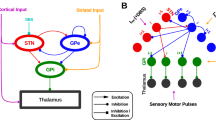
Abstract
Parkinson’s disease (PD) is a movement disorder characterized by alpha-synuclein (a-Syn) aggregation, dopaminergic degeneration, and pathological beta oscillations (13–30 Hz) in the basal ganglia circuit. Deep brain stimulation (DBS) is an effective neurosurgical treatment for the motor symptoms of PD. However, the extent to which mitigation of beta oscillations mediates DBS therapeutic effects remains uncertain. Using an adeno-associated virus-mediated nigral A53T a-Syn overexpression rat model, we examined basal ganglia-thalamo-cortical electrophysiology and the model’s responsiveness to DBS. In vivo recordings revealed early beta emergence in the motor cortex (MCx), spreading to the subthalamic nucleus (STN) and entopeduncular nucleus (EP) with neurodegeneration. This was accompanied by alterations in STN and EP single-unit activity. Awake-state beta oscillations manifested as transient bursts. Low- and high-frequency DBS differentially modulated beta bursts and motor performance. Our results demonstrate that the A53T a-Syn model replicates key PD-like electrophysiological features, providing a platform to investigate DBS mechanisms and optimize therapies targeting aberrant beta activity.
Similar content being viewed by others
Data availability
All data generated or analyzed in this study are included in this article and supplementary files, or are available from the corresponding author upon reasonable request. The underlying code for this study is not publicly available but may be made available to qualified researchers on reasonable request from the corresponding author.
Code availability
The underlying code for this study is not publicly available but may be made available to qualified researchers on reasonable request from the corresponding author.
References
Ben-Shlomo, Y. et al. The epidemiology of Parkinson’s disease. Lancet 403, 283–292 (2024).
Dorsey, E. R. et al. Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology 68, 384–386 (2007).
Lee, L.-H. N. et al. An electrophysiological perspective on Parkinson’s disease: symptomatic pathogenesis and therapeutic approaches. J. Biomed. Sci. 28, 85 (2021).
DeLong, M. R. & Wichmann, T. Circuits and circuit disorders of the basal ganglia. Arch. Neurol. 64, 20 (2007).
Little, S. & Brown, P. The functional role of beta oscillations in Parkinson’s disease. Parkinsonism Relat. Disord. 20, S44–S48 (2014).
Brown, P. Oscillatory nature of human basal ganglia activity: Relationship to the pathophysiology of Parkinson’s disease. Mov. Disord. 18, 357–363 (2003).
Feldmann, L. K. et al. Toward therapeutic electrophysiology: beta-band suppression as a biomarker in chronic local field potential recordings. NPJ Parkinsons Dis. 8, 44 (2022).
Haumesser, J. K. et al. Subthalamic beta oscillations correlate with dopaminergic degeneration in experimental Parkinsonism. Exp. Neurol. 335, 113513 (2021).
Beudel, M. et al. Oscillatory beta power correlates with akinesia-rigidity in the Parkinsonian subthalamic nucleus. Mov. Disord. 32, 174–175 (2017).
Kühn, A. A. et al. Event-related beta desynchronization in human subthalamic nucleus correlates with motor performance. Brain 127, 735–746 (2004).
Sumarac, S. et al. Interrogating basal ganglia circuit function in people with Parkinson’s disease and dystonia. Elife 12, RP90454 (2024).
Engel, A. K. & Fries, P. Beta-band oscillations—signalling the status quo? Curr. Opin. Neurobiol. 20, 156–165 (2010).
Brown, P. Bad oscillations in Parkinson’s disease. J. Neural Transm. Suppl. 70, 27–30 (2006).
Iskhakova, L. et al. Modulation of dopamine tone induces frequency shifts in cortico-basal ganglia beta oscillations. Nat. Commun. 12, 7026 (2021).
David, F. J., Munoz, M. J. & Corcos, D. M. The effect of STN DBS on modulating brain oscillations: consequences for motor and cognitive behavior. Exp. Brain Res. 238, 1659–1676 (2020).
Neumann, W.-J., Steiner, L. A. & Milosevic, L. Neurophysiological mechanisms of deep brain stimulation across spatiotemporal resolutions. Brain 146, 4456–4468 (2023).
Wang, D. D. et al. Pallidal deep-brain stimulation disrupts pallidal beta oscillations and coherence with primary motor cortex in Parkinson’s disease. J. Neurosci. 38, 4556–4568 (2018).
