Wang, Y. et al. In-situ monitoring of pH and Cl− concentration and rebar corrosion at the rebar/mortar interface. Constr. Build. Mater. 400, 132712 (2023).
Wang, Y. et al. Interfacial properties determine the performance of embedded IrOx pH sensor in cement mortar. J. Mater. Res. Technol. 36, 5211–5221 (2025).
Hsu, C. H. & Mansfeld, F. Technical note: concerning the conversion of the constant phase element parameter Y0 into a capacitance. Corrosion 57, 747–748 (2001).
Williams, D. C. et al. Hydrogen embrittlement and strain rate sensitivity of electrodeposited copper: part I – the effect of hydrogen content. Npj Mater. Degrad. 8, 79 (2024).
Nakahara, S. & Okinaka, Y. Defects induced in copper by cathodic charging of hydrogen. J. Electrochem. Soc. 136, 1892 (1989).
Lazanas, A. C. & Prodromidis, M. I. Electrochemical impedance spectroscopy─a tutorial. ACS Meas. Sci. Au 3, 162–193 (2023).
Bisquert, J. Influence of the boundaries in the impedance of porous film electrodes. Phys. Chem. Chem. Phys. 2, 4185–4192 (2000).
Franceschetti, D. R. Small-signal A.C. response theory for systems exhibiting impurity oxygen transport. Solid State Ionics 5, 613–616 (1981).
Diard, J.-P., Le Gorrec, B. & Montella, C. EIS study of electrochemical battery discharge on constant load. J. Power Sources 70, 78–84 (1998).
GB/T 5231-2022, Designation and chemical composition of wrought copper and copper alloys, Standardization Administration of the P.R.C. (2022).
Zheng, M. et al. Determination of representative ground-water for corrosion assessment of candidate materials used in beishan area preselected for high-level radioactive waste disposal repository. J. Chin. Soc. Corros. Prot. 36, 185–190 (2016).
Acknowledgements
This study was funded by the National Natural Science Foundation of China (No. 52201092, 51901222), Shandong Provincial Natural Science Foundation (No. ZR2022QB128) and the CAS Pioneer Hundred Talents Program.
Author information
Authors and Affiliations
Contributions
Liu X.J. Methodology, investigation, resources, formal analysis, writing – original draft, and funding acquisition. Liu N.Z. Conceptualization, formal analysis, writing – review & editing, project administration and funding acquisition. Ma Z. writing – review & editing. Wang Y.X. and Dai Y.C. investigation. Li C.T., Song L.J., Jiang Q.T. and Duan J.Z. writing – review & editing. Hou B.R. supervision and funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, X., Liu, N., Ma, Z. et al. Uniform corrosion of hydrogen-charged copper and localized corrosion of non-charged copper in anoxic groundwater containing diluted sulfide. npj Mater Degrad (2026). https://doi.org/10.1038/s41529-026-00787-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41529-026-00787-8