Piña-Fuentes, D. et al. The characteristics of pallidal low-frequency and beta bursts could help implementing adaptive brain stimulation in the Parkinsonian and dystonic internal globus pallidus. Neurobiol. Dis. 121, 47–57 (2019).
Davidson, B., Milosevic, L., Kondrataviciute, L., Kalia, L. V. & Kalia, S. K. Neuroscience fundamentals relevant to neuromodulation: Neurobiology of deep brain stimulation in Parkinson’s disease. Neurotherapeutics 21, e00348 (2024).
Musacchio, T., Koprich, J. B. & Ip, C. W. Modeling with the A53T α-synuclein model of Parkinson’s disease. Genet. Neurol. Behav. Diet. Parkinson’s. Dis. 2, 677–687 (2020).
Koprich, J. B., Kalia, L. V. & Brotchie, J. M. Animal models of α-synucleinopathy for Parkinson disease drug development. Nat. Rev. Neurosci. 18, 515–529 (2017).
Chung, C. Y., Koprich, J. B., Siddiqi, H. & Isacson, O. Dynamic changes in presynaptic and axonal transport proteins combined with striatal neuroinflammation precede dopaminergic neuronal loss in a rat model of AAV α-synucleinopathy. J. Neurosci. 29, 3365–3373 (2009).
Koprich, J. B. et al. Progressive neurodegeneration or endogenous compensation in an animal model of Parkinson’s disease produced by decreasing doses of alpha-synuclein. PLoS One 6, e17698 (2011).
Koprich, J. B., Johnston, T. H., Reyes, M. G., Sun, X. & Brotchie, J. M. Expression of human A53T alpha-synuclein in the rat substantia nigra using a novel AAV1/2 vector produces a rapidly evolving pathology with protein aggregation, dystrophic neurite architecture and nigrostriatal degeneration with potential to model the pathology of Parkinson’s disease. Mol. Neurodegener. 5, 43 (2010).
Hofman, K. et al. Low β predicts motor output and cell degeneration in the A53T Parkinson’s disease rat model. Brain 148, 4058–4071 (2025).
Musacchio, T. et al. Subthalamic nucleus deep brain stimulation is neuroprotective in the A53T α-synuclein Parkinson’s disease rat model. Ann. Neurol. 81, 825–836 (2017).
Musacchio, T. et al. Temporal, spatial and molecular pattern of dopaminergic neurodegeneration in the AAV-A53T α-synuclein rat model of Parkinson’s disease. Behav. Brain Res. 432, 113968 (2022).
Benhamou, L. & Cohen, D. Electrophysiological characterization of entopeduncular nucleus neurons in anesthetized and freely moving rats. Front. Syst. Neurosci. 8, 7 (2014).
Nim, S. et al. Disrupting the α-synuclein-ESCRT interaction with a peptide inhibitor mitigates neurodegeneration in preclinical models of Parkinson’s disease. Nat. Commun. 14, 2150 (2023).
Hui, S. et al. Mitophagy upregulation occurs early in the neurodegenerative process mediated by α-synuclein. Mol. Neurobiol. 61, 9032–9042 (2024).
Lee, E. J. et al. Reduction of alpha-synuclein oligomers in preclinical models of Parkinson’s disease by electrical stimulation in vitro and deep brain stimulation in vivo. Brain Stimul. 17, 166–175 (2024).
Kondrataviciute, L. et al. Characterization of motor and non-motor features associated with bilateral nigral degeneration due to A53T alpha-synuclein in female rats. Sci. Rep. (2026).
Feingold, J., Gibson, D. J., DePasquale, B. & Graybiel, A. M. Bursts of beta oscillation differentiate postperformance activity in the striatum and motor cortex of monkeys performing movement tasks. Proc. Natl. Acad. Sci. USA 112, 13687–13692 (2015).
Tinkhauser, G. et al. The modulatory effect of adaptive deep brain stimulation on beta bursts in Parkinson’s disease. Brain 140, 1053–1067 (2017).
Yu, Y. et al. Parkinsonism alters beta burst dynamics across the basal ganglia–motor cortical network. J. Neurosci. 41, 2274–2286 (2021).
Little, S., Bonaiuto, J., Barnes, G. & Bestmann, S. Human motor cortical beta bursts relate to movement planning and response errors. PLoS Biol. 17, e3000479 (2019).
Sumarac, S. et al. Clinico-physiological correlates of Parkinson’s disease from multi-resolution basal ganglia recordings. NPJ Parkinsons Dis. 10, 175 (2024).
Tinkhauser, G. et al. Beta burst coupling across the motor circuit in Parkinson’s disease. Neurobiol. Dis. 117, 217–225 (2018).
Avila, I. et al. Beta frequency synchronization in basal ganglia output during rest and walk in a hemiparkinsonian rat. Exp. Neurol. 221, 307–319 (2010).
Polar, C. A., Gupta, R., Lehmkuhle, M. J. & Dorval, A. D. Correlation between cortical beta power and gait speed is suppressed in a Parkinsonian model, but restored by therapeutic deep brain stimulation. Neurobiol. Dis. 117, 137–148 (2018).
Lofredi, R. et al. Subthalamic beta bursts correlate with dopamine-dependent motor symptoms in 106 Parkinson’s patients. NPJ Parkinsons Dis. 9, 2 (2023).
West, T. O. et al. Propagation of beta/gamma rhythms in the cortico-basal ganglia circuits of the Parkinsonian rat. J. Neurophysiol. 119, 1608–1628 (2018).
Pavlides, A., Hogan, S. J. & Bogacz, R. Computational models describing possible mechanisms for generation of excessive beta oscillations in Parkinson’s disease. PLoS Comput. Biol. 11, e1004609 (2015).
Connolly, A. T. et al. Modulations in oscillatory frequency and coupling in Globus Pallidus with increasing Parkinsonian severity. J. Neurosci. 35, 6231–6240 (2015).
Muralidharan, A. et al. Physiological changes in the pallidum in a progressive model of Parkinson’s disease: Are oscillations enough? Exp. Neurol. 279, 187–196 (2016).
Lundqvist, M., Miller, E. K., Nordmark, J., Liljefors, J. & Herman, P. Beta: bursts of cognition. Trends Cogn. Sci. 28, 662–676 (2024).
Bos, M. J. et al. Effect of anesthesia on microelectrode recordings during deep brain stimulation surgery: a narrative review. J. Neurosurg. Anesthesiol. 33, 300–307 (2021).
Janssen, M. L. F. & Bos, M. J. Microelectrode assisted deep brain stimulation: Considerations for anesthesia. Deep Brain Stimulation 4, 13–23 (2024).
Huang, P.-H., Pan, Y.-S., Chen, S.-Y. & Lin, S.-H. Anesthetic effect on the subthalamic nucleus in microelectrode recording and local field potential of Parkinson’s disease. Neuromodulation Technol. Neural Interface 28, 414–424 (2025).
Devlin-Hegedus, J. A., McGain, F., Harris, R. D. & Sherman, J. D. Action guidance for addressing pollution from inhalational anaesthetics. Anaesthesia 77, 1023–1029 (2022).
Aksenov, D. P., Miller, M. J., Dixon, C. J. & Wyrwicz, A. M. The effect of sevoflurane and isoflurane anesthesia on single unit and local field potentials. Exp. Brain Res. 237, 1521–1529 (2019).
Eisinger, R. S. et al. Parkinsonian beta dynamics during rest and movement in the dorsal pallidum and subthalamic nucleus. J. Neurosci. 40, 2859–2867 (2020).
Foffani, G. & Alegre, M. Brain oscillations and Parkinson disease. Handb. Clin. Neurol. 184, 259–271 (2022).
Scherer, M. et al. Single-neuron bursts encode pathological oscillations in subcortical nuclei of patients with Parkinson’s disease and essential tremor. Proc. Natl. Acad. Sci. USA 119, e2205881119 (2022).
Binns, T. S. et al. Shared pathway-specific network mechanisms of dopamine and deep brain stimulation for the treatment of Parkinson’s disease. Nat. Commun. 16, 3587 (2025).
Stein, E. & Bar-Gad, I. Beta oscillations in the cortico-basal ganglia loop during Parkinsonism. Exp. Neurol. 245, 52–59 (2013).
Brazhnik, E., Novikov, N., McCoy, A. J., Cruz, A. V. & Walters, J. R. Functional correlates of exaggerated oscillatory activity in basal ganglia output in hemiparkinsonian rats. Exp. Neurol. 261, 563–577 (2014).
Marceglia, S. et al. Gender-related differences in the human subthalamic area: a local field potential study. Eur. J. Neurosci. 24, 3213–3222 (2006).
Yin, Z. et al. Local field potentials in Parkinson’s disease: a frequency-based review. Neurobiol. Dis. 155, 105372 (2021).
Khan, H. F. et al. Site-specific seeding of Lewy pathology induces distinct pre-motor cellular and dendritic vulnerabilities in the cortex. Nat. Commun. 15, 10775 (2024).
Khanna, P. & Carmena, J. M. Neural oscillations: beta band activity across motor networks. Curr. Opin. Neurobiol. 32, 60–67 (2015).
Bonaiuto, J. J. et al. Laminar dynamics of high amplitude beta bursts in human motor cortex. Neuroimage 242, 118479 (2021).
Ni, Z.-G., Bouali-Benazzouz, R., Gao, D.-M., Benabid, A.-L. & Benazzouz, A. Time-course of changes in firing rates and firing patterns of subthalamic nucleus neuronal activity after 6-OHDA-induced dopamine depletion in rats. Brain Res. 899, 142–147 (2001).
Park, S. E., Song, K.-I., Kim, H., Chung, S. & Youn, I. Graded 6-OHDA-induced dopamine depletion in the nigrostriatal pathway evokes progressive pathological neuronal activities in the subthalamic nucleus of a hemi-Parkinsonian mouse. Behav. Brain Res. 344, 42–47 (2018).
Bezard, E. & Gross, C. E. Compensatory mechanisms in experimental and human Parkinsonism: towards a dynamic approach. Prog. Neurobiol. 55, 93–116 (1998).
Zigmond, M. J., Abercrombie, E. D., Berger, T. W., Grace, A. A. & Stricker, E. M. Compensations after lesions of central dopaminergic neurons: some clinical and basic implications. Trends Neurosci. 13, 290–296 (1990).
Detsch, O., Kochs, E., Siemers, M., Bromm, B. & Vahle-Hinz, C. Differential effects of isoflurane on excitatory and inhibitory synaptic inputs to thalamic neurones in vivo. Br. J. Anaesth. 89, 294–300 (2002).
Ellens, D. J. & Leventhal, D. K. Review: electrophysiology of basal ganglia and cortex in models of Parkinson disease. J. Parkinsons Dis. 3, 241–254 (2013).
Hamani, C. The subthalamic nucleus in the context of movement disorders. Brain 127, 4–20 (2004).
Hamani, C. et al. Insertional effect following electrode implantation: an underreported but important phenomenon. Brain Commun. 6, fcae093 (2024).
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Coordinates (2013).
Fim Neto, A. et al. Subthalamic low beta bursts differ in Parkinson’s disease phenotypes. Clin. Neurophysiol. 140, 45–58 (2022).
Hirschmann, J., Schoffelen, J. M., Schnitzler, A. & van Gerven, M. A. J. Parkinsonian rest tremor can be detected accurately based on neuronal oscillations recorded from the subthalamic nucleus. Clin. Neurophysiol. 128, 2029–2036 (2017).
Cassidy, M. et al. Movement-related changes in synchronization in the human basal ganglia. Brain 125, 1235–1246 (2002).
Underwood, C. F. & Parr-Brownlie, L. C. Primary motor cortex in Parkinson’s disease: Functional changes and opportunities for neurostimulation. Neurobiol. Dis. 147, 105159 (2021).
Verma Rodriguez, A. K., Ramírez-Jarquin, J. O., Rossi-Pool, R. & Tecuapetla, F. Basal ganglia output (entopeduncular nucleus) coding of contextual kinematics and reward in the freely moving mouse. Elife 13, RP98159 (2025).
Brys, I., Nunes, J. & Fuentes, R. Motor deficits and beta oscillations are dissociable in an alpha-synuclein model of Parkinson’s disease. Eur. J. Neurosci. 46, 1906–1917 (2017).
Gerster, M. et al. Separating neural oscillations from aperiodic 1/f activity: challenges and recommendations. Neuroinformatics 20, 991–1012 (2022).
Weinberger, M. et al. Beta oscillatory activity in the subthalamic nucleus and its relation to dopaminergic response in Parkinson’s disease. J. Neurophysiol. 96, 3248–3256 (2006).
Bouthour, W. et al. Biomarkers for closed-loop deep brain stimulation in Parkinson disease and beyond. Nat. Rev. Neurol. 15, 343–352 (2019).
Spencer, K. A. et al. Modulating inhibitory synaptic plasticity to restore basal ganglia dynamics in Parkinson’s disease. Brain 148, 2299–2305 (2025).
Feldmann, L. K. et al. Subthalamic beta band suppression reflects effective neuromodulation in chronic recordings. Eur. J. Neurol. 28, 2372–2377 (2021).
Temperli, P. et al. How do Parkinsonian signs return after discontinuation of subthalamic DBS? Neurology 60, 78–81 (2003).
Kuhn, A. A. et al. High-frequency stimulation of the subthalamic nucleus suppresses oscillatory activity in patients with Parkinson’s disease in parallel with improvement in motor performance. J. Neurosci. 28, 6165–6173 (2008).
Tai, C.-H. Subthalamic burst firing: a pathophysiological target in Parkinson’s disease. Neurosci. Biobehav. Rev. 132, 410–419 (2022).
Gage, G. J. et al. Surgical implantation of chronic neural electrodes for recording single unit activity and electrocorticographic signals. J. Vis. Exp. 60, 3565 (2012).
Chin, G. D. & Hutchison, W. D. Effects of cobalt and bicuculline on focal microstimulation of rat pallidal neurons in vivo. Brain Stimul. 1, 134–150 (2008).
McConnell, G. C., So, R. Q., Hilliard, J. D., Lopomo, P. & Grill, W. M. Effective deep brain stimulation suppresses low-frequency network oscillations in the basal ganglia by regularizing neural firing patterns. J. Neurosci. 32, 15657–15668 (2012).
Zhang, K. K., Matin, R., Gorodetsky, C., Ibrahim, G. M. & Gouveia, F. V. Systematic review of rodent studies of deep brain stimulation for the treatment of neurological, developmental and neuropsychiatric disorders. Transl. Psychiatry 14, 186 (2024).
Schor, J. S. & Nelson, A. B. Multiple stimulation parameters influence efficacy of deep brain stimulation in Parkinsonian mice. J. Clin. Investig. 129, 3833–3838 (2019).
Mottaghi, S., Buchholz, O. & Hofmann, U. G. Systematic evaluation of DBS parameters in the hemi-Parkinsonian rat model. Front. Neurosci. 14, 561008 (2020).
Barbosa, E. H., Vallim, J. H., Lachat, J.-J. & de Castro, V. L. S. S. Assessments of motor abnormalities on the grid-walking and foot-fault tests from undernutrition in Wistar rats. J. Mot. Behav. 48, 5–12 (2016).
Chao, O. Y., Pum, M. E., Li, J.-S. & Huston, J. P. The grid-walking test: assessment of sensorimotor deficits after moderate or severe dopamine depletion by 6-hydroxydopamine lesions in the dorsal striatum and medial forebrain bundle. Neuroscience 202, 318–325 (2012).
Jain, A., Nandakumar, K. & Ross, A. Score normalization in multimodal biometric systems. Pattern Recognit. 38, 2270–2285 (2005).
Nath, T. et al. Using DeepLabCut for 3D markerless pose estimation across species and behaviors. Nat. Protoc. 14, 2152–2176 (2019).
Acknowledgements
This study was funded by Natural Sciences and Engineering Council (NSERC) RGPIN-2022-05181 (L.M.), the Canadian Institute for Health Research (CIHR) PJT 191880 (L.M., S.K.), Canadian Foundation for Innovation (CFI) Project 42608 (L.M., S.K., T.V.) and the R.R. Tasker Chair in Stereotactic and Functional Neurosurgery (S.K.).
Author information
Authors and Affiliations
Contributions
Conceptualization: L.K., M.K., L.V. K., S.K.K.; Methodology: L.K., M.K., W.D.H.; Formal analysis: L.K., M.K., I.S., W.D.H., S.S., L.Z.; Investigation: L.K.; Resources: S.K.K., L.V.K., L.M., T.V., C.H., X.L.; Data curation: L.K., C.T., S.J., H.C., Y.H., S.B.; Writing–original draft: L.K.; Writing–review & editing: L.K., M.K., S.K.K., L.V.K., W.D.H.; Visualization: L.K.; Supervision: S.K.K., L.V.K., T.V., L.M; Project administration: S.K.K., L.V.K.; Funding acquisition: S.K.K., L.V.K., T.V., L.M. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kondrataviciute, L., Kapadia, M., Skelin, I. et al. Cortical and basal ganglia beta oscillations and frequency-dependent DBS effects in the A53T Parkinson’s disease rat model. npj Parkinsons Dis. (2026). https://doi.org/10.1038/s41531-026-01304-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-026-01304-